Abstract
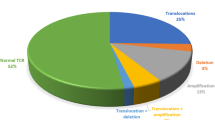
Contemporary protocols ensure high-remission rate and long-term free survival in children with acute lymphoblastic leukemia (ALL), but small percentage of patients is still incurable. Molecular genetic methods helped to establish submicroscopic classification as well as minimal residual disease follow-up, considered to be responsible for relapse. Our study enrolled 70 pediatric patients with de novo ALL, analyzed using reverse transcriptase-polymerase chain reaction for the presence of four major risk-stratifying translocations (BCR/ABL, MLL/AF4, TEL/AML1, and E2A/PBX1). Bone marrow samples were collected at diagnosis, at the end of induction phase, and after intensive chemotherapy with the aim to establish the correlation between chromosomal aberration, clinical features, and treatment response. Presenting the results of this study, we offer another evidence of variable incidence and clinical characteristics of ALL subtypes.
Similar content being viewed by others
References
Silverman LB, et al. Improved outcome for children with acute lymphoblastic leukemia: results of Dana-Faber Consortium Protocol 91–01. Blood. 2001;97:1211–8. doi:10.1182/blood.V97.5.1211.
Mitelman F, Johansson B, Mertens F. The impact of translocations and gene fusions on cancer causation. Nat Rev Cancer. 2007;7(4):233–5. doi:10.1038/nrc2091.
Jones LK, Saha V. Philadelphia positive acute lymphoblastic leukaemia of childhood. Br J Haematol. 2005;130:489–500. doi:10.1111/j.1365-2141.2005.05611.x.
Pui Ch, et al. Clinical characteristics and treatment outcome of childhood lymphoblastic leukemia with t (4;11)(q21:q23): a collaborative study of 40 cases. Blood. 1991;77:440–7.
Zelent A, Greaves M, Enver T. Role of the TEL-AML1 fusion gene in the molecular pathogenesis of childhood acute lymphoblastic leukaemia. Oncogene. 2004;23:4275–83. doi:10.1038/sj.onc.1207672.
Wiemels JL, et al. Site-specific translocation and evidence of postnatal origin of the t(1;19) E2A-PBX1 fusion in childhood acute lymphoblastic leukemia. Proc Natl Acad Sci USA. 2002;99:15101–6. doi:10.1073/pnas.222481199.
Van Dongen JJM, et al. Standardized RT-PCR analysis of fusion gene transcripts from chromosome aberrations in acute leukemia for detection of minimal residual disease. Report of the BIOMED-1 Concerted Action: Investigation of minimal residual disease in acute leukemia. Leukemia. 1999;13:1901–28. doi:10.1038/sj/leu/2401592.
Schlieben S, et al. Incidence and clinical outcome of children with BCR/ABL positive acute lymphoblastic leukemia (ALL). A prospective RT-PCR study based on 673 patients enrolled in the German pediatric multicenter therapy trials ALLBFM-90 and CoALL-05–92. Leukemia. 1996;10:957–63.
Gurbuxani S, et al. Detection of BCR/ABL transcripts in acute lymphoblastic leukemia in Indian patients. Leuk Res. 1998;22:77–80. doi:10.1016/S0145-2126(97)87415-2.
Iqbal Z, Iqbal M, Akhter T. Frequency of BCR/ABL fusion oncogene in Pakistani Childhood acute lymphoblastic leukemia (ALL). Patients reflect ethnic differences in molecular genetics of ALL. J Pediatr Hematol Oncol. 2007;29:585. doi:10.1097/MPH.0b013e3180f61bcf.
Silva ML, et al. M-BCR rearrangement in a case of T-cell childhood acute lymphoblastic leukemia. Med Pediatr Oncology. 1999;32:455–6. doi:10.1002/(SICI)1096-911X(199906)32:6<455::AID-MPO15>3.0.CO;2-S.
Lo Nigro L, et al. Association of cytogenetic abnormalities with detection of BCR/ABL fusion transcripts in children with T-lineage lymphoproliferative diseases (T-ALL and T-NHL). Pediatr Blood Cancer. 2004;42:278–80. doi:10.1002/pbc.10453.
Tchirkov A, et al. Molecular detection of a late-appearing BCR/ABL gene in a child with T-cell acute lymphoblastic leukemia. Ann Hematol. 1998;77:55–9. doi:10.1007/s002770050412.
Arico M, et al. Outcome of treatment in children with Philadelphia chromosome-positive acute lymphoblastic leukemia. N Engl J Med. 2000;342:998–1006. doi:10.1056/NEJM200004063421402.
Saglio G, et al. Consistent amounts of acute leukemia-associated P190BCR/ABL transcripts are expressed by chronic myelogenous leukemia patients at diagnosis. Blood. 1996;87:1075–80.
Serrano J, et al. Molecular analysis of lineage-specific chimerism and minimal residual disease by RT-PCR of p210 (BCR-ABL) and p190 (BCR-ABL) after alogenic bone marrow transplantation for chronic myeloid leukemia: increasing mixed myeloid chimerism and p190 (BCR-ABL) detection precede cytogenetic relapse. Blood. 2000;95:2659–65.
Alvarado Y, Apostolidou E, Swords R, Giles FJ. Emerging therapeutic options for Philadelphia-positive acute lymphocytic leukemia. Expert Opin Emerg Drugs. 2007;12(1):165–79. doi:10.1517/14728214.12.1.165.
Codrington R, et al. Analysis of ETV6/AML1 abnormalities in acute lymphoblastic leukemia: incidence, alternative spliced forms and minimal residual disease value. Br J Haematol. 2000;111:1071–9. doi:10.1046/j.1365-2141.2000.02464.x.
Jimenez-Morales S, Miranda-Peralta E, Saldana-Alvarez Y. BCR-ABL, ETV6-RUNX1 and E2A-PBX1: prevalence of the most common acute lymphoblastic leukemia fusion genes in Mexican patients. Leuk Res. 2008;32:1518–22. doi:10.1016/j.leukres.2008.03.021.
Borkhardt A, et al. Incidence and clinical relevance of TEL/AML1 fusion genes in children with acute lymphoblastic leukemia enrolled in the German and Italian multicenter therapy trials. Blood. 1997;90(2):571–7.
Shurtleff SA, et al. TEL/AML1 fusion resulting from a cryptic t(12;21) in the most common genetic lesion in pediatric ALL and defines a subgroup of patients with an excellent prognosis. Leukemia. 1995;9:1985–9.
Lanza C, et al. Persistence of E2A/PBX1 transcripts in t(l;19) childhood acute lymphoblastic leukemia: correlation with chemotherapy intensity and clinical outcome. Leuk Res. 1996;20(5):441–3. doi:10.1016/0145-2126(95)00115-8.
Hunger SP, et al. E2A-PBX1 chimeric transcript status at end of consolidation in not predictive of treatment outcome in childhood acute lymphoblastic leukemias with t(1;19)(q23;p13), a Pediatric Oncology Group study. Blood. 1998;91:1021–8.
Mann G, Cazzaniga G, Van der Velden VHJ. Acute lymphoblastic leukemia with t(4;11) in children 1 year and older: The ‘big sister’ of the infant disease? Leukemia. 2007;21:642–6.
Garcia-Sanz R, et al. Low frequency of the TEL/AML1 fusion gene in acute lymphoblastic leukaemia in Spain. Br J Hematol. 1999;107:667–669. doi:10.1046/j.1365-2141.1999.01747.x.
Acknowledgments
This work was supported by Ministry of Science, Republic of Serbia (Grant No. 143051).
Author information
Authors and Affiliations
Corresponding author
Additional information
J. Lazic and N. Tosic equally contributed to the manuscript.
Rights and permissions
About this article
Cite this article
Lazic, J., Tosic, N., Dokmanovic, L. et al. Clinical features of the most common fusion genes in childhood acute lymphoblastic leukemia. Med Oncol 27, 449–453 (2010). https://doi.org/10.1007/s12032-009-9232-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-009-9232-x