Abstract
Food protein-induced enterocolitis syndrome (FPIES) is a non-IgE-mediated food hypersensitivity with usual onset in infancy. The most common FPIES triggers are cow milk, soy and rice; in addition, oats, vegetables, egg, poultry and seafood have been reported. In the acute form, when food is ingested on an intermittent basis or following a period of avoidance, FPIES presents with profuse vomiting within 1–3 h after ingestion, occasionally accompanied by diarrhea and dehydration. In the chronic form, when food is ingested on a regular basis, FPIES presents with intermittent vomiting, diarrhea, weight loss and failure to thrive. FPIES is diagnosed based on history and typical symptoms, which improve with food avoidance, and exclusion of other etiologies. Oral food challenge remains the gold standard for FPIES diagnosis. Most CM or soy FPIESs resolve within the first 3–5 years; solid food FPIES or FPIES associated with positive food-specific IgE may have a more protracted course. The prevalence of FPIES is unknown.
Similar content being viewed by others
Introduction
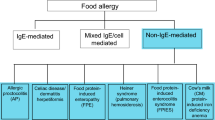
Non-IgE and mixed pathophysiology gastrointestinal food allergic disorders are estimated to account for 40 % of cow’s milk allergy in infants and young children [1]. They encompass food protein-induced enterocolitis syndrome (FPIES), allergic proctocolitis, enteropathy and eosinophilic gastrointestinal disorders. Most are self-limiting and resolve by 3–5 years of age. FPIES is an under-recognized and frequently misdiagnosed non-IgE-mediated food hypersensitivity, manifesting as a severe, repetitive vomiting within 1–3 h following food ingestion, lethargy, pallor and/or diarrhea, usually within 5–10 h following food ingestion. Classic allergic symptoms of the skin, such as urticaria, pruritus or angioedema, and of the respiratory tract, such as cough, wheezing or shortness of breath, are absent [2•]. In the US, the most common food triggers are cow milk (CM), soybean and rice [3]. Symptoms induced by milk or soy typically begin in the first month of life in association with failure to thrive and may progress to acidemia and shock. FPIES to cereal grains, vegetables and meats usually starts after these solid foods have been introduced into the infant’s diet, whereas FPIES to fish or shellfish may have onset in older children or adults. Despite the potential severity of acute reactions, FPIES can be considered self-limiting as avoidance of the incriminating allergen(s) leads to resolution of symptoms.
Defining FPIES
Allergic reactions to foods affecting the gastrointestinal tract have been known since ancient times. Hippocrates noted that cow’s milk caused gastrointestinal symptoms as well as urticaria, and that some infants fed cow’s milk developed prolonged diarrhea, vomiting and failure to thrive that resolved only after removal of cow’s milk from their diet. Rubin reported intestinal bleeding due to cow’s milk allergy in newborns [4]. Gryboski and Powell described infants presenting in the first 6 weeks of life with recurrent vomiting, bloody diarrhea and abdominal distension while being fed with cow’s milk-based formula [5, 6]. They appeared dehydrated and severely ill. Sepsis evaluations were negative, but they improved with intravenous fluids or hydrolyzed casein-based formula, but not with soy-based formula. Reintroduction of cow’s milk-based formula resulted in recurrence of severe emesis, diarrhea and elevation of the peripheral blood neutrophil count. Powell characterized major features of the disorder and established criteria for the diagnosis of cow’s milk-induced enterocolitis and a standard challenge protocol [7]. Reports of large series of infants with food protein-induced enterocolitis by Sicherer et al. [8] (16 patients) and Burks et al. [9] (43 patients) further characterized clinical features and modified food challenge protocols. More recent reports have identified various solid foods as triggers for FPIES, including oat, wheat, vegetables and shellfish in adults [3, 10–18].
Prevalence of FPIES
The prevalence of FPIES in the US is not known; generally, FPIES is considered to be an uncommon food allergy. However, in a large Israeli population-based birth cohort FPIES incidence was 0.34 % (44/13,019); by comparison, the incidence of IgE-mediated CM allergy in this population was 0.5 % [19••]. The Israeli study suggested that FPIES may be more common than previously appreciated. It is likely that mild phenotypes of CM and soy FPIES are not being diagnosed as FPIES and are managed empirically with formula changes in the first year of life with resolution of symptoms.
Risk factors for development of FPIES include cesarean delivery, male gender and FPIES to another food [19••]. FPIES is associated with atopy, with 30 % of FPIES patients having a personal history of atopic disease, 40–80 % reporting family history of atopic disease and 20 % family history of food allergy [3, 8]. Our own unpublished data suggest that early introduction of CM may be a risk factor for FPIES. In addition, although egg is not a common cause of FPIES, when it was introduced at 5.5 months as a control allergen, it caused FPIES symptoms in 30 % of the infants with milk or soy FPIES [20].
Wheat is also not a common trigger; however, wheat FPIES prevalence is likely to be modified by the common practice of delaying wheat introduction in infants with rice or oat FPIES past 12 months of age. These observations and the optimal timing of the food introductions need to be validated by prospective studies in larger populations.
Breastfeeding appears to have a protective role against FPIES as infants exclusively breast-fed without formula supplementation are asymptomatic until direct feeding with the offending food. CM-FPIES to the mother’s milk in exclusively breastfed infants is rare, with only four case reports to date [3, 21, 22].
FPIES Manifestations
FPIES manifestations depend on the frequency and the dose of food ingested. FPIES may present in either acute or chronic form. It is important to point out that there are no classic allergic symptoms from the skin (such as itching, hives, swelling) or respiratory tract (wheezing, cough, sneezing) in either form of FPIES (Table 1). Chronic FPIES occurs with frequent ingestion of the trigger food and typically presents in a formula-fed infant in the first weeks of life, with symptoms of intermittent emesis, watery or mucous diarrhea, poor weight gain and dehydration [5–7]. These symptoms are non-specific and have a very broad differential diagnosis including infection, metabolic disorders, structural defects and inflammatory gastrointestinal disorders, among others. Acute FPIES occurs with intermittent ingestion of the trigger food, with symptoms’ onset 1–3 h after ingestion, typically with somewhat more severe presentation than chronic FPIES. In addition to profuse emesis (which may be projectile and occur up to 10–20 times) and dehydration, infants may develop lethargy, pallor, hypotension, hypothermia and methemoglobinemia [5, 6, 23–25]. Typically, avoidance of the culprit food for several days results in the resolution of symptoms; upon subsequent food ingestions FPIES symptoms usually re-occur within 1–3 h. In children with ongoing gastrointestinal symptoms that persist despite extensive dietary eliminations or while the child is exclusively fed with an amino acid-based formula, further evaluation is necessary to confirm the diagnosis of FPIES and to avoid misdiagnosing other conditions (e.g., metabolic disorders, eosinophilic gastroenteropathies or gastrointestinal inflammatory diseases) as chronic FPIES.
Food Triggers and Age of Onset
In the US, the most common triggers are CM and soy formulas and rice in young infants [3, 5–7, 26]. In Israel, Australia and Italy, soy FPIES is rare [19••, 24, 25]. In infants fed with the infant formula, CM and soy FPIES usually starts within the first few months of life. When the direct introduction of milk or soy is delayed, e.g., because of breastfeeding, initial reactions may begin after the first year of age. Solid food FPIES includes grains (rice, oats, barley, corn), meat and poultry (beef, chicken, turkey), egg white, vegetables (white potato, sweet potato, squash), fruit (tomato) and legumes (peanut, green pea, lentil, string bean) [27]. It usually occurs within several days after the solid foods are introduced. It is not uncommon for the parents to report that the solid food was initially tolerated fine for several sequential feedings; however, when feedings were interrupted for several days or weeks, subsequent ingestion resulted in acute FPIES reaction. FPIES to seafood (fish, crustaceans, molluscs) may occur in older children and adults [28].
Diagnosis
The NIAID Food Allergy Guidelines recommend using the medical history and oral food challenge (OFC) to establish a diagnosis of FPIES [1]. When the history indicates that infants or children have had experienced hypotensive episodes or multiple reactions to the same food, a diagnosis may be based on a convincing history and absence of symptoms when the causative food is eliminated from the diet. The original diagnostic criteria as proposed by Powell included: (1) exposure to the incriminating food elicits repetitive vomiting and/or diarrhea within 4 h, without any other cause for the symptoms; (2) symptoms are limited to the gastrointestinal tract; (3) avoidance of the offending protein from the diet results in resolution of symptoms; (4) a standardized OFC or isolated re-exposure elicits the typical symptoms [7]. An International Working Group on Consensus Guidelines for FPIES has been formed under the auspices of the AAAAI Adverse Reactions to Food Committee and the International Association of Food Protein-induced Enterocolitis in 2013. The Expert Panel is working on evidence-based guidelines for the diagnosis and management of FPIES to improve the care provided for patients with FPIES.
There are no pathognomomic laboratory or radiographic findings specific to FPIES. An elevated white blood count with left shift (peaking at 6 h after food ingestion and after the onset of the first symptoms) and methemoglobinemia following food ingestion support diagnosis of FPIES.
Food-specific IgE and/or skin prick testing may be performed to provide complete evaluation for food sensitization, particularly when considering a food challenge. Though the majority of patients with FPIES have undetectable serum IgE at the time of diagnosis, 18–30 % of FPIES patients may develop IgE mediated food sensitivity to the same food at some point during their course, with some developing immediate type symptoms of classic IgE-mediated food allergy [8, 24, 29].
Gastric juice leukocytosis at 3 h after a food challenge and atopy patch testing with fresh foods (APT) have been investigated in FPIES patients [30–33]. However, the results are equivocal, and therefore their diagnostic utility remains unclear and they are not recommended for routine evaluation of FPIES [1, 26].
Delays in Diagnosis/Misdiagnosis in FPIES
Infants with FPIES often present with multiple reactions and undergo extensive evaluations before the diagnosis of FPIES is considered [3, 24, 25]. Diagnostic tests include sepsis workup, abdominal imaging, electrocardiograms and electroencephalograms. In one case presenting with severe abdominal distension, an exploratory laparoscopy was performed [34]. Delays in diagnosis of infants with FPIES may be due to the non-specific nature of the symptoms FPIES patients are experiencing, lack of classic allergic skin and respiratory reactions, broad differential diagnosis (anaphylaxis and allergic gastrointestinal disorders, infections, metabolic disease, gastrointestinal obstruction, inflammatory bowel disease, neurologic disorders), relative lack of knowledge among physicians and, in the case of FPIES to solid foods, the perception that grains and vegetables are hypoallergenic as they rarely cause IgE-mediated food allergy [35, 36].
Oral Food Challenge in FPIES
The physician-supervised OFC remains the gold standard for an initial diagnosis of FPIES as well as for monitoring the resolution of FPIES [1]. The OFC is usually done in an open manner under physician supervision in a facility appropriately equipped for managing dehydration and allergic reactions (Table 2) [37]. In our practice, a peripheral intravenous line is placed before the challenge to secure immediate access for rapid intravenous fluid rehydration. The baseline complete blood count with differential is obtained immediately before the challenge [2•]. The challenge food amount is based on the food protein content (maximum 3 g of protein or 10 g of food or 100 ml of liquid food) and administered in three equal portions over 30 min, followed by a minimum 4 h of observation prior to discharge (Table 2). Patients who tolerated the challenge without any symptoms are usually discharged 4 h after eating the last portion of the challenge food; a post-challenge blood sample is obtained for a complete blood count with differential at 4 h. Patients who reacted to the challenge are usually discharged when 6 h have passed since ingestion of the food and their symptoms have resolved. A post-challenge blood sample is obtained for a complete blood count with differential at 6 h post challenge to calculate the increase in peripheral blood neutrophils, which is one of the major criteria for challenge positivity, as proposed by Powell (Table 2) [7]. In recent experience, diarrhea is seen in 20–40 % of the patients [24, 25, 38]. In addition, the magnitude of the neutrophil count increase during the challenge seems to be less than 3,500/cu mm. These observations suggest that criteria for challenge positivity should be updated and revised based on observations from a large number of food challenges. The initial criteria were established based on the outcomes of 14 challenges in 9 infants with median age of 36 days who had just recently removed the offending food from their diet and who were clearly at the peak of their disease [6, 7]. In our clinical practice, we do not perform challenges in infants. The challenge is usually delayed by 12–18 months from the most recent FPIES-like reaction; therefore, the magnitude of the inflammatory response could be lower. Cooperative efforts among the centers providing the challenges for patients with FPIES are needed to standardize the challenge criteria.
Management
Management of acute and chronic FPIES consists of dietary food elimination, supportive therapies for acute and chronic FPIES on presentation, and providing an emergency treatment plan for episodes due to accidental exposures [2•]. Children are re-evaluated periodically and oral food challenges are repeated every 12–18 months to evaluate for resolution (Fig. 1). This is an empiric approach and shorter intervals between the food challenges may be appropriate for individual patients.
Infants with suspected FPIES to cow or soy milk protein should strictly avoid all forms of the inciting food, including baked and processed foods. They may either exclusively breastfeed or start a casein hydrosylate-based formula. Ten to 20 % may require an amino acid-based formula [3, 39]. Infants with chronic FPIES usually return to their usual state of health within 3–10 days of switching to a hypoallergenic formula; infants with acute FPIES reactions generally recover rapidly with rehydration alone [6, 7].
For the rare cases of FPIES in breastfed infants, breastfeeding mothers should eliminate the suspected trigger food(s) from her diet. For infants with FPIES to one solid food, in our practice we recommended delaying introduction of grain, legumes, poultry, as well as cow and soy milk until the first year because of the high rate of FPIES to multiple foods [2•, 40].
On presentation, the first line in management of FPIES is vigorous intravenous hydration, usually a 10–20 ml/kg bolus of normal saline, repeated as needed. The second line includes a single dose of intravenous methylprednisolone (dosed at 1 mg/kg, with a maximum of 60–80 mg) in order to decrease presumed cell-mediated inflammation [37]. A recent small case series highlighted the effectiveness of intravenous ondansetron for stopping emesis during FPIES OFC [41•]. In severe reactions, patients may need other supportive therapies including supplemental oxygen, vasopressors, bicarbonate and methylene blue for methemoglobinemia. Epinephrine and antihistamines do not appear to stop emesis in acute FPIES [28].
Emergency treatment plans outlining clinical features and management of acute reactions should be provided to patients with FPIES (a template can be accessed on the International Association for Food Protein Enterocolitis website, http://iaffpe.org/docs/Emergency_Plan.pdf). Mild reactions may be managed with careful oral rehydration at home. Infants with more severe reactions require resuscitation in the emergency department or inpatient unit (see “Acute episode” above).
Natural History of FPIES
Age of resolution of FPIES varies widely depending on the type of food, the country and the population studied. For CM-FPIES, resolution rates by age 3 years range from about 50 % in a US referral population to 90 % in an Israeli population-based birth cohort [3, 19••]. For soy FPIES, resolution rates range from 25 % by age 3 years in a US referral population [5] to 90 % by age 10 months in a Korean cohort [3, 19••, 38]. For solid food FPIES, a retrospective study in Italy reported a resolution rate of 48 % by 29 months of age [25]. In US referral populations, resolution rates range from 40 % for rice, 66 % for oats and 67 % for vegetables by 3 years of age [3]. These differences could be explained by the more severe FPIES phenotype reported from the referral allergy clinic in the US compared to the milder phenotypes among the children with FPIES from a general population.
Taking into account the average age of resolution, in our practice we usually recommend OFC to trigger foods every 12–18 months after the first year of life along with testing to evaluate for the development of food-specific IgE [2•, 8] (Fig. 1). Though the majority of FPIES patients have negative food-specific serum IgEs and skin prick tests at diagnosis, those with positive IgE tests tend to have a more protracted course and are at risk for developing IgE-mediated food allergy [8].
Pathophysiology of FPIES
T Lymphocyte-mediated Immune Response
The mechanisms underlying FPIES remain poorly characterized. FPIES is often considered to be a T cell-mediated disorder; however, few studies have investigated T cells in FPIES. There is some evidence of T cell proliferation upon stimulation with food antigens; however, the stimulation index is not consistently different from control, non-allergic subjects [42–47]. T-cell activation by food allergens may mediate local intestinal inflammation through the release of proinflammatory cytokines, e.g., TNF-α and IFN-γ, causing increased intestinal permeability and fluid shift that is thought to underlie the pallor, poor perfusion, hypothermia and methemoglobinemia [48]. Local inflammation may be mediated by activated peripheral mononuclear cells, increased TNF-α and decreased expression of TGF-β receptors in the intestinal mucosa [44, 49–51]. However, baseline antigen absorption is normal in FPIES [52]. Acute FPIES reactions seem to be associated with a Th2 skewing of the T cell cytokine profile, in keeping with the classical IgE-mediated allergic reactions to foods. An increase in IL-4 and decrease in IFN-γ expression in peripheral blood T cells has been shown after a positive oral challenge. After tolerance had been acquired, IFN-γ and IL-10 increased significantly [48].
Neutrophils, Platelets and Eosinophils
Powell reported leukocytosis with a left shift as a common finding for patients presenting with acute FPIES and included it as one of the diagnostic criteria. In the Powell study, peripheral blood neutrophil counts were elevated in all positive challenges, peaking at 6 h with a mean increase of 9,900 cells/μl [6, 7]. These results were confirmed by subsequent studies [8, 9]. Neutrophils have also been found in stool mucous of FPIES patients and in the gastric juice aspirate. This increase in peripheral neutrophils is likely due to the acute inflammatory reaction of the gastrointestinal tract leading to secretion of different cytokines (TNF-α) and chemokines.
Thrombocytosis was recorded in 63 % of episodes in a report from Australia [24]. One possible explanation for this acute thrombocytosis is a response to epinephrine induced by stress, which can shift platelets from the spleen into the circulation. The potential active contribution of neutrophils and platelets in FPIES pathophysiology requires further investigation.
Eosinophils reside in the GI tract, except in the esophageal squamous mucosa. Eosinophil accumulation in the GI tract is commonly found in many GI disorders, including eosinophilic gastroenteropathies, food-induced proctocolitis as well as classic IgE-mediated food allergy, inflammatory bowel diseases and gastroesophageal reflux [53]. Eosinophil inflammation has been found in intestinal biopsies from infants with FPIES [50]. In FPIES with chronic diarrhea, eosinophils and Charcot-Leyden crystals were detected with Hansel’s stain in stool samples. Blood eosinophilia may be present in conjunction with or independent of eosinophil accumulation in the GI tract. These findings, however, are not specific for FPIES.
Humoral Immune Responses
Humoral responses are poorly characterized in FPIES. Since FPIES is a GI-food allergy disorder and considering the importance of IgA in mucosal immunity, impairment in IgA production or secretion might play a role in FPIES pathophysiology. This is supported by the epidemiological observations that FPIES occurs predominantly in cow’s milk-formula-fed infants, and it is extremely rare in exclusively breastfed infants [22]. It is not clear how exclusive breastfeeding prevents FPIES, but it has been hypothesized that breast milk IgA, either alone or as a complex with secreted antigens, may play a protective role [54]. However, there are only a few, small studies in which the potential role of IgA in FPIES has been examined [20, 42]. In a study by McDonald et al. [20], children with FPIES to cow’s milk had comparable levels of serum milk-specific IgA levels to children with FPIES to other foods. Similar results were observed in the infants with FPIES to egg and soy. These studies also reported near absence of serum allergen-specific IgG4 in FPIES. IgG4 antibodies fix complement poorly and have a protective role in competing with other antibody subclasses that activate complement. The relative lack of IgG4 in FPIES patients may be involved in the pathogenesis of this disorder. However, these are findings from the peripheral blood and not from the GI tract. Jejunal biopsies reveal increased numbers of IgM- and IgA-containing plasma cells [50, 55], but it is unclear how this increase affects antibody secretion in the GI tract.
Systemic food protein-specific IgE antibody responses are usually absent in FPIES [3, 8]. However, if skin tests are positive to the trigger food, case series suggest that these patients have a decreased probability of developing tolerance to the implicated food. The relationship between IgE and non-IgE mechanisms in FPIES requires further investigation. The gastrointestinal inflammation caused by FPIES could enhance penetrability of food proteins and their presentation to the immune system with subsequent generation of food-specific IgE antibodies. Conversely, local intestinal mucosal IgE antibodies could facilitate antigen uptake and intestinal inflammation.
Neuroendocrine Pathways
A recent case series of children with FPIES successfully treated with intravenous ondansetron during the supervised OFC raised questions about the role of serotonin signaling in FPIES [41•]. Ondansetron is a serotonin 5-HT3 receptor antagonist used mainly to treat nausea and vomiting, often following chemotherapy but also in viral gastroenteritis. It reduces activity of the vagus nerve both peripherally and centrally. The effectiveness of ondansetron suggests the potential role of an impaired neuroendocrinologic pathomechanism in FPIES reactions and warrants further study.
Conclusions
FPIES represents a more severe spectrum of the non-IgE-mediated gastrointestinal food allergic disorders, with potential for hypothermia, methemoglobinemia, acidemia, hypotension and shock. Because of the delayed onset of symptoms and absence of classic allergic cutaneous and respiratory manifestations, the FPIES diagnosis is frequently delayed or missed. A low index of suspicion for the cereal grains, vegetables and fruit that have a low potential for inducing IgE-mediated reactions but are common triggers of FPIES also likely contributes to misdiagnosis. Management relies on food avoidance and symptomatic treatment of dehydration. The pathophysiology of FPIES remains obscure, and there are no biomarkers for diagnosis. While most patients with FPIES to cow’s milk and soy resolve within the first 3–5 years of life, those with FPIES to solid food or with detectable systemic food-specific IgE tend to have a more protracted course. Studies documenting FPIES prevalence, natural history and pathophysiology are necessary to improve patient care and to develop evidence-based approaches to diagnosis and treatment.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance. •• Of major importance
Boyce JA, Assa’ad A, Burks AW, Jones SM, Sampson HA, Wood RA, Plaut M, Cooper SF, Fenton MJ, Arshad SH, et al. Guidelines for the diagnosis and management of food allergy in the United States: summary of the NIAID-sponsored expert panel report. J Allergy Clin Immunol. 2010;126:1105–18.
• Jarvinen KM, Nowak-Węgrzyn A. Food protein-induced enterocolitis syndrome: current management strategies. J Allergy Clin Immunol. 2013;1:317. This is a comprehensive and contemporary review on the manifestations, diagnosis and management of FPIES.
Nowak-Wegrzyn A, Sampson HA, Wood RA, Sicherer SH. Food protein-induced enterocolitis syndrome caused by solid food proteins. Pediatrics. 2003;111:829–35.
MI R. Allergic intestinal bleeding in the newborn: a clinical syndrome. Am J Med Sci. 1940;200:385–90.
Gryboski JD. Gastrointestinal milk allergy in infants. Pediatrics. 1967;40:354–62.
Powell GK. Enterocolitis in low-birth-weight infants associated with milk and soy protein intolerance. J Pediatr. 1976;88:840–4.
Powell GK. Milk- and soy-induced enterocolitis of infancy. Clinical features and standardization of challenge. J Pediatr. 1978;93:553–60.
Sicherer SH, Eigenmann PA, Sampson HA. Clinical features of food protein-induced enterocolitis syndrome. J Pediatr. 1998;133:214–9.
Burks AW, Casteel HB, Fiedorek SC, Williams LW, Pumphrey CL. Prospective oral food challenge study of two soybean protein isolates in patients with possible milk or soy protein enterocolitis. Pediatr Allergy Immunol. 1994;5:40–5.
Caubet JC, Nowak-Wegrzyn A. Food protein-induced enterocolitis to hen’s egg. J Allergy Clin Immunol. 2011;128:1386–8.
Levy Y, Danon YL. Food protein-induced enterocolitis syndrome–not only due to cow’s milk and soy. Pediatr Allergy Immunol. 2003;14:325–9.
Vandenplas Y, Edelman R, Sacre L. Chicken-induced anaphylactoid reaction and colitis. J Pediatr Gastroenterol Nutr. 1994;19:240–1.
Hojsak I, Kljaic-Turkalj M, Misak Z, Kolacek S. Rice protein-induced enterocolitis syndrome. Clin Nutr. 2006;25:533–6.
Bruni F, Peroni DG, Piacentini GL, De Luca G, Boner AL. Fruit proteins: another cause of food protein-induced enterocolitis syndrome. Allergy. 2008;63:1645–6.
Monti G, Viola S, Tarasco V, Lupica MM, Cosentino V, Castagno E. A case of severe allergic reaction to cooked potato. Acta Paediatr. 2011;100:e236–8.
Zapatero Remon L, Alonso Lebrero E, Martin Fernandez E, Martinez Molero MI. Food-protein-induced enterocolitis syndrome caused by fish. Allergol Immunopathol (Madr). 2005;33:312–6.
Vitoria JC, Camarero C, Sojo A, Ruiz A, Rodriguez-Soriano J. Enteropathy related to fish, rice, and chicken. Arch Dis Child. 1982;57:44–8.
Sopo SM, Filoni S, Giorgio V, Monaco S, Onesimo R. Food protein-induced enterocolitis syndrome (FPIES) to corn: a case report. J Investig Allergol Clin Immunol. 2012;22:391–2.
•• Katz Y, Goldberg MR, Rajuan N, Cohen A, Leshno M. The prevalence and natural course of food protein-induced enterocolitis syndrome to cow’s milk: a large-scale, prospective population-based study. J Allergy Clin Immunol. 2011;127:647–653 e641–643. This is the only population-based study conducted in Israel that documentedd the prevalence of FPIES.
McDonald PJ, Goldblum RM, Van Sickle GJ, Powell GK. Food protein-induced enterocolitis: altered antibody response to ingested antigen. Pediatr Res. 1984;18:751–5.
Tan J, Campbell D, Mehr S. Food protein-induced enterocolitis syndrome in an exclusively breast-fed infant-an uncommon entity. J Allergy Clin Immunol. 2012;129:873–4.
Monti G, Castagno E, Liguori SA, Lupica MM, Tarasco V, Viola S, Tovo PA. Food protein-induced enterocolitis syndrome by cow’s milk proteins passed through breast milk. J Allergy Clin Immunol. 2011;127:679–80.
Murray KF, Christie DL. Dietary protein intolerance in infants with transient methemoglobinemia and diarrhea. J Pediatr. 1993;122:90–2.
Mehr S, Kakakios A, Frith K, Kemp AS. Food protein-induced enterocolitis syndrome: 16 years experience. Pediatrics. 2009;123:e459–64.
Sopo SM, Giorgio V, Dello Iacono I, Novembre E, Mori F, Onesimo R. A multicentre retrospective study of 66 Italian children with food protein-induced enterocolitis syndrome: different management for different phenotypes. Clin Exp Allergy. 2012;42:1257–65.
Ruffner MRK, Barni S, Cianferoni A, BrownWhitehorn T, Spergel JM. Food protein-induced enterocolitis syndrome: insights from review f a large referral population. J Allergy Clin Immuno. 2013;1:343–9.
Beauchamp JN, Gaboury I, Ni A, Boland MP, Mack DR. Solid-food introduction in infants diagnosed as having a cow’s-milk protein-induced enterocolitis. J Pediatr Gastroenterol Nutr. 2011;52:639–43.
Fernandes BN, Boyle RJ, Gore C, Simpson A, Custovic A. Food protein-induced enterocolitis syndrome can occur in adults. J Allergy Clin Immunol. 2012;130:1199–200.
Onesimo R, Dello Iacono I, Giorgio V, Limongelli MG, Miceli Sopo S. Can food protein induced enterocolitis syndrome shift to immediate gastrointestinal hypersensitivity? A report of two cases. Eur Ann Allergy Clin Immunol. 2011;43:61–3.
Fogg MI, Brown-Whitehorn TA, Pawlowski NA, Spergel JM. Atopy patch test for the diagnosis of food protein-induced enterocolitis syndrome. Pediatr Allergy Immunol. 2006;17:351–5.
Jarvinen KM, Caubet JC, Sickles L, Ford LS, Sampson HA, Nowak-Wegrzyn A. Poor utility of atopy patch test in predicting tolerance development in food protein-induced enterocolitis syndrome. Ann Allergy Asthma Immunol. 2012;109:221–2.
Hwang JB, Song JY, Kang YN, Kim SP, Suh SI, Kam S, Choi WJ. The significance of gastric juice analysis for a positive challenge by a standard oral challenge test in typical cow’s milk protein-induced enterocolitis. J Korean Med Sci. 2008;23:251–5.
Scaparrotta A, Di Pillo S, Consilvio NP, Attanasi A, Cingolani A, Rapino D, Cerasa N, Pucci N, Di Gioacchino M, Chiarelli F. Usefulness of atopy patch test on a child with milk protein-induced enterocolitis syndrome: a case report. Int J Immunopathol Pharmacol. 2013;26:795–800.
Jayasooriya S, Fox AT, Murch SH. Do not laparotomize food-protein-induced enterocolitis syndrome. Pediatr Emerg Care. 2007;23:173–5.
Coates RW, Weaver KR, Lloyd R, Ceccacci N, Greenberg MR. Food protein-induced enterocolitis syndrome as a cause for infant hypotension. West J Emerg Med. 2011;12:512–4.
Menon N, Feuille E, Huang F, Nowak-Wegrzyn A. Knowledge of food protein-induced enterocolitis syndrome among general pediatricians. J Allergy Clin Immunol. 2013;131:AB177.
Nowak-Wegrzyn A, Assa’ad AH, Bahna SL, Bock SA, Sicherer SH, Teuber SS. Adverse reactions to food committee of American academy of allergy, A., and immunology. Work group report: oral food challenge testing. J Allergy Clin Immunol. 2009;123:S365–83.
Hwang JB, Sohn SM, Kim AS. Prospective follow-up oral food challenge in food protein-induced enterocolitis syndrome. Arch Dis Child. 2009;94:425–8.
Hill DJ, Murch SH, Rafferty K, Wallis P, Green CJ. The efficacy of amino acid-based formulas in relieving the symptoms of cow’s milk allergy: a systematic review. Clin Exp Allergy. 2007;37:808–22.
Sicherer SH. Food protein-induced enterocolitis syndrome: case presentations and management lessons. J Allergy Clin Immunol. 2005;115:149–56.
• Holbrook T, Keet CA, Frischmeyer-Guerrerio PA, Wood RA. Use of ondansetron for food protein-induced enterocolitis syndrome. J Allergy Clin Immunol. 2013;132:1219–1220. Case series reporting the utility of ondansetron for acute FPIES during oral food challenge.
Shek LP, Bardina L, Castro R, Sampson HA, Beyer K. Humoral and cellular responses to cow milk proteins in patients with milk-induced IgE-mediated and non-IgE-mediated disorders. Allergy. 2005;60:912–9.
Van Sickle GJ, Powell GK, McDonald PJ, Goldblum RM. Milk- and soy protein-induced enterocolitis: evidence for lymphocyte sensitization to specific food proteins. Gastroenterology. 1985;88:1915–21.
Benlounes N, Dupont C, Candalh C, Blaton MA, Darmon N, Desjeux JF, Heyman M. The threshold for immune cell reactivity to milk antigens decreases in cow’s milk allergy with intestinal symptoms. J Allergy Clin Immunol. 1996;98:781–9.
Heyman M, Darmon N, Dupont C, Dugas B, Hirribaren A, Blaton MA, Desjeux JF. Mononuclear cells from infants allergic to cow’s milk secrete tumor necrosis factor alpha, altering intestinal function. Gastroenterology. 1994;106:1514–23.
Karlsson MR, Rugtveit J, Brandtzaeg P. Allergen-responsive CD4 + CD25 + regulatory T cells in children who have outgrown cow’s milk allergy. J Exp Med. 2004;199:1679–88.
Hoffman KM, Ho DG, Sampson HA. Evaluation of the usefulness of lymphocyte proliferation assays in the diagnosis of allergy to cow’s milk. J Allergy Clin Immunol. 1997;99:360–6.
Mori F, Barni S, Cianferoni A, Pucci N, de Martino M, Novembre E. Cytokine expression in CD3 + cells in an infant with food protein-induced enterocolitis syndrome (FPIES): case report. Clin Dev Immunol. 2009;2009:679381.
Benlounes N, Candalh C, Matarazzo P, Dupont C, Heyman M. The time-course of milk antigen-induced TNF-alpha secretion differs according to the clinical symptoms in children with cow’s milk allergy. J Allergy Clin Immunol. 1999;104:863–9.
Chung HL, Hwang JB, Kwon YD, Park MH, Shin WJ, Park JB. Deposition of eosinophil-granule major basic protein and expression of intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 in the mucosa of the small intestine in infants with cow’s milk-sensitive enteropathy. J Allergy Clin Immunol. 1999;103:1195–201.
Chung HL, Hwang JB, Park JJ, Kim SG. Expression of transforming growth factor beta1, transforming growth factor type I and II receptors, and TNF-alpha in the mucosa of the small intestine in infants with food protein-induced enterocolitis syndrome. J Allergy Clin Immunol. 2002;109:150–4.
Powell GK, McDonald PJ, Van Sickle GJ, Goldblum RM. Absorption of food protein antigen in infants with food protein-induced enterocolitis. Dig Dis Sci. 1989;34:781–8.
Rothenberg ME. Eosinophilic gastrointestinal disorders (EGID). J Allergy Clin Immunol. 2004;113:11–28.
Lake AM. Food-induced eosinophilic proctocolitis. J Pediatr Gastroenterol Nutr. 2000;30:S58–60.
Fontaine JL, Navarro J. Small intestinal biopsy in cows milk protein allergy in infancy. Arch Dis Child. 1975;50:357–62.
Disclosure
A. Nowak-Wegrzyn received research funding from NIAD, FARE, Nutricia and Nestle, and honoraria from Thermofisher Scientific and Nestle, served as board member for Merck and Stallergenes, and received royalties from UpToDate. George Konstantinou reports no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nowak-Węgrzyn, A., Konstantinou, G. Non-IgE-Mediated Food Allergy: FPIES. Curr Pediatr Rep 2, 135–143 (2014). https://doi.org/10.1007/s40124-014-0043-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40124-014-0043-y