Abstract
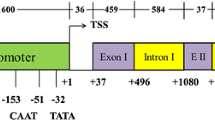
Two genomic clones (λUbi-1 and λUbi-2) encoding the highly conserved 76 amino acid protein ubiquitin have been isolated from maize. Sequence analysis shows that both genes contain seven contiguous direct repeats of the protein coding region in a polyprotein conformation. The deduced amino acid sequence of all 14 repeats is identical and is the same as for other plant ubiquitins. The use of transcript-specific oligonucleotide probes shows that Ubi-1 and Ubi-2 are expressed constitutively at 25°C but are inducible to higher levels at elevated temperatures in maize seedlings. Both genes contain an intron in the 5′ untranslated region which is inefficiently processed following a brief, severe heat shock. The transcription start site of Ubi-1 has been determined and a transcriptional fusion of 0.9 kb of the 5′ flanking region and the entire 5′ untranslated sequence of Ubi-1 with the coding sequence of the gene encoding the reporter molecule chloramphenicol acetyl transferase (CAT) has been constructed (pUBI-CAT). CAT assays of extracts of protoplasts electroporated with this construct show that the ubiquitin gene fragment confers a high level of CAT expression in maize and other monocot protoplasts but not in protoplasts of the dicot tobacco. Expression from the Ubi-1 promoter of pUBI-CAT yields more than a 10-fold higher level of CAT activity in maize protoplasts than expression from the widely used cauliflower mosaic virus 35S promoter of a 35S-CAT construct. Conversely, in tobacco protoplasts CAT activity from transcription of pUBI-CAT is less than one tenth of the level from p35S-CAT.
Similar content being viewed by others
References
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K: Current Protocols in Molecular Biology. John Wiley, New York (1988).
Ball E, Karlik CC, Beall CJ, Saville DL, Sparrow JC, Bullard B, Fyrberg EA: Arthrin, a myofibrillar protein of insect flight muscle is an actin-ubiquitin conjugate. Cell 51: 221–228 (1987).
Barsoum J, Varshavsky A: Preferential localization of variant nucleosomes near the 5′ end of the mouse dihydrofolate reductase gene. J Biol Chem 260: 7688–7697 (1985).
Bond U, Schlesinger MJ: The chicken ubiquitin gene contains a heat shock promoter and expresses an unstable mRNA in heat-shocked cells. Mol Cell Biol 6: 4602–4610 (1986).
Burke TJ, Callis J, Vierstra RD: Characterization of a polyubiquitin gene fromArabidopsis thaliana. Mol Gen Genet 213: 435–443 (1988).
Callis J, Vierstra RD: Ubiquitin and ubiquitin genes in higher plants. Oxford Surv Plant Mol Cell Biol 6: 1–30 (1989).
Callis J, Fromm M, Walbot V: Introns increase gene expression in cultured maize cells. Genes Devel 1: 1183–1200 (1987).
Callis J, Fromm M, Walbot V: Heat inducible expression of a chimeric maize hsp70-CAT gene in maize protoplasts. Plant Physiol 88: 965–968 (1988).
Callis J, Raasch JA, Vierstra RD: Ubiquitin extension proteins ofArabidopsis thaliana. J Biol Chem 265: 12486–12493 (1990).
Christensen AH, Quail PH: Sequence analysis and transcriptional regulation by heat shock of polyubiquitin transcripts from maize. Plant Mol Biol 12: 619–632 (1989).
Czarnecka E, Nagao RT, Key JL, Gurley WB: Characterization of Gmhsp26-A, a stress gene encoding a divergent heat shock protein of soybean: heavy metal induced inhibition of intron processing. Mol Cell Biol 8: 1113–1122 (1988).
Dekeyser R, Claes B, Marichal M, VanMontagu M, Caplan A: Evaluation of selectable markers for rice transformation. Plant Physiol 90: 217–223 (1989).
Devereux J, Haeberli P, Smithies O: A comprehensive set of sequence analysis programs for the VAX: Nucl Acids Res 12: 387–395 (1984).
Finley D, Bartel B, Varshavsky A: The tails of ubiquitin precursors are ribosomal proteins whose fusion to ubiquitin facilitates ribosome biogenesis. Nature 338: 394–401 (1989).
Finley D, Ozkaynak E, Varshavsky A: The yeast polyubiquitin gene is essential for resistance at high temperatures, starvation and other stresses. Cell 48: 1035–1046 (1987).
Fried VA, Smith HT, Hildebrandt E, Weiner K: Ubiquitin has instrinsic proteolytic activity: Implications for cellular regulation. Proc Natl Acad Sci USA 84: 3685–3689 (1987).
Fromm M, Taylor LP, Walbot V: Expression of genes transferred into monocot and dicot plant cells by electroporation. Proc Natl Acad Sci USA 82: 5824–5828 (1985).
Fromm M, Callis J, Taylor LP, Walbot V: Electroporation of DNA and RNA into plant protoplasts. Meth Enzymol 153: 351–366 (1987).
Gausing K, Barkardottir R: Structure and expression of ubiquitin genes in higher plants. Eur J Biochem 158: 57–62 (1986).
Gilmour DS, Thomas GH, Elgin SCR:Drosophila nuclear proteins bind to regions of alternating C and T residues in gene promoters. Science 245: 1487–1490 (1989).
Hanley BA, Schuler MA: Plant intron sequences: evidence for distinct groups of introns. Nucl Acids Res 16: 7159–7176 (1988).
Henikoff S: Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene 28: 351–359 (1984).
Hershko A: Ubiquitin-mediated protein degradation. J Biol Chem 263: 15237–15240 (1988).
Jabben M, Shanklin J, Vierstra RD: Red light-induced accumulation of ubiquitin-phytochrome conjugated in both monocots and dicots. Plant Physiol 90: 380–384 (1989).
Jentsch S, McGrath JP, Varshavsky A: The yeast DNA repair gene RAD 6 encodes a ubiquitin-conjugating enzyme. Nature 329: 131–134 (1987).
Keith B, Chua N-H: Monocot and dicot pre-mRNAs are processed with different efficiencies in transgenic tobacco. EMBO J 5: 2149–2425 (1986).
Maniatis T, Fritsch EF, Sambrook J: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY (1982).
Pelham HRB: A regulatory upstream promoter element inDrosophila HSP70 heat-shock gene. Cell 30: 517–528 (1982).
Rechsteiner M: Ubiquitin-mediated pathways for intracellular proteolysis. Annu Rev Cell Biol 3: 1–30 (1987).
Rechsteiner M: Natural substrates of the ubiquitin proteolytic pathway. Cell 66: 615–618 (1991).
Redman KL, Rechsteiner M: Identification of the long ubiquitin extension as ribosomal protein S27a. Nature 338: 438–440 (1989).
Schlesinger M, Bond U: Ubiquitin genes. Oxford Surv Eukaryotic Genes 4: 77–89 (1987).
Shanklin J, Jabben M, Vierstra RD: Red light-induced formation of ubiquitin-phytochrome conjugates: Identification of possible intermediates of phytochrome degradation. Proc Natl Acad Sci USA 84: 359–363 (1987).
Sharp PM, Li W-H: Ubiquitin genes as a paradigm of concerted evolution of tandem repeats. J Mol Evol 25: 58–64 (1987).
Sullivan TD, Christensen AH, Quail PH: Isolation and characterization of a maize chlorophyll a/b binding protein gene that produces high levels of mRNA in the dark. Mol Gen Genet 215: 431–440 (1989).
Winter J, Wright R, Duck N, Gasser C, Fraley R, Shah D: The inhibition of petunia hsp70 mRNA processing during CdCl2 stress. Mol Gen Genet 211: 315–319 (1988).
Yost HJ, Lindquist S: RNA splicing is interrupted by heat shock and is rescued by heat shock protein synthesis. Cell 45: 185–193 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Christensen, A.H., Sharrock, R.A. & Quail, P.H. Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol Biol 18, 675–689 (1992). https://doi.org/10.1007/BF00020010
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00020010