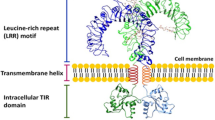
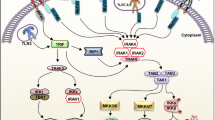
Abstract
Animals and plants are exposed to myriads of potential microbial invaders. In case of animals, Toll-like receptors (TLRs) act as the primary defense against infection by pathogens. Arguably, less is known regarding the activation of TLRs that connect the innate and adaptive immune systems. Some TLR ligands have been used as adjuvants in various vaccines and have gained a great deal of attention due to their ability to elicit an effective immune response. Understanding the intricate relationships between various molecules involved in TLR signaling and their positive or negative regulation is a key focus for the development of effective therapeutics. In this review, recent developments in TLR signaling that will be very important in providing new drug target molecules and a better understanding of molecular regulation of innate immunity are discussed.
Article PDF
Similar content being viewed by others
References
Adams, M., Navabi, H., Jasani, B., Man, S., Fiander, A., Evans, A. S., Donninger, C., and Mason, M., Dendritic cell (DC) based therapy for cervical cancer: use of DC pulsed with tumour lysate and matured with a novel synthetic clinically non-toxic double stranded RNA analogue poly [I]:poly [C(12)U] (Ampligen R). Vaccine, 21, 787–790 (2003).
Akira, S., Uematsu, S., and Takeuchi, O., Pathogen recognition and innate immunity. Cell, 124, 783–801 (2006).
An, H., Zhao, W., Hou, J., Zhang, Y., Xie, Y., Zheng, Y., Xu, H., Qian, C., Zhou, J., Yu, Y., Liu, S., Feng, G., and Cao, X., SHP-2 phosphatase negatively regulates the TRIF adaptor protein-dependent type I interferon and proinflammatory cytokine production. Immunity, 25, 919–928 (2006).
Anantharaman, V. and Aravind, L., The GOLD domain, a novel protein module involved in Golgi function and secretion. Genome Biol, 3, research0023 (2002).
Applequist, S. E., Rollman, E., Wareing, M. D., Liden, M., Rozell, B., Hinkula, J., and Ljunggren, H. G., Activation of innate immunity, inflammation, and potentiation of DNA vaccination through mammalian expression of the TLR5 agonist flagellin. J. Immunol., 175, 3882–3891 (2005).
Asea, A., Kraeft, S. K., Kurt-Jones, E. A., Stevenson, M. A., Chen, L. B., Finberg, R. W., Koo, G. C., and Calderwood, S. K., HSP70 stimulates cytokine production through a CD14-dependant pathway, demonstrating its dual role as a chaperone and cytokine. Nat. Med., 6, 435–442 (2000).
Atkinson, T. J., Toll-like receptors, transduction-effector pathways, and disease diversity: Evidence of an immunobiological paradigm explaining all human illness? Int. Rev. Immunol., 27, 255–281 (2008).
Barton, G. M., Kagan, J. C., and Medzhitov, R., Intracellular localization of Toll-like receptor 9 prevents recognition of self DNA but facilitates access to viral DNA. Nat. Immunol., 7, 49–56 (2006).
Beutler, B., Inferences, questions and possibilities in Tolllike receptor signalling. Nature, 430, 257–263 (2004).
Biragyn, A., Ruffini, P. A., Leifer, C. A., Klyushnenkova, E., Shakhov, A., Chertov, O., Shirakawa, A. K., Farber, J. M., Segal, D. M., Oppenheim, J. J., and Kwak, L. W., Toll-like receptor 4-dependent activation of dendritic cells by betadefensin 2. Science, 298, 1025–1029 (2002).
Boehm, U., Klamp, T., Groot, M., and Howard, J. C., Cellular responses to interferon-gamma. Annu. Rev. Immunol., 15, 749–795 (1997).
Boone, D. L., Turer, E. E., Lee, E. G., Ahmad, R. C., Wheeler, M. T., Tsui, C., Hurley, P., Chien, M., Chai, S., Hitotsumatsu, O., Mcnally, E., Pickart, C., and Ma, A., The ubiquitinmodifying enzyme A20 is required for termination of Tolllike receptor responses. Nat. Immunol., 5, 1052–1060 (2004).
Bowie, A., Kiss-Toth, E., Symons, J. A., Smith, G. L., Dower, S. K., and O’neill, L. A., A46R and A52R from vaccinia virus are antagonists of host IL-1 and toll-like receptor signaling. Proc. Natl. Acad. Sci. USA, 97, 10162–10167 (2000).
Brint, E. K., Xu, D., Liu, H., Dunne, A., Mckenzie, A. N., O’neill, L. A., and Liew, F. Y., ST2 is an inhibitor of interleukin 1 receptor and Toll-like receptor 4 signaling and maintains endotoxin tolerance. Nat. Immunol., 5, 373–379 (2004).
Broide, D. H., Immunostimulatory sequences of DNA and conjugates in the treatment of allergic rhinitis. Curr. Allergy Asthma Rep., 5, 182–185 (2005).
Burns, K., Clatworthy, J., Martin, L., Martinon, F., Plumpton, C., Maschera, B., Lewis, A., Ray, K., Tschopp, J., and Volpe, F., Tollip, a new component of the IL-1RI pathway, links IRAK to the IL-1 receptor. Nat. Cell Biol., 2, 346–351 (2000).
Carty, M., Goodbody, R., Schroder, M., Stack, J., Moynagh, P. N., and Bowie, A. G., The human adaptor SARM negatively regulates adaptor protein TRIF-dependent Toll-like receptor signaling. Nat. Immunol., 7, 1074–1081 (2006).
Chang, Y. C., Madkan, V., Cook-Norris, R., Sra, K., and Tyring, S., Current and potential uses of imiquimod. South Med. J., 98, 914–920 (2005).
Chung, C. D., Liao, J., Liu, B., Rao, X., Jay, P., Berta, P., and Shuai, K., Specific inhibition of Stat3 signal transduction by PIAS3. Science, 278, 1803–1805 (1997).
Cluff, C. W., Baldridge, J. R., Stover, A. G., Evans, J. T., Johnson, D. A., Lacy, M. J., Clawson, V. G., Yorgensen, V. M., Johnson, C. L., Livesay, M. T., Hershberg, R. M., and Persing, D. H., Synthetic toll-like receptor 4 agonists stimulate innate resistance to infectious challenge. Infect. Immun., 73, 3044–3052 (2005).
Dan, H. C., Sun, M., Kaneko, S., Feldman, R. I., Nicosia, S. V., Wang, H. G., Tsang, B. K., and Cheng, J. Q., Akt phosphorylation and stabilization of X-linked inhibitor of apoptosis protein (XIAP). J. Biol. Chem., 279, 5405–5412 (2004).
Deng, L., Wang, C., Spencer, E., Yang, L., Braun, A., You, J., Slaughter, C., Pickart, C., and Chen, Z. J., Activation of the IkappaB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell, 103, 351–361 (2000).
Dieckmann, T., Withers-Ward, E. S., Jarosinski, M. A., Liu, C. F., Chen, I. S., and Feigon, J., Structure of a human DNA repair protein UBA domain that interacts with HIV-1 Vpr. Nat. Struct. Biol., 5, 1042–1047 (1998).
Diehl, G. E., Yue, H. H., Hsieh, K., Kuang, A. A., Ho, M., Morici, L. A., Lenz, L. L., Cado, D., Riley, L. W., and Winoto, A., TRAIL-R as a negative regulator of innate immune cell responses. Immunity, 21, 877–889 (2004).
Divanovic, S., Trompette, A., Atabani, S. F., Madan, R., Golenbock, D. T., Visintin, A., Finberg, R. W., Tarakhovsky, A., Vogel, S. N., Belkaid, Y., Kurt-Jones, E. A., and Karp, C. L., Negative regulation of Toll-like receptor 4 signaling by the Toll-like receptor homolog RP105. Nat. Immunol., 6, 571–578 (2005).
Evans, J. T., Cluff, C. W., Johnson, D. A., Lacy, M. J., Persing, D. H., and Baldridge, J. R., Enhancement of antigen-specific immunity via the TLR4 ligands MPL adjuvant and Ribi.529. Expert. Rev. Vaccines, 2, 219–229 (2003).
Fritz, J. H., Ferrero, R. L., Philpott, D. J., and Girardin, S. E., Nod-like proteins in immunity, inflammation and disease. Nat. Immunol., 7, 1250–1257 (2006).
Gilchrist, M., Thorsson, V., Li, B., Rust, A. G., Korb, M., Roach, J. C., Kennedy, K., Hai, T., Bolouri, H., and Aderem, A., Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4. Nature, 441, 173–178 (2006).
Guillot, L., Balloy, V., Mccormack, F. X., Golenbock, D. T., Chignard, M., and Si-Tahar, M., Cutting edge: the immunostimulatory activity of the lung surfactant protein-A involves Toll-like receptor 4. J. Immunol., 168, 5989–5992 (2002).
Halperin, S. A., Dobson, S., Mcneil, S., Langley, J. M., Smith, B., Mccall-Sani, R., Levitt, D., Nest, G. V., Gennevois, D., and Eiden, J. J., Comparison of the safety and immunogenicity of hepatitis B virus surface antigen co-administered with an immunostimulatory phosphorothioate oligonucleotide and a licensed hepatitis B vaccine in healthy young adults. Vaccine, 24, 20–26 (2006).
Hardy, M. P., and O’neill, L. A., The murine IRAK2 gene encodes four alternatively spliced isoforms, two of which are inhibitory. J. Biol. Chem., 279, 27699–27708 (2004).
Harmey, J. H., Bucana, C. D., Lu, W., Byrne, A. M., Mcdonnell, S., Lynch, C., Bouchier-Hayes, D., and Dong, Z., Lipopolysaccharide-induced metastatic growth is associated with increased angiogenesis, vascular permeability and tumor cell invasion. Int. J. Cancer, 101, 415–422 (2002).
Harte, M. T., Haga, I. R., Maloney, G., Gray, P., Reading, P. C., Bartlett, N. W., Smith, G. L., Bowie, A., and O’neill, L. A., The poxvirus protein A52R targets Toll-like receptor signaling complexes to suppress host defense. J. Exp. Med., 197, 343–351 (2003).
Heyninck, K. and Beyaert, R., The cytokine-inducible zinc finger protein A20 inhibits IL-1-induced NF-kappaB activation at the level of TRAF6. FEBS Lett., 442, 147–150 (1999).
Higgins, D., Marshall, J. D., Traquina, P., Van Nest, G., and Livingston, B. D., Immunostimulatory DNA as a vaccine adjuvant. Expert. Rev. Vaccines, 6, 747–759 (2007).
Hirotani, T., Lee, P. Y., Kuwata, H., Yamamoto, M., Matsumoto, M., Kawase, I., Akira, S., and Takeda, K., The nuclear IkappaB protein IkappaBNS selectively inhibits lipopolysaccharide-induced IL-6 production in macrophages of the colonic lamina propria. J. Immunol., 174, 3650–3657 (2005).
Hoebe, K., Du, X., Georgel, P., Janssen, E., Tabeta, K., Kim, S. O., Goode, J., Lin, P., Mann, N., Mudd, S., Crozat, K., Sovath, S., Han, J., and Beutler, B., Identification of Lps2 as a key transducer of MyD88-independent TIR signalling. Nature, 424, 743–748 (2003a).
Hoebe, K., Janssen, E. M., Kim, S. O., Alexopoulou, L., Flavell, R. A., Han, J., and Beutler, B., Upregulation of costimulatory molecules induced by lipopolysaccharide and double-stranded RNA occurs by Trif-dependent and Trif-independent pathways. Nat. Immunol., 4, 1223–1229 (2003b).
Hoffman, E. S., Smith, R. E., and Renaud, R. C., Jr., From the analyst’s couch: TLR-targeted therapeutics. Nat. Rev. Drug Discov., 4, 879–880 (2005).
Honko, A. N., Sriranganathan, N., Lees, C. J., and Mizel, S. B., Flagellin is an effective adjuvant for immunization against lethal respiratory challenge with Yersinia pestis. Infect Immun., 74, 1113–1120 (2006).
Horng, T., Barton, G. M., Flavell, R. A., and Medzhitov, R., The adaptor molecule TIRAP provides signalling specificity for Toll-like receptors. Nature, 420, 329–333 (2002).
Hunter, R. L., Overview of vaccine adjuvants: present and future. Vaccine, 20Suppl 3, S7–12 (2002).
Ilvesaro, J. M., Merrell, M. A., Swain, T. M., Davidson, J., Zayzafoon, M., Harris, K. W., and Selander, K. S., Toll like receptor-9 agonists stimulate prostate cancer invasion in vitro. Prostate, 67, 774–781 (2007).
Inohara, Chamaillard, Mcdonald, C., and Nunez, G., NODLRR proteins: Role in host-microbial interactions and inflammatory disease. Annu. Rev. Biochem., 74, 355–383 (2005).
Ishii, K. J. and Akira, S., Toll or toll-free adjuvant path toward the optimal vaccine development. J. Clin. Immunol., 27, 363–371 (2007).
Iwami, K. I., Matsuguchi, T., Masuda, A., Kikuchi, T., Musikacharoen, T., and Yoshikai, Y., Cutting edge: naturally occurring soluble form of mouse Toll-like receptor 4 inhibits lipopolysaccharide signaling. J. Immunol., 165, 6682–6686 (2000).
Janssens, S., Burns, K., Tschopp, J., and Beyaert, R., Regulation of interleukin-1- and lipopolysaccharide-in duced NF-kappaB activation by alternative splicing of MyD88. Curr. Biol., 12, 467–471 (2002).
Johnson, G. B., Brunn, G. J., Kodaira, Y., and Platt, J. L., Receptor-mediated monitoring of tissue well-being via detection of soluble heparan sulfate by Toll-like receptor 4. J. Immunol., 168, 5233–5239 (2002).
Kanayama, A., Seth, R. B., Sun, L., Ea, C. K., Hong, M., Shaito, A., Chiu, Y. H., Deng, L., and Chen, Z. J., TAB2 and TAB3 activate the NF-kappaB pathway through binding to polyubiquitin chains. Mol. Cell, 15, 535–548 (2004).
Kanzler, H., Barrat, F. J., Hessel, E. M., and Coffman, R. L., Therapeutic targeting of innate immunity with Toll-like receptor agonists and antagonists. Nat. Med., 13, 552–559 (2007).
Kaplan, B. I. and Tipirneni, P., Lessons for the future: a review of sepsis past and present. IDrugs, 10, 264–269 (2007).
Kawagoe, T., Sato, S., Matsushita, K., Kato, H., Matsui, K., Kumagai, Y., Saitoh, T., Kawai, T., Takeuchi, O., and Akira, S., Sequential control of Toll-like receptor-dependent responses by IRAK1 and IRAK2. Nat. Immunol., 9, 684–691 (2008).
Kawai, T. and Akira, S., TLR signaling. Semin. Immunol., 19, 24–32 (2007).
Kawai, T., Sato, S., Ishii, K. J., Coban, C., Hemmi, H., Yamamoto, M., Terai, K., Matsuda, M., Inoue, J., Uematsu, S., Takeuchi, O., and Akira, S., Interferon-alpha induction through Toll-like receptors involves a direct interaction of IRF7 with MyD88 and TRAF6. Nat. Immunol., 5, 1061–1068 (2004).
Kayagaki, N., Phung, Q., Chan, S., Chaudhari, R., Quan, C., O’rourke, K. M., Eby, M., Pietras, E., Cheng, G., Bazan, J. F., Zhang, Z., Arnott, D., and Dixit, V. M., DUBA: A deubiquitinase that regulates type I interferon production. Science, 318, 1628–1632 (2007).
Kobayashi, K., Hernandez, L. D., Galan, J. E., Janeway, C. A., Jr., Medzhitov, R., and Flavell, R. A., IRAK-M is a negative regulator of Toll-like receptor signaling. Cell, 110, 191–202 (2002).
Kol, A., Lichtman, A. H., Finberg, R. W., Libby, P., and Kurt-Jones, E. A., Cutting edge: heat shock protein (HSP) 60 activates the innate immune response: CD14 is an essential receptor for HSP60 activation of mononuclear cells. J. Immunol., 164, 13–17 (2000).
Krieg, A. M., Therapeutic potential of Toll-like receptor 9 activation. Nat. Rev. Drug Discov., 5, 471–484 (2006).
Krishnan, J., Selvarajoo, K., Tsuchiya, M., Lee, G., and Choi, S., Toll-like receptor signal transduction. Exp. Mol. Med., 39, 421–438 (2007).
Kuwata, H., Watanabe, Y., Miyoshi, H., Yamamoto, M., Kaisho, T., Takeda, K., and Akira, S., IL-10-inducible Bcl-3 negatively regulates LPS-induced TNF-alpha production in macrophages. Blood, 102, 4123–4129 (2003).
Latz, E., Franko, J., Golenbock, D. T., and Schreiber, J. R., Haemophilus influenzae type b-outer membrane protein complex glycoconjugate vaccine induces cytokine production by engaging human toll-like receptor 2 (TLR2) and requires the presence of TLR2 for optimal immunogenicity. J. Immunol., 172, 2431–2438 (2004).
Lebouder, E., Rey-Nores, J. E., Rushmere, N. K., Grigorov, M., Lawn, S. D., Affolter, M., Griffin, G. E., Ferrara, P., Schiffrin, E. J., Morgan, B. P., and Labeta, M. O., Soluble forms of Toll-like receptor (TLR)2 capable of modulating TLR2 signaling are present in human plasma and breast milk. J. Immunol., 171, 6680–6689 (2003).
Lee, E. G., Boone, D. L., Chai, S., Libby, S. L., Chien, M., Lodolce, J. P., and Ma, A., Failure to regulate TNFinduced NF-kappaB and cell death responses in A20-deficient mice. Science, 289, 2350–2354 (2000).
Lee, M. S. and Kim, Y. J., Signaling pathways downstream of pattern-recognition receptors and their cross talk. Annu. Rev. Biochem., 76, 447–480 (2007).
Liu, B., Liao, J., Rao, X., Kushner, S. A., Chung, C. D., Chang, D. D., and Shuai, K., Inhibition of Stat1-mediated gene activation by PIAS1. Proc. Natl. Acad Sci. USA, 95, 10626–10631 (1998).
Liu, B., Mink, S., Wong, K. A., Stein, N., Getman, C., Dempsey, P. W., Wu, H., and Shuai, K., PIAS1 selectively inhibits interferon-inducible genes and is important in innate immunity. Nat. Immunol., 5, 891–898 (2004).
Liu, B., Yang, Y., Chernishof, V., Loo, R. R., Jang, H., Tahk, S., Yang, R., Mink, S., Shultz, D., Bellone, C. J., Loo, J. A., and Shuai, K., Proinflammatory stimuli induce IKKalphamediated phosphorylation of PIAS1 to restrict inflammation and immunity. Cell, 129, 903–914 (2007).
Mansell, A., Smith, R., Doyle, S. L., Gray, P., Fenner, J. E., Crack, P. J., Nicholson, S. E., Hilton, D. J., O’neill, L. A., and Hertzog, P. J., Suppressor of cytokine signaling 1 negatively regulates Toll-like receptor signaling by mediating Mal degradation. Nat. Immunol., 7, 148–155 (2006).
Masihi, K. N., Lange, W., Brehmer, W., and Ribi, E., Immunobiological activities of nontoxic lipid A: enhancement of nonspecific resistance in combination with trehalose dimycolate against viral infection and adjuvant effects. Int. J. Immunopharmacol, 8, 339–345 (1986).
Mcguirk, P., Mccann, C., and Mills, K. H., Pathogen-specific T regulatory 1 cells induced in the respiratory tract by a bacterial molecule that stimulates interleukin 10 production by dendritic cells: a novel strategy for evasion of protective T helper type 1 responses by Bordetella pertussis. J. Exp. Med., 195, 221–231 (2002).
Medzhitov, R., Preston-Hurlburt, P., Kopp, E., Stadlen, A., Chen, C., Ghosh, S., and Janeway, C. A., Jr., MyD88 is an adaptor protein in the hToll/IL-1 receptor family signaling pathways. Mol. Cell, 2, 253–258 (1998).
Mellor, A. L., Baban, B., Chandler, P. R., Manlapat, A., Kahler, D. J., and Munn, D. H., Cutting edge: CpG oligonucleotides induce splenic CD19+ dendritic cells to acquire potent indoleamine 2,3-dioxygenase-dependent T cell regulatory functions via IFN Type 1 signaling. J. Immunol., 175, 5601–5605 (2005).
Merrell, M. A., Ilvesaro, J. M., Lehtonen, N., Sorsa, T., Gehrs, B., Rosenthal, E., Chen, D., Shackley, B., Harris, K. W., and Selander, K. S., Toll-like receptor 9 agonists promote cellular invasion by increasing matrix metalloproteinase activity. Mol. Cancer Res., 4, 437–447 (2006).
Meyer, T. and Stockfleth, E., Clinical investigations of Tolllike receptor agonists. Expert. Opin. Investig. Drugs, 17, 1051–1065 (2008).
Miyake, K., Yamashita, Y., Hitoshi, Y., Takatsu, K., and Kimoto, M., Murine B cell proliferation and protection from apoptosis with an antibody against a 105-kD molecule: unresponsiveness of X-linked immunodeficient B cells. J. Exp. Med., 180, 1217–1224 (1994).
Motoyama, M., Yamazaki, S., Eto-Kimura, A., Takeshige, K., and Muta, T., Positive and negative regulation of nuclear factor-kappaB-mediated transcription by IkappaBzeta, an inducible nuclear protein. J. Biol. Chem., 280, 7444–7451 (2005).
Mullen, G. E., Giersing, B. K., Ajose-Popoola, O., Davis, H. L., Kothe, C., Zhou, H., Aebig, J., Dobrescu, G., Saul, A., and Long, C. A., Enhancement of functional antibody responses to AMA1-C1/Alhydrogel, a Plasmodium falciparum malaria vaccine, with CpG oligodeoxynucleotide. Vaccine, 24, 2497–2505 (2006).
Negishi, H., Ohba, Y., Yanai, H., Takaoka, A., Honma, K., Yui, K., Matsuyama, T., Taniguchi, T., and Honda, K., Negative regulation of Toll-like-receptor signaling by IRF-4. Proc. Natl. Acad. Sci. USA, 102, 15989–15994 (2005).
Okamoto, M., Oshikawa, T., Tano, T., Ahmed, S. U., Kan, S., Sasai, A., Akashi, S., Miyake, K., Moriya, Y., Ryoma, Y., Saito, M., and Sato, M., Mechanism of anticancer host response induced by OK-432, a streptococcal preparation, mediated by phagocytosis and Toll-like receptor 4 signaling. J. Immunother, 29, 78–86 (2006).
Okamura, Y., Watari, M., Jerud, E. S., Young, D. W., Ishizaka, S. T., Rose, J., Chow, J. C., and Strauss, J. F., 3rd The extra domain A of fibronectin activates Toll-like receptor 4. J. Biol. Chem., 276, 10229–10233 (2001).
Ondiveeran, H. K., and Fox-Robichaud, A., Drug evaluation: E-5564. IDrugs, 7, 582–590 (2004).
Opipari, A. W., Jr., Boguski, M. S., and Dixit, V. M., The A20 cDNA induced by tumor necrosis factor alpha encodes a novel type of zinc finger protein. J. Biol. Chem., 265, 14705–14708 (1990).
Palsson-Mcdermott, E. M., Doyle, S. L., Mcgettrick, A. F., Hardy, M., Husebye, H., Banahan, K., Gong, M., Golenbock, D., Espevik, T., and O’neill, L. A., TAG, a splice variant of the adaptor TRAM, negatively regulates the adaptor MyD88-independent TLR4 pathway. Nat. Immunol., 10, 579–586 (2009).
Park, J. S., Svetkauskaite, D., He, Q., Kim, J. Y., Strassheim, D., Ishizaka, A., and Abraham, E., Involvement of toll-like receptors 2 and 4 in cellular activation by high mobility group box 1 protein. J. Biol. Chem., 279, 7370–7377 (2004).
Rappuoli, R., Bridging the knowledge gaps in vaccine design. Nat. Biotechnol., 25, 1361–1366 (2007).
Robinson, R. A., Devita, V. T., Levy, H. B., Baron, S., Hubbard, S. P., and Levine, A. S., A phase I-II trial of multiple-dose polyriboinosic-polyribocytidylic acid in patieonts with leukemia or solid tumors. J. Natl. Cancer Inst., 57, 599–602 (1976).
Rottapel, R., Ilangumaran, S., Neale, C., La Rose, J., Ho, J. M., Nguyen, M. H., Barber, D., Dubreuil, P., and De Sepulveda, P., The tumor suppressor activity of SOCS-1. Oncogene, 21, 4351–4362 (2002).
Ryo, A., Liou, Y. C., Lu, K. P., and Wulf, G., Prolyl isomerase Pin1: A catalyst for oncogenesis and a potential therapeutic target in cancer. J. Cell Sci., 116, 773–783 (2003a).
Ryo, A., Liou, Y. C., Wulf, G., Nakamura, M., Lee, S. W., and Lu, K. P., PIN1 is an E2F target gene essential for Neu/Ras-induced transformation of mammary epithelial cells. Mol. Cell Biol., 22, 5281–5295 (2002).
Ryo, A., Nakamura, M., Wulf, G., Liou, Y. C., and Lu, K. P., Pin1 regulates turnover and subcellular localization of beta-catenin by inhibiting its interaction with APC. Nat. Cell Biol., 3, 793–801 (2001).
Ryo, A., Suizu, F., Yoshida, Y., Perrem, K., Liou, Y. C., Wulf, G., Rottapel, R., Yamaoka, S., and Lu, K. P., Regulation of NF-kappaB signaling by Pin1-dependent prolyl isomerization and ubiquitin-mediated proteolysis of p65/RelA. Mol. Cell, 12, 1413–1426 (2003b).
Saitoh, T., Tun-Kyi, A., Ryo, A., Yamamoto, M., Finn, G., Fujita, T., Akira, S., Yamamoto, N., Lu, K. P., and Yamaoka, S., Negative regulation of interferon-regulatory factor 3-dependent innate antiviral response by the prolyl isomerase Pin1. Nat. Immunol., 7, 598–605 (2006).
Schroder, N. W. and Schumann, R. R., Single nucleotide polymorphisms of Toll-like receptors and susceptibility to infectious disease. Lancet Infect. Dis., 5, 156–164 (2005).
Seiwert, S. D., Andrews, S. W., Jiang, Y., Serebryany, V., Tan, H., Kossen, K., Rajagopalan, P. T., Misialek, S., Stevens, S. K., Stoycheva, A., Hong, J., Lim, S. R., Qin, X., Rieger, R., Condroski, K. R., Zhang, H., Do, M. G., Lemieux, C., Hingorani, G. P., Hartley, D. P., Josey, J. A., Pan, L., Beigelman, L., and Blatt, L. M., Preclinical characteristics of the hepatitis C virus NS3/4A protease inhibitor ITMN-191 (R7227). Antimicrob. Agents Chemother., 52, 4432–4441 (2008).
Sharrocks, A. D., PIAS proteins and transcriptional regulation—more than just SUMO E3 ligases? Genes Dev., 20, 754–758 (2006).
Shi, M., Deng, W., Bi, E., Mao, K., Ji, Y., Lin, G., Wu, X., Tao, Z., Li, Z., Cai, X., Sun, S., Xiang, C., and Sun, B., TRIM30 alpha negatively regulates TLR-mediated NF-kappa B activation by targeting TAB2 and TAB3 for degradation. Nat. Immunol., 9, 369–377 (2008).
Singh-Jasuja, H., Scherer, H. U., Hilf, N., Arnold-Schild, D., Rammensee, H. G., Toes, R. E., and Schild, H., The heat shock protein gp96 induces maturation of dendritic cells and down-regulation of its receptor. Eur. J. Immunol., 30, 2211–2215 (2000).
Smiley, S. T., King, J. A., and Hancock, W. W., Fibrinogen stimulates macrophage chemokine secretion through tolllike receptor 4. J. Immunol., 167, 2887–2894 (2001).
Stack, J., Haga, I. R., Schroder, M., Bartlett, N. W., Maloney, G., Reading, P. C., Fitzgerald, K. A., Smith, G. L., and Bowie, A. G., Vaccinia virus protein A46R targets multiple Toll-like-interleukin-1 receptor adaptors and contributes to virulence. J. Exp. Med., 201, 1007–1018 (2005).
Stark, G. R., Kerr, I. M., Williams, B. R., Silverman, R. H., and Schreiber, R. D., How cells respond to interferons. Annu. Rev. Biochem., 67, 227–264 (1998).
Uematsu, S., Sato, S., Yamamoto, M., Hirotani, T., Kato, H., Takeshita, F., Matsuda, M., Coban, C., Ishii, K. J., Kawai, T., Takeuchi, O., and Akira, S., Interleukin-1 receptorassociated kinase-1 plays an essential role for Toll-like receptor (TLR)7- and TLR9-mediated interferon-{alpha} induction. J. Exp. Med., 201, 915–923 (2005).
Vollmer, J., TLR9 in health and disease. Int. Rev. Immunol., 25, 155–181 (2006).
Wang, J. H., Manning, B. J., Wu, Q. D., Blankson, S., Bouchier-Hayes, D., and Redmond, H. P., Endotoxin/lipopolysaccharide activates NF-kappa B and enhances tumor cell adhesion and invasion through a beta 1 integrin-dependent mechanism. J. Immunol., 170, 795–804 (2003).
Whitmore, M. M., Iparraguirre, A., Kubelka, L., Weninger, W., Hai, T., and Williams, B. R., Negative regulation of TLR-signaling pathways by activating transcription factor-3. J. Immunol., 179, 3622–3630 (2007).
Wingender, G., Garbi, N., Schumak, B., Jungerkes, F., Endl, E., Von Bubnoff, D., Steitz, J., Striegler, J., Moldenhauer, G., Tuting, T., Heit, A., Huster, K. M., Takikawa, O., Akira, S., Busch, D. H., Wagner, H., Hammerling, G. J., Knolle, P. A., and Limmer, A., Systemic application of CpG-rich DNA suppresses adaptive T cell immunity via induction of IDO. Eur. J. Immunol., 36, 12–20 (2006).
Xiang, A. X., Webber, S. E., Kerr, B. M., Rueden, E. J., Lennox, J. R., Haley, G. J., Wang, T., Ng, J. S., Herbert, M. R., Clark, D. L., Banh, V. N., Li, W., Fletcher, S. P., Steffy, K. R., Bartkowski, D. M., Kirkovsky, L. I., Bauman, L. A., and Averett, D. R., Discovery of ANA975: an oral prodrug of the TLR-7 agonist isatoribine. Nucleosides Nucleotides Nucleic Acids, 26, 635–640 (2007).
Yamamoto, M., Sato, S., Hemmi, H., Hoshino, K., Kaisho, T., Sanjo, H., Takeuchi, O., Sugiyama, M., Okabe, M., Takeda, K., and Akira, S., Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science, 301, 640–643 (2003a).
Yamamoto, M., Sato, S., Hemmi, H., Sanjo, H., Uematsu, S., Kaisho, T., Hoshino, K., Takeuchi, O., Kobayashi, M., Fujita, T., Takeda, K., and Akira, S., Essential role for TIRAP in activation of the signalling cascade shared by TLR2 and TLR4. Nature, 420, 324–329 (2002).
Yamamoto, M., Sato, S., Hemmi, H., Uematsu, S., Hoshino, K., Kaisho, T., Takeuchi, O., Takeda, K., and Akira, S., TRAM is specifically involved in the Toll-like receptor 4-mediated MyD88-independent signaling pathway. Nat. Immunol., 4, 1144–1150 (2003b).
Yamazaki, S., Muta, T., and Takeshige, K., A novel IkappaB protein, IkappaB-zeta, induced by proinflammatory stimuli, negatively regulates nuclear factor-kappaB in the nuclei. J. Biol. Chem., 276, 27657–27662 (2001).
Yoder, A., Wang, X., Ma, Y., Philipp, M. T., Heilbrun, M., Weis, J. H., Kirschning, C. J., Wooten, R. M., and Weis, J. J., Tripalmitoyl-S-glyceryl-cysteine-dependent OspA vaccination of toll-like receptor 2-deficient mice results in effective protection from Borrelia burgdorferi challenge. Infect. Immun., 71, 3894–3900 (2003).
Yoshida, H., Jono, H., Kai, H., and Li, J. D., The tumor suppressor cylindromatosis (CYLD) acts as a negative regulator for toll-like receptor 2 signaling via negative cross-talk with TRAF6 AND TRAF7. J. Biol. Chem., 280, 41111–41121 (2005).
Yoshikawa, H., Matsubara, K., Qian, G. S., Jackson, P., Groopman, J. D., Manning, J. E., Harris, C. C., and Herman, J. G., SOCS-1, a negative regulator of the JAK/STAT pathway, is silenced by methylation in human hepatocellular carcinoma and shows growth-suppression activity. Nat. Genet., 28, 29–35 (2001).
Yu, D., Wang, D., Zhu, F. G., Bhagat, L., Dai, M., Kandimalla, E. R., and Agrawal, S., Modifications Incorporated in CpG Motifs of Oligodeoxynucleotides Lead to Antagonist Activity of Toll-like Receptors 7 and 9. J. Med. Chem., 52, 5108–5114 (2009).
Yuan, Z. Q., Feldman, R. I., Sussman, G. E., Coppola, D., Nicosia, S. V., and Cheng, J. Q., AKT2 inhibition of cisplatin-induced JNK/p38 and Bax activation by phosphorylation of ASK1: implication of AKT2 in chemoresistance. J. Biol. Chem., 278, 23432–23440 (2003).
Zhang, G. and Ghosh, S., Negative regulation of toll-like receptor-mediated signaling by Tollip. J. Biol. Chem., 277, 7059–7065 (2002).
Zhang, X., Meng, Z., Qiu, S., Xu, Y., Yang, D., Schlaak, J. F., Roggendorf, M., and Lu, M., Lipopolysaccharide induced innate immune responses in primary hepatocytes downregulates woodchuck hepatitis virus replication via interferon-independent pathways. Cell Microbiol., 11, 1624–1637 (2009).
Zhu, X., Chang, M. S., Hsueh, R. C., Taussig, R., Smith, K. D., Simon, M. I., and Choi, S., Dual ligand stimulation of RAW 264.7 cells uncovers feedback mechanisms that regulate TLR-mediated gene expression. J. Immunol., 177, 4299–4310 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krishnan, J., Lee, G. & Choi, S. Drugs targeting toll-like receptors. Arch. Pharm. Res. 32, 1485–1502 (2009). https://doi.org/10.1007/s12272-009-2100-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-009-2100-6