Abstract
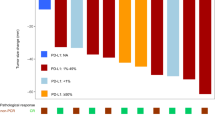
A phase II clinical trial was performed to evaluate the efficacy and safety of gefitinib on pretreated Chinese female non-small-cell lung cancer (NSCLC) patients. Chinese female patients with locally advanced or metastatic NSCLC who failed at least one platinum-based chemotherapy received gefitinib monotherapy (250 mg/day) between April 2002 and January 2010. The primary endpoint was overall response rate (ORR), and secondary endpoints were overall survival (OS) and progression-free survival (PFS). Of the 40 evaluable female patients, the ORR was 62.5%. All patients have responded with one (2.5%) complete response, 24 (60%) partial response, 12 (30%) stable disease, and 3 (7.5%) progressive disease. The OS and PFS were 20 months (95% CI: 11.9–28 months) and 13 months (95% CI: 8.0–17.9 months), respectively. Survival (OS and PFS) were longer in patients with good performance status and in patients older than 65 years (P < 0.05). The most frequently observed toxicities were rash/dry skin (80%), diarrhea (42.5%), and vomiting/anorexia (32.5%). Four patients developed grade 3 toxicities (rash and diarrhea) but did not require either dose reduction or discontinuation. Gefitinib is a highly effective and well-tolerated agent for Chinese women with pretreated advanced NSCLC.


Similar content being viewed by others
References
Yang L, Li LD, Chen YD, Parkin DM. Mortality time trends and the incidence and mortality estimation and projection for lung cancer in China. Chin J Lung Cancer. 2005;8(4):274–8.
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55(2):74–108.
Lee CT. Epidemiology of lung cancer in Korea. Cancer Res Treat. 2002;34:3–7.
Parkin DM. Global cancer statistics in the year 2000. Lancet Oncol. 2001;2:533–43.
Thomas L, Doyle LA, Edelman MJ. Lung cancer in women: emerging differences in epidemiology, biology, and therapy. Chest. 2005;128:370–81.
Chun BY, Yang JH, Song JH, Lim JS. Annual average cancer incidence and trend of cancer incidence in Daegu for 5 years, 1997–2001. Korean J Epidemiol. 2007;29:59–69.
Yang GH, Wang JF, Wan X, Wang LJ, Chen AP. Quantitative analysis of factors affected mortality trend in Chinese, 2002. Chin J Epidemiol. 2005;26(12):934–8.
Patel JD. Lung cancer in women. J Clin Oncol. 2005;23(14):3212–8.
Ries LAG, Hankey BF, Miller BA, Hartman AM, Edwards BK. Cancer statistics review 1973–88. Bethesda, MD: National Cancer Institute; 1991.
Radzikowska E, Glaz P, Roszkowski K. Lung cancer in women: age, smoking, histology, performance status, stage, initial treatment and survival. Population based study of 20 561 cases. Ann Oncol. 2002;13:1087–93.
Minami H, Yoshimura M, Miyamoto Y, Matsuoka H, Tsubota N. Lung cancer in women: sex-associated differences in survival of patients undergoing resection for lung cancer. Chest. 2000;118:1603–9.
Rosell R, et al. Screening for epidermal growth factor receptor mutations in lung cancer. N Engl J Med. 2009;361(10):958–67.
Mitsudomi T, et al. Mutations of the epidermal growth factor receptor gene predict prolonged survival after gefitinib treatment in patients with non-small-cell lung cancer with postoperative recurrence. J Clin Oncol. 2005;23:2513–20.
Taron M, et al. Activating mutations in the tyrosine kinase domain of the epidermal growth factor receptor are associated with improved survival in gefitinib-treated chemorefractory lung adenocarcinomas. Clin Cancer Res. 2005;11:5878–85.
Han SW, et al. Predictive and prognostic impact of epidermal growth factor receptor mutation in non-small-cell lung cancer patients treated with gefitinib. J Clin Oncol. 2005;23:2493–501.
Takano T, et al. Epidermal growth factor receptor gene mutations and increased copy numbers predict gefitinib sensitivity in patients with recurrent non-small-cell lung cancer. J Clin Oncol. 2005;23:6829–37.
Paez JG, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304:1497–500.
Bell DW, et al. Epidermal growth factor receptor mutations and gene amplification in non-small-cell lung cancer: molecular analysis of the IDEAL/INTACT gefitinib trials. J Clin Oncol. 2005;23:8081–92.
Lynch TJ, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non–small-cell lung cancer to gefitinib. N Engl J Med. 2004;350:2129–39.
Kim ES, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomised phase III trial. Lancet. 2008;372(9652):1809–18.
Maemondo M, et al. Gefitinib or chemotherapy for non–small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362(25):2380–8.
Tiseo M, Bartolotti M, Gelsomino F, Bordi P. Emerging role of gefitinib in the treatment of non-small-cell lung cancer (NSCLC). Drug Des Dev Ther. 2010;4:81–98.
Li J, et al. CYP3A phenotyping approach to predict systemic exposure to EGFR tyrosine kinase inhibitors. J Natl Cancer Inst. 2006;98(23):1714–23.
Myrand SP, et al. Pharmacokinetics/genotype associations for major cytochrome P450 enzymes in native and first- and third-generation Japanese populations: comparison with Korean, Chinese, and Caucasian populations. Clin Pharmacol Ther. 2008;84(3):347–61.
Wildt SN, Kearns GL, Leeder JS, van den Anker JN. Cytochrome P450 3A: ontogeny and drug disposition. Clin Pharmacokinet. 1999;37(6):485–505.
Ranson M, et al. ZD1839, a selective oral epidermal growth factor receptor-tyrosine kinase inhibitor, is well tolerated and active in patients with solid malignant tumours: results of a phase I trial. J Clin Oncol. 2002;20:2240–50.
Nakagawa K, et al. Phase I pharmacokinetic trial of the selective oral epidermal growth factor receptor tyrosine kinase inhibitor gefitinib (‘Iressa’, ZD1839) in Japanese patients with solid malignant tumors. Ann Oncol. 2003;14(6):922–30.
Cohen MH, Williams GA, Sridhara R, Chen G, Pazdur R. FDA drug approval summary: gefitinib (ZD1839) (Iressa) tablets. Oncologist. 2003;8:303–6.
Ando M, et al. Predictive factors for interstitial lung disease, antitumor response, and survival in non-small-cell lung cancer patients treated with gefitinib. J Clin Oncol. 2006;24:2549–56.
Kudoh S, et al. Interstitial lung disease in Japanese patients with lung cancer: a cohort and nested case-control study. Am J Respir Crit Care Med. 2008;177:1348–57.
Yoshida S. The results of gefitinib prospective investigation. Med Drug J. 2005;41:772–89.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Deng, J., Fang, W.J., Zhang, X.C. et al. Phase II trial of gefitinib in pretreated Chinese women with advanced non-small-cell lung cancer. Med Oncol 29, 595–599 (2012). https://doi.org/10.1007/s12032-011-9891-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-9891-2