Summary
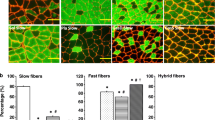
Rats were used in this study to determine the time course of conversion of muscle fiber types. The right or left gastrocnemius muscle was removed thereby causing an overload on the ipsilateral soleus and plantaris muscles. The contralateral limb served as a control. The type II to type I fiber conversion was followed histochemically in the soleus and plantaris muscles for one to six weeks following surgery. Muscle sections were stained for myofibrillar actomyosin ATPase and NADH tetrazolium reductase. The type I population in the soleus muscle was 99.3% six weeks after synergist removal. The plantaris muscle underwent a two fold increase in the percentage of type I fibers after six weeks. Transitional fibers were prominent in the plantaris muscle and reached their peak at 4% (P<0.05) of the total population, four weeks after surgery.
Similar content being viewed by others
References
Armstrong R, Marum P, Tulison P, Saubert IVC (1979) Acute hypertrophic response of skeletal muscle to removal of synergists. J Appl Physiol 46:835–842
Baldwin K, Valdez V, Herrick R, MacIntosh A, Roy R (1982) Biochemical properties of overloaded fast-twitch skeletal muscle. J Appl Physiol 52:467–472
Baldwin K, Roy R, Sacks R, Blanco C, Edgerton V (1984) Relative independence of metabolic enzymes and neuromuscular activity. J Appl Physiol 56:1602–1607
Gollnick P, Timson B, Moore R, Riedy M (1981) Muscular enlargement and number of fibers in skeletal muscles of rats. J Appl Physiol 50:936–943
Gollnick P, Parsons D, Oakley C (1983) Differentiation of fiber types in skeletal muscle from the sequential inactivation of myofibrillar actomyosin ATPase during acid preincubation. Histochemistry 77:543–555
Gollnick P, Matoba H (1984) Identification of fiber types in rat skeletal muscle based on the sensitivity of myofibrillar actomyosin ATPase to copper. Histochemistry 81:379–383
Green H, Klug G, Reichmann H, Seedorf U, Wiehrer W, Pette D (1984) Excrcise-induced fibre type transitions with regard to myosin, parvalbumin, and sarcoplasmic reticulum in muscles of the rat. Pfluegers Arch 400:432–438
Guth L, Yellin H (1971) The dynamic nature of the so-called “Fiber-Types” of mammalian skeletal muscle. Exp Neurol 31:277–300
Ianuzzo C, Chen V (1979) Metabolic character of hypertrophied rat muscle. J Appl Physiol 46:738–742
Ianuzzo C, Gollnick P, Armstrong R (1976) Compensatory adaptations of skeletal muscle fiber types to a long term functional overload. Life Sci 19:1517–1524
Karpati G, Engel W (1968) Correlative histochemical study of skeletal muscle after suprasegmental denervation, peripheral nerve section and skeletal fixation. Neurology 18:681–692
Kugelberg e (1976) Adaptive transformation of rat soleus motor neurons during growth: histochemistry and contraction speed. J Neurol Sci 27:269–289
Martin A, Rabinowitz M, Blough R, Prior G, Zak R (1977) Measurements of half-life of rat cardiac myosin heavy chain with leucyl-tRNA used as a precursor pool. J Biol Chem 252:3422–3429
Matoba H, Gollnick P (1984) Influence of ionic composition, buffering agent, and pH on the histochemical demonstration of myofibrillar actomyosin ATPase. Histochemistry 80:609–614
Noble E, Dabrowski B, Ianuzzo C (1983) Myosin transformation in hypertrophied rat muscle. Pfluegers Arch 396:260–262
Novikoff A, Shin W, Drucker J (1961) Mitochondrial localization of oxidative enzymes: staining results with two tetrazolium salts. J Biophys Biochem Cytol 9:47–61
Pette D, Schnez U (1977a) Coexistance of fast and slow type myosin light chains in single muscle fibers during transformation as induced by long term stimulation. FEBS Lett 83:128–130
Pette D, Schnez U (1977b) Myosin light chain patterns of individual fast and slow twitch fibers in rabbit muscle. Histochemistry 54:97–107
Pette D, Muller W, Leisner E, Vrbova G (1976) Time dependent effects on contractile properties, fibre population, myosin light chains and enzymes of energy metabolism in intermittently and continuously stimulated fast twitch muscles of the rabbit. Pfluegers Arch 364:103–112
Rubinstein N, Kelly A (1978) Myogenic and neurogenic contributions to the development of fast and slow twitch muscles in rat. Dev Biol 62:473–485
Schiaffino S, Pierobon-Bormioli S (1973) Adaptive changes in developing rat skeletal muscles in response to functional overload. Exp Neurol 40:126–137
Timson B, Bowlin B, Dudenhoeffer G, George J (1985) Fiber number, area, and composition of mouse soleus muscle following enlargement. J Appl Physiol 58:619–624
Vaughn H, Aziz-Ullah, Goldspink G, Nowell N (1974) Sex and stock differences in the histochemical myofibrillar adenosine triphosphatase reaction in the soleus muscle of the mouse. J Histochem Cytochem 22:155–159
Zak R, Martin A, Prior G, Rabinowitz M (1977) Comparison of turnover of several myofibrillar proteins and critical evaluation of double isotope method. J Biol Chem 252:3430–3435
Author information
Authors and Affiliations
Additional information
This research was funded in part by grants from The Graduate School at Washington State University, and The Society of the Sigma Xi
Rights and permissions
About this article
Cite this article
Oakley, C.R., Gollnick, P.D. Conversion of rat muscle fiber types. Histochemistry 83, 555–560 (1985). https://doi.org/10.1007/BF00492460
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00492460