Abstract
Introduction
As healthcare spending on diabetes and its complications continues to rise, the optimization of prescribed insulin regimens is becoming increasingly important from both clinical and economic perspectives. The aim of the present study was to evaluate the costeffectiveness of 75/25 biphasic insulin lispro and 50/50 biphasic insulin lispro (Humalog® Mix75/25 and Humalog® Mix50/50, respectively; Eli Lilly and Company, Indianapolis, IN, USA) compared with a long-acting analog insulin regimen in patients with type 2 diabetes.
Methods
A published and validated computer simulation model of diabetes was used to evaluate the cost-effectiveness of 75/25 and 50/50 biphasic insulin lispro versus a longacting analog insulin (insulin glargine) from the perspective of a healthcare payer in the UK. Treatment effects in terms of glycated hemoglobin (HbA1c) benefits were taken from a recent meta-analysis. Direct medical costs including pharmacy, complication, and patient management costs were obtained from published sources. All costs were expressed in 2008 British pounds sterling (GBP), and future costs and clinical benefits were discounted at 3.5% per annum. Sensitivity analyses were performed.
Results
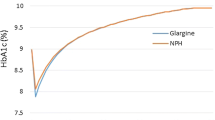
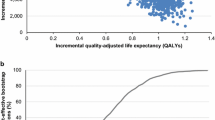
75/25 and 50/50 biphasic insulin lispro were associated with improvements in life expectancy of 0.09 and 0.13 years, respectively, improvements in quality-adjusted life expectancy of 0.09 quality-adjusted life years (QALYs) and 0.12 QALYs, respectively, and reductions in cost of GBP 1,217 and GBP 430, respectively, when compared with long-acting analog insulin.
Conclusion
Based on a recently published metaanalysis, biphasic analog insulins are likely to improve clinical outcomes and reduce costs versus long-acting analog insulins in the longterm treatment of patients with type 2 diabetes in the UK.
Similar content being viewed by others
References
González EL, Johansson S, Wallander MA, Rodríguez LA. Trends in the prevalence and incidence of diabetes in the UK: 1996–2005. J Epidemiol Community Health. 2009;63:332–336.
Thomas MC, Hardoon SL, Papacosta AO, et al. Evidence of an accelerating increase in prevalence of diagnosed Type 2 diabetes in British men, 1978–2005. Diabet Med. 2009;26:766–772.
Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med. 2003;348:383–393.
Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med. 2008;358:580–591.
Colhoun HM, Thomason MJ, Mackness MI, et al. Collaborative AtoRvastatin Diabetes Study (CARDS). Design of the Collaborative AtoRvastatin Diabetes Study (CARDS) in patients with type 2 diabetes. Diabet Med. 2002;19:201–211.
Filion KB, Joseph L, Boivin JF, Suissa S, Brophy JM. Trends in the prescription of anti-diabetic medications in the United Kingdom: a population-based analysis. Pharmacoepidemiol Drug Saf. 2009;18:973–976.
The NHS Information Centre, Prescribing Support and Primary Care Services. Prescribing for Diabetes in England: 2004/5 to 2009/10. Available at: http://www.ic.nhs.uk/pubs/prescribingdiabetes0410. Accessed Nov 12 2012.
Palmer AJ, Roze S, Valentine WJ, et al. The CORE Diabetes Model: Projecting long-term clinical outcomes, costs and cost-effectiveness of interventions in diabetes mellitus (types 1 and 2) to support clinical and reimbursement decision-making. Curr Med Res Opin. 2004;20(Suppl. 1): S5–26.
Palmer AJ, Roze S, Valentine WJ, et al. Validation of the CORE Diabetes Model against epidemiological and clinical studies. Curr Med Res Opin. 2004;20(Suppl. 1):S27–S40.
Jones S, Benroubi M, Castell C, et al. Characteristics of patients with type 2 diabetes mellitus initiating insulin therapy: baseline data from the INSTIGATE study. Curr Med Res Opin. 2009;25:691–700.
Qayyum R, Bolen S, Maruthur N, et al. Systematic review: comparative effectiveness and safety of premixed insulin analogues in type 2 diabetes. Ann Intern Med. 2008;149:549–559.
Ray JA, Boye KS, Yurgin N, et al. Exenatide versus insulin glargine in patients with type 2 diabetes in the UK: a model of long-term clinical and cost outcomes. Curr Med Res Opin. 2007;23:609–622.
National Health Service Electronic Drug Tariff 2009. Available at: http://www.ppa.org.uk/ppa/drug_tariff_back_copies.htm. Accessed May 12 2010.
UK Office for National Statistics. Consumer Price Indices. Available at: http://www.statistics.gov.uk/statbase/Product.asp?vlnk=10919. Accessed May 20 2010.
World Health Organization Collaborating Centre for Drug Statistics Methodology. Anatomical Therapeutic Chemical/Defined Daily Dose Index 2010. Available at: http://www.whocc.no/atc_ddd_index/?code=A10AE. Accessed May 11 2010.
National Institute for Health and Clinical Excellence. Guide to the Methods of Technology Appraisal (reference N0515). Available at: http://www.nice.org.uk/niceMedia/pdf/TAP_Methods.pdf. Accessed May 11 2010.
Levy AR, Christensen TL, Johnson JA. Utility values for symptomatic non-severe hypoglycaemia elicited from persons with and without diabetes in Canada and the United Kingdom. Health Qual Life Outcomes. 2008;29:73.
Clarke P, Gray A, Holman R. Estimating utility values for health states of type 2 diabetic patients using the EQ-5D (UKPDS 62). Med Decis Making. 2002;22:340–349.
Coffey JT, Brandle M, Zhou H, et al. Valuing health-related quality of life in diabetes. Diabetes Care. 2002;25:2238–2243.
Valentine WJ, Palmer AJ, Erny-Albrecht KM, et al. Cost-effectiveness of basal insulin from a US health system perspective: comparative analyses of detemir, glargine, and NPH. Adv Ther. 2006;23:191–207.
Ray JA, Valentine WJ, Roze S, et al. Insulin therapy in type 2 diabetes patients failing oral agents: cost-effectiveness of biphasic insulin aspart 70/30 vs. insulin glargine in the US. Diabetes Obes Metab. 2007;9:103–113.
Lasserson DS, Glasziou P, Perera R, Holman RR, Farmer AJ. Optimal insulin regimens in type 2 diabetes mellitus: systematic review and meta-analyses. Diabetologia. 2009;52:1990–2000.
Nathan DM, Buse JB, Davidson MB, et al. American Diabetes Association; European Association for Study of Diabetes. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2009;32:193–203.
National Institute for Health and Clinical Excellence. CG87 Type 2 diabetes - newer agents (a partial update of CG66): NICE guideline. Available at: http://www.nice.org.uk/nicemedia/live/12165/44320/44320.pdf. Accessed Nov 7 2012.
Holden SE, Poole CD, Morgan CL, Currie CJ. Evaluation of the incremental cost to the National Health Service of prescribing analogue insulin. BMJ Open. 2011;1:e000258.
Swinnen SG, Hoekstra JB, DeVries JH. Insulin therapy for type 2 diabetes. Diabetes Care. 2009;32(Suppl. 2):S253–S259.
Data on file: USA; Eli Lilly and Company, 2010.
Author information
Authors and Affiliations
Corresponding author
Additional information
To view enhanced content go to www.advancesintherapy.com
Rights and permissions
About this article
Cite this article
Pollock, R.F., Curtis, B.H., Smith-Palmer, J. et al. A UK Analysis of the Cost-Effectiveness of Humalog Mix75/25 and Mix50/50 Versus Long-Acting Basal Insulin. Adv Therapy 29, 1051–1066 (2012). https://doi.org/10.1007/s12325-012-0065-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-012-0065-1