Abstract
Objective
This study investigated the correlation between the chronological age, telomere length in peripheral blood leukocytes and blood laboratory data of female patients with mild hypertension to identify laboratory data that reflect the biological aging of individuals.
Design
Cross-sectional population-based study.
Setting
Outpatient clinic of the Department of Cardiovascular, Respiratory, and Geriatric Medicine Kyushu University Hospital at Beppu in Japan.
Participants
Outpatients with mild hypertension treated with a low dose of amlodipine.
Measurements
The laboratory data of female patients were collected and the telomere length parameters in their peripheral blood leukocytes were determined by Southern blotting. Any correlations between the laboratory data and the telomere length parameters were assessed.
Results
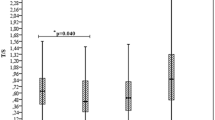
The patients showed a positive correlation between the telomere length and the high density lipoprotein, albumin, creatinine, hemoglobin levels, red blood cell counts, and a negative correlation with the globulin level. The extent of subtelomeric methylation of long telomeres tended to correlate negatively with the telomeric attrition. Only the creatinine level correlated with subtelomeric methylation, but not with telomeric length.
Conclusion
HDL and the albumin/globulin ratio were potential indicators for individual somatic genomic aging. Creatinine may therefore be a useful indicator for a predisposition for telomeric attrition.
Similar content being viewed by others
References
Blackburn EH Structure and function of telomeres. Nature (London). 1991;350:569–573.
Zakian VA. Telomeres: beginning to understand the end. Science. 1995;270:1601–1607.
Aubert G, Lansdorp PM. Telomeres and aging. Physiol Rev. 2008;88:557–795.
Iwama H, Ohyashiki K, Ohyashiki JH, et al. Telomeric length and telomerase activity vary with age in peripheral blood cells obtained from normal individuals. Hum Genet. 1998;102:397–402.
Guan JZ, Maeda T, Sugano M, Oyama J, Higuchi Y, Makino N. Change in the telomere length distribution with age in the Japanese population. Mol Cell Biochem. 2007;304:253–260.
Sidorova I, Kimurab M, Yashinc A, Aviv A. Leukocyte telomere dynamics and human hematopoietic stem cell kinetics during somatic growth. Experimental Hematology 2009;37:514–524.
Okuda K, Khan MY, Skurnick J, et al. Telomere attrition of the human abdominal aorta: relationships with age and atherosclerosis. Atherosclerosis. 2000;152:391–398.
Uziel O, Singer JA, Danicek V, et al. Telomere dynamics in arteries and mononuclear cells of diabetic patients: effect of diabetes and of glycemic control. Exp Gerontol. 2007;42:971–978.
Panossian LA, Porter VR, Valenzuela HF, et al. Telomere shortening in T cells correlates with Alzheimer’s disease status. Neurobiol ageing. 2003;24:77–84.
Valdes AM, Andrew T, Gardner JP, et al. Obesity, cigarette smoking, and telomere length in women. Lancet. 2005;366:662–664.
Epel ES, Blackburn EH, Lin F, et al. Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci U S A. 2004;101:17312–17315.
Guan JZ, Maeda T, Sugano M, et al. A Percentage analysis of the telomere length in Parkinson’s disease patients. J Gerontol A Biol Sci Med Sci. 2008;63A:467–473.
Guan JZ, Maeda T, Sugano M, et al. An analysis of telomere length in sarcoidosis. J Gerontol A Biol Sci Med Sci. 2007;62:1199–1203.
Brouilette S, Singh RK, Thompson JR, Goodall AH, Samani NJ. White cell telomere length and risk of premature myocardial infarction. Arterioscler Thromb Vasc Biol. 2003;23:842–46.
Ogami M, Ikura Y, Ohsawa M, et al. Telomere shortening in human coronary artery diseases. Arterioscler Thromb Vasc Biol. 2004;24:546–50.
Hansen RS, Wijmenga C, Luo P, et al. The DNMT3B DNA methyltransferase gene is mutated in the ICF immunodeficiency syndrome. Proc Natl Acad Sci U S A. 1999;96:14412–7.
Yehezkel S, Segev Y, Viegas-Péquignot E, Skorecki K, Selig S. Hypomethylation of subtelomeric regions in ICF syndrome is associated with abnormally short telomeres and enhanced transcription from telomeric regions. Hum Mol Genet. 2008;17:2776–89.
Maeda T, Guan JZ, Oyama J, Higuchi Y, Makino N. Aging-associated alteration in subtelomeric methylation in Parkinson’s disease. J Gerontol A Biol Sci Med Sci. 2009 64:949–955.
Maeda T, Guan JZ, Higuchi Y, Oyama J, Makino N. Age-related alterations in subtelomeric methylation in sarcoidosis patients. J Gerontol A Biol Sci Med Sci. 2009 64:752–760.
Yamaguchi H, Calado RT, Ly H, et al. Mutations in TERT, the gene for telomerase reverse transcriptase, in aplastic anemia. N Engl J Med. 2005;352:1413–1424.
Chen W, Gardner JP, Kimura M, et al. Leukocyte telomere length is associated with HDL cholesterol levels: The Bogalusa heart study. Atherosclerosis. 2009;205:620–625
Shay JW, Wright WE. Telomerase therapeutics for cancer: challenges and new directions. Nat Rev Drug Discov. 2006;5:577–584.
van Overveld PG, Lemmers RJ, Sandkuijl LA, et al. Hypomethylation of D4Z4 in 4q-linked and non-4q-linked facioscapulohumeral muscular dystrophy. Nat Genet. 2003;35:315–317.
Pedram M, Sprung CN, Gao Q, Lo AW, Reynolds GE, Murnane JP. Telomere position effect and silencing of transgenes near telomeres in the mouse. Mol Cell Biol. 2006;26:1865–1878.
Blanco R, Muñoz P, Flores JM, Klatt P, Blasco MA. Telomerase abrogation dramatically accelerates TRF2-induced epithelial carcinogenesis. Genes Dev. 2007;21:206–220.
Wu L, Multani AS, He H, et al. Pot1 deficiency initiates DNA damage checkpoint activation and aberrant homologous recombination at telomeres. Cell. 2006;126:49–62.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maeda, T., Oyama, J.I., Sasaki, M. et al. The correlation between the clinical laboratory data and the telomere length in peripheral blood leukocytes of Japanese female patients with hypertension. J Nutr Health Aging 15, 240–244 (2011). https://doi.org/10.1007/s12603-010-0137-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-010-0137-3