Abstract
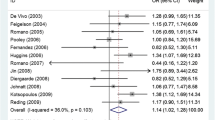
Progesterone and its receptor, progesterone receptor (PGR), have been widely studied for their roles in the onset and development of ovarian cancer. Although numerous epidemiological studies have focused on the association of PGR PROGINS and +331G/A polymorphisms with ovarian cancer susceptibility, presently, available results remain controversial, in part due to low sample sizes. Thus, a meta-analysis is required to evaluate this association. A literature search of PubMed, Embase, Web of Science, CNKI, and CBM databases was performed to retrieve eligible studies published before August 15, 2013. Summary odds ratios (ORs) with 95 % confidence intervals (CIs) were used to evaluate the strength of this association. All analyses were done using STATA 12.0 software (Stata Corp., College Station, TX, USA). Seventeen case–control studies with a total of 6,365 cases and 9,998 controls were identified. While no statistically significant association between the PROGINS allele and ovarian cancer risk was found in an overall analysis, a stratified analysis revealed that for Caucasians, never-oral contraceptive (OC) users, and serous tumor patients, there were statistically significant ORs for ovarian cancer risk associated with the mutated PROGINS allele. No significant association, however, between the +331G/A polymorphism and ovarian cancer susceptibility was observed in the overall analyses and subgroup analyses based on ethnicity and histological type. This meta-analysis provides evidence that the PROGINS allele occurs more frequently in ovarian cancer patients and especially in non-OC users and serous cancer patients, indicating that PROGINS may be a risk modifier. No significant association between the +331G/A polymorphism and ovarian cancer was found, even in stratified analyses by ethnicity and histological type. More detailed and well-designed studies are still needed to confirm the role of the PROGINS allele in ovarian cancer development.




Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30.
Lowe KA, Chia VM, Taylor A, O'Malley C, Kelsh M, et al. An international assessment of ovarian cancer incidence and mortality. Gynecol Oncol. 2013;130:107–14.
Boyd J, Rubin SC. Hereditary ovarian cancer: molecular genetics and clinical implications. Gynecol Oncol. 1997;64:196–206.
Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science. 1994;266:66–71.
Wooster R, Weber BL. Breast and ovarian cancer. N Engl J Med. 2003;348:2339–47.
Whittemore AS, Harris R, Itnyre J. Characteristics relating to ovarian cancer risk: collaborative analysis of 12 US case–control studies. II. Invasive epithelial ovarian cancers in white women. Collaborative Ovarian Cancer Group. Am J Epidemiol. 1992;136:1184–203.
King RJ. Biology of female sex hormone action in relation to contraceptive agents and neoplasia. Contraception. 1991;43:527–42.
Ivarsson K, Sundfeldt K, Brannstrom M, Janson PO. Production of steroids by human ovarian surface epithelial cells in culture: possible role of progesterone as growth inhibitor. Gynecol Oncol. 2001;82:116–21.
Rodriguez GC, Nagarsheth NP, Lee KL, Bentley RC, Walmer DK, et al. Progestin-induced apoptosis in the macaque ovarian epithelium: differential regulation of transforming growth factor-beta. J Natl Cancer Inst. 2002;94:50–60.
Lydon JP, DeMayo FJ, Funk CR, Mani SK, Hughes AR, et al. Mice lacking progesterone receptor exhibit pleiotropic reproductive abnormalities. Genes Dev. 1995;9:2266–78.
Mattei MG, Krust A, Stropp U, Mattei JF, Chambon P. Assignment of the human progesterone receptor to the q22 band of chromosome 11. Hum Genet. 1988;78:96–7.
Wen DX, Xu YF, Mais DE, Goldman ME, McDonnell DP. The A and B isoforms of the human progesterone receptor operate through distinct signaling pathways within target cells. Mol Cell Biol. 1994;14:8356–64.
McKenna NJ, Kieback DG, Carney DN, Fanning M, McLinden J, et al. A germline TaqI restriction fragment length polymorphism in the progesterone receptor gene in ovarian carcinoma. Br J Cancer. 1995;71:451–5.
Spurdle AB, Webb PM, Purdie DM, Chen X, Green A, et al. No significant association between progesterone receptor exon 4 Val660Leu G/T polymorphism and risk of ovarian cancer. Carcinogenesis. 2001;22:717–21.
Lancaster JM, Wenham RM, Halabi S, Calingaert B, Marks JR, et al. No relationship between ovarian cancer risk and progesterone receptor gene polymorphism in a population-based, case–control study in North Carolina. Cancer Epidemiol, Biomark Prev Publ Am Assoc Cancer Res, Cosponsored Am Soc Prev Oncol. 2003;12:226–7.
Berchuck A, Schildkraut JM, Wenham RM, Calingaert B, Ali S, et al. Progesterone receptor promoter +331A polymorphism is associated with a reduced risk of endometrioid and clear cell ovarian cancers. Cancer Epidemiol, Biomark Prev Publ Am Assoc Cancer Res, Cosponsored Am Soc Prev Oncol. 2004;13:2141–7.
Risch HA, Bale AE, Beck PA, Zheng W. PGR +331 A/G and increased risk of epithelial ovarian cancer. Cancer Epidemiol, Biomark Prev Publ Am Assoc Cancer Res, Cosponsored Am Soc Prev Oncol. 2006;15:1738–41.
Quaye L, Tyrer J, Ramus SJ, Song H, Wozniak E, et al. Association between common germline genetic variation in 94 candidate genes or regions and risks of invasive epithelial ovarian cancer. PloS One. 2009;4:e5983.
Chaudhary S, Panda AK, Mishra DR, Mishra SK. Association of +331G/A PGR polymorphism with susceptibility to female reproductive cancer: evidence from a meta-analysis. PloS One. 2013;8:e53308.
da Costa BR, Cevallos M, Altman DG, Rutjes AW, Egger M. Uses and misuses of the strobe statement: bibliographic study. BMJ Open. 2011;1:e000048.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58.
Zintzaras E, Ioannidis JP. Heterogeneity testing in meta-analysis of genome searches. Genet Epidemiol. 2005;28:123–37.
Peters JL, Sutton AJ, Jones DR, Abrams KR, Rushton L. Comparison of two methods to detect publication bias in meta-analysis. JAMA. 2006;295:676–80.
Manolitsas TP, Englefield P, Eccles DM, Campbell IG. No association of a 306-bp insertion polymorphism in the progesterone receptor gene with ovarian and breast cancer. Br J Cancer. 1997;75:1398–9.
Runnebaum IB, Wang-Gohrke S, Vesprini D, Kreienberg R, Lynch H, et al. Progesterone receptor variant increases ovarian cancer risk in BRCA1 and BRCA2 mutation carriers who were never exposed to oral contraceptives. Pharmacogenetics. 2001;11:635–8.
Tong D, Fabjani G, Heinze G, Obermair A, Leodolter S, et al. Analysis of the human progesterone receptor gene polymorphism PROGINS in Austrian ovarian carcinoma patients. Int J Cancer [Journal International du Cancer]. 2001;95:394–7.
Agoulnik IU, Tong XW, Fischer DC, Korner K, Atkinson NE, et al. A germline variation in the progesterone receptor gene increases transcriptional activity and may modify ovarian cancer risk. J Clin Endocrinol Metab. 2004;89:6340–7.
Pearce CL, Hirschhorn JN, Wu AH, Burtt NP, Stram DO, et al. Clarifying the PROGINS allele association in ovarian and breast cancer risk: a haplotype-based analysis. J Natl Cancer Inst. 2005;97:51–9.
Terry KL, De Vivo I, Titus-Ernstoff L, Sluss PM, Cramer DW. Genetic variation in the progesterone receptor gene and ovarian cancer risk. Am J Epidemiol. 2005;161:442–51.
Romano A, Lindsey PJ, Fischer DC, Delvoux B, Paulussen AD, et al. Two functionally relevant polymorphisms in the human progesterone receptor gene (+331 G/A and PROGINS) and the predisposition for breast and/or ovarian cancer. Gynecol Oncol. 2006;101:287–95.
Delort L, Chalabi N, Satih S, Rabiau N, Kwiatkowski F, et al. Association between genetic polymorphisms and ovarian cancer risk. Anticancer Res. 2008;28:3079–81.
Leite DB, Junqueira MG, de Carvalho CV, Massad-Costa AM, Goncalves WJ, et al. Progesterone receptor (PROGINS) polymorphism and the risk of ovarian cancer. Steroids. 2008;73:676–80.
Ludwig AH, Murawska M, Panek G, Timorek A, Kupryjanczyk J. Androgen, progesterone, and FSH receptor polymorphisms in ovarian cancer risk and outcome. Endocr-Relat Cancer. 2009;16:1005–16.
Lancaster JM, Berchuck A, Carney ME, Wiseman R, Taylor JA. Progesterone receptor gene polymorphism and risk for breast and ovarian cancer. Br J Cancer. 1998;78:277.
Rosenberg L, Palmer JR, Zauber AG, Warshauer ME, Lewis Jr JL, et al. A case–control study of oral contraceptive use and invasive epithelial ovarian cancer. Am J Epidemiol. 1994;139:654–61.
Risch HA. Hormonal etiology of epithelial ovarian cancer, with a hypothesis concerning the role of androgens and progesterone. J Natl Cancer Inst. 1998;90:1774–86.
Narod SA, Risch H, Moslehi R, Dorum A, Neuhausen S, et al. Oral contraceptives and the risk of hereditary ovarian cancer. Hereditary ovarian cancer clinical study group. New Engl J Med. 1998;339:424–8.
De Vivo I, Huggins GS, Hankinson SE, Lescault PJ, Boezen M, et al. A functional polymorphism in the promoter of the progesterone receptor gene associated with endometrial cancer risk. Proc Natl Acad Sci U S A. 2002;99:12263–8.
Fujimoto J, Ichigo S, Hirose R, Sakaguchi H, Tamaya T. Clinical implication of expression of progesterone receptor form A and B mRNAs in secondary spreading of gynecologic cancers. J Steroid Biochem Mol Biol. 1997;62:449–54.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Ting Liu and Lilan Chen have contributed equally to this work and are co-first authors.
Rights and permissions
About this article
Cite this article
Liu, T., Chen, L., Sun, X. et al. Progesterone receptor PROGINS and +331G/A polymorphisms confer susceptibility to ovarian cancer: a meta-analysis based on 17 studies. Tumor Biol. 35, 2427–2436 (2014). https://doi.org/10.1007/s13277-013-1322-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-1322-x