Abstract
Background
Cerebral vasospasm is a significant cause of morbidity in patients after aneurysmal subarachnoid hemorrhage (aSAH). There are few effective treatments. The search for new treatments has focused predominantly on dilating cerebral blood vessels. Growing evidence supports a role for inflammation in its pathogenesis but no potential target for intervention has emerged.
Methods
CSF and clinical information from patients with aSAH were collected. Additionally, tyrosine modifications by stable isotope dilution HPLC with online tandem mass spectrometry were quantified in CSF samples.
Results
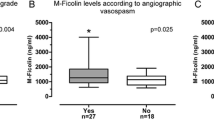
We report an association between neutrophil accumulation in the cerebrospinal fluid of patients with aSAH and the development of vasospasm. In particular, CSF neutrophil content of >62% on the third day after aSAH is an independent predictor of the later development of vasospasm (OR 6.8, 95% CI 2.0–23.3, P = 0.002). Further, activity of myeloperoxidase and NADPH oxidase is elevated in aSAH suggesting a role for modification of CSF proteins by reactive oxidant species.
Conclusions
Neutrophil percentage is an independent predictor of vasospasm in aSAH patients, days prior to its onset suggesting a role of neutrophils in vasospasm. The activity of neutrophil enzymes is also increased suggesting a mechanism for blood vessel damage. Inflammation mediated by neutrophils is a potential target for therapies in vasospasm. More study is necessary to determine the mechanism by which neutrophils damage cerebral blood vessels.


Similar content being viewed by others
Abbreviations
- HOCl:
-
Hypochlorous acid
- HOBr:
-
Hypobromous acid
- LC/ESI/MS/MS:
-
Liquid chromatography electrospray, ionization tandem mass spectrometry
- EPO:
-
Eosinophil peroxidase
- MPO:
-
Myeloperoxidase
References
van Gijn J, Rinkel GJ. Subarachnoid haemorrhage: diagnosis, causes and management. Brain. 2001;124:249–78.
Provencio JJ, Vora N. Subarachnoid hemorrhage and inflammation: bench to bedside and back. Semin Neurol. 2005;25:435–44.
Clatterbuck RE, Oshiro EM, Hoffman PA, Dietsch GN, Pardoll DM, Tamargo RJ. Inhibition of vasospasm with lymphocyte function-associated antigen-1 monoclonal antibody in a femoral artery model in rats. J Neurosurg. 2002;97:676–82.
Chaichana KL, Levy AP, Miller-Lotan R, Shakur S, Tamargo RJ. Haptoglobin 2–2 genotype determines chronic vasospasm after experimental subarachnoid hemorrhage. Stroke. 2007;38:3266–71.
Pradilla G, Wang PP, Legnani FG, Ogata L, Dietsch GN, Tamargo RJ. Prevention of vasospasm by anti-CD11/CD18 monoclonal antibody therapy following subarachnoid hemorrhage in rabbits. J Neurosurg. 2004;101:88–92.
Naruko T, Ueda M, Haze K, et al. Neutrophil infiltration of culprit lesions in acute coronary syndromes. Circulation. 2002;106:2894–900.
Brennan ML, Hazen SL. Emerging role of myeloperoxidase and oxidant stress markers in cardiovascular risk assessment. Curr Opin Lipidol. 2003;14:353–9.
Abu-Soud HM, Hazen SL. Nitric oxide is a physiological substrate for mammalian peroxidases. J Biol Chem. 2000;275:37524–32.
Eiserich JP, Baldus S, Brennan ML, et al. Myeloperoxidase, a leukocyte-derived vascular NO oxidase. Science. 2002;296:2391–4.
Vita JA, Brennan ML, Gokce N, et al. Serum myeloperoxidase levels independently predict endothelial dysfunction in humans. Circulation. 2004;110:1134–9.
Frontera JA, Claassen J, Schmidt JM, et al. Prediction of symptomatic vasospasm after subarachnoid hemorrhage: the modified fisher scale. Neurosurgery. 2006;59:21–7. discussion 7.
Brennan ML, Wu W, Fu X, et al. A tale of two controversies: defining both the role of peroxidases in nitrotyrosine formation in vivo using eosinophil peroxidase and myeloperoxidase-deficient mice, and the nature of peroxidase-generated reactive nitrogen species. J Biol Chem. 2002;277:17415–27.
Weir B, Grace M, Hansen J, Rothberg C. Time course of vasospasm in man. J Neurosurg. 1978;48:173–8.
Neil-Dwyer G, Cruickshank J. The blood leucocyte count and its prognostic significance in subarachnoid haemorrhage. Brain. 1974;97:79–86.
Niikawa S, Hara S, Ohe N, Miwa Y, Ohkuma A. Correlation between blood parameters and symptomatic vasospasm in subarachnoid hemorrhage patients. Neurol Med Chir (Tokyo). 1997;37:881–4. discussion 4–5.
Maiuri F, Gallicchio B, Donati P, Carandente M. The blood leukocyte count and its prognostic significance in subarachnoid hemorrhage. J Neurosurg Sci. 1987;31:45–8.
Fassbender K, Hodapp B, Rossol S, et al. Inflammatory cytokines in subarachnoid haemorrhage: association with abnormal blood flow velocities in basal cerebral arteries. J Neurol Neurosurg Psychiatry. 2001;70:534–7.
Takizawa T, Tada T, Kitazawa K, et al. Inflammatory cytokine cascade released by leukocytes in cerebrospinal fluid after subarachnoid hemorrhage. Neurol Res. 2001;23:724–30.
Polin RS, Bavbek M, Shaffrey ME, et al. Detection of soluble E-selectin, ICAM-1, VCAM-1, and L-selectin in the cerebrospinal fluid of patients after subarachnoid hemorrhage. J Neurosurg. 1998;89:559–67.
Dhar R, Diringer MN. The burden of the systemic inflammatory response predicts vasospasm and outcome after subarachnoid hemorrhage. Neurocrit Care. 2008;8:404–12.
Lin C-L, Dumont AS, Calisaneller T, Kwan A-L, Hwong S-L, Lee KS. Monoclonal antibody against E selectin attenuates subarachnoid hemorrhage-induced cerebral vasospasm. Surg Neurol. 2005;64:201–5. discussion 5–6.
Lebkowska U, Malyszko J, Lebkowska A, et al. Neutrophil gelatinase-associated lipocalin and cystatin C could predict renal outcome in patients undergoing kidney allograft transplantation: a prospective study. Transplant Proc. 2009;41:154–7.
Turunen AJ, Lindgren L, Salmela KT, et al. Association of graft neutrophil sequestration with delayed graft function in clinical renal transplantation. Transplantation. 2004;77:1821–6.
Devouassoux G, Drouet C, Pin I, et al. Alveolar neutrophilia is a predictor for the bronchiolitis obliterans syndrome, and increases with degree of severity. Transpl Immunol. 2002;10:303–10.
Trabold B, Rothoerl R, Wittmann S, Woertgen C, Frohlich D. Cerebrospinal fluid and neutrophil respiratory burst after subarachnoid hemorrhage. Neuroimmunomodulation. 2005;12:152–6.
Provencio JJ, Kivisakk P, Tucky BH, Luciano MG, Ransohoff RM. Comparison of ventricular and lumbar cerebrospinal fluid T cells in non-inflammatory neurological disorder (NIND) patients. J Neuroimmunol. 2005;163:179–84.
Sommer JB, Gaul C, Heckmann J, Neundorfer B, Erbguth FJ. Does lumbar cerebrospinal fluid reflect ventricular cerebrospinal fluid? A prospective study in patients with external ventricular drainage. Eur Neurol. 2002;47:224–32.
Fukuda S, Hashimoto N, Naritomi H, et al. Prevention of rat cerebral aneurysm formation by inhibition of nitric oxide synthase. Circulation. 2000;101:2532–8.
Acknowledgments
The authors would like to thank Michael Chow, MD, MPH, University of Alberta for his help with the 2000–2002 database; Rishi Gupta, MD for statistical help and review of the manuscript; and Edward Manno, MD and Saksith Smithason, MD for review of the manuscript. This work was supported by National Institutes of Health Grants K08 NS051350 (to J.J.P), and P01 HL076491 (to S.L.H.), and K24 NS51400 (to R.M.R).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Provencio, J.J., Fu, X., Siu, A. et al. CSF Neutrophils Are Implicated in the Development of Vasospasm in Subarachnoid Hemorrhage. Neurocrit Care 12, 244–251 (2010). https://doi.org/10.1007/s12028-009-9308-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-009-9308-7