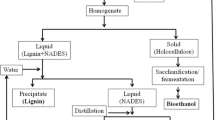
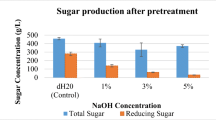
Abstract
In this paper, two new strians, Issatchenkia occidentalis (Lj-3, CCTCC M 2006097) and Issatchenkia orienalis (S-7, CCTCC M 2006098), isolated from different environments on solid media, were used in the detoxification process of the hemicellulosic hydrolysate of sugarcane bagasse. High-pressure liquid chromatography elution curve of UV-absorption compounds represented by acetic acid, furfural, and guaiacol (toxic compounds found in the hemicellulosic hydrolysate) showed that several chromatographic peaks were evidently diminished for the case of detoxified hydrolysate with isolate strains compared to the high peaks resulted for no detoxified hydrolysate. It was clear that these inhibitors were degraded by the two new isolates during their cultivation process. Fermentation results for the biodetoxified hydrolysate showed an increase in xylitol productivity (Q p) by 1.97 and 1.95 times (2.03 and 2.01 g l−1 h−1) and in xylitol yield (Y p) by 1.72 and 1.65 times (0.93 and 0.89 g xylitol per gram xylose) for hydrolysate treated with S-7 and Lj-3, respectively, in comparison with no detoxified hydrolysate (1.03 g l−1 h−1 and 0.54 g xylitol per gram xylose). This present work demonstrated the importance of Issatchenkia yeast in providing an effective biological detoxification approach to remove inhibitors and improve hydrolysate fermentability, leading to a high xylitol productivity and yield.





Similar content being viewed by others
References
Sarrouh, B. F., Santos, D. T., & Silva, S. S. (2007). BTJ, 2(6), 759–763.
Silva, S. S., Converti, A., Zilli, M., Sene, L., & Felipe, M. G. A. (2001). Applied Biochemistry and Biotechnology, 57, 738–743.
Silva, S. S., & Afschar, A. S. (1994). Bioprocess Engineering, 11, 129–34.
Silva, S. S., Santos, J. C., Carvalho, W., Aracava, K. K., & Vitolo, M. (2003). Process Biochemistry, 38(6), 903–907.
Hsu, T. (1996) Handbook on bioethanol production and utilization. In: Wyman CE (Ed) Washington, DC: Taylor and Francis, pp. 179–212
Sun, Y., & Cheng, J. (2002). Bioresource Technology, 83, 1–11.
Parajó, J. C., Dominguez, H., & Dominguez, J. M. (1998). Bioresource Technology, 66, 25–40.
Palmqvist, E., & Hahn-Hagerdal, B. (2000). Bioresource Technology, 74, 25–33.
Berson, R. E., Young, J. S., & Kamer, S. N. (2005). Applied Biochemistry and Biotechnology, 123, 923–934.
Agblevor, F. A., Fu, J., & Hames, B. (2004). Applied Biochemistry and Biotechnology, 119, 97–120.
Carvalhero, F., Duarte, L. C., & Lopes, S. (2005). Process Biochemistry, 40, 1215–1223.
Mussatto, S. I., & Roberto, I. C. (2004). Bioresource Technology, 93, 1–10.
Carvalho, G. B. M., Mussatto, S. I., & Cândido, E. J. (2006). Journal of Chemical Technology and Biotechnology, 81, 152–157.
López, M. J., Nichols, N. N., Deien, B. B., Moreno, J., & Bothast, R. J. (2004). Applied Microbiology and Biotechnology, 64, 125–131.
Peterson, S. W., & Kurtzman, C. P. (1991). Systematic and Applied Microbiology, 14, 124–129.
Raeder, U., & Broda, P. (1985). Applied Microbiology, 1, 17–20.
Guadet, J., Julien, J., Lafey, J. F., & Brygoo, Y. (1989). Molecular Biology and Evolution, 6, 227–242.
Kurtzman, C. P., & Robnett, C. J. (1997). Journal of Clinical Microbiology, 35, 1216–1223.
Thompson, J. D., Higgins, D. G., & Gibson, T. J. (1994). Nucleic Acids Research, 22, 4673–4680.
Barnett, J. A., Payne, R. W., Yarrow, D. (1990) Yeasts: characteristics and identification. (pp. 683–684) Cambridge: Cambridge University Press.
Luo, C., Brink, D. L., & Blanch, H. W. (2002). Biomass Bioenergy, 22, 125–130.
Jönsson, L. J., Palmqvist, E., Nilvebrant, N. O., & Hahn-Hägerdal, B. (1998). Applied Microbiology and Biotechnology, 49, 691–697.
Rodrigues, R. C. L. B., Felipe, M. G. A., Almeida e Silva, J. B., Vitolo, M., & Gómez, P. V. (2001). Brazilian Journal of Chemical Engineering, 18(3), 299–311.
Silva, C. J. S. M., & Roberto, I. C. (2001). Letters in Applied Microbiology, 32, 248–252.
Nichols, N. N., Dien, B. S., & Guisado, G. M. (2005). Applied Biochemistry and Biotechnology, 124, 379–390.
Patrícia, A., Ramalho, M., & Helena, C. (2004). Applied and Environmental Microbiology, 70(4), 2279–2288.
Tamaki, H., Kumagai, H., & Shimada, Y. (1991). Agricultural and Biological Chemistry, 55(4), 951–956.
Arroyo, L. F. N., Duran, Q. M. C., & Garrido, F. A. (2006). Journal of Food Protection, 69(6), 1354–1364.
Lee, J. H., Lim, Y. B., & Park, K. M. (2003). Asian–Australasian Journal of Animal Sciences, 16(7), 1011–1014.
Middelhoven, W. J. (2001) Methods in biotechnology. In: Spencer, J. F. T., Spencer, R. (Eds.) Food microbiology protocols (pp. 209–224). New York: Humana.
Costas, M, Deive, F. J., & Longo, M. A. (2004). Process Biochemistry, 39, 2109–2114.
Martinez, A., Rodriguez, M. E., Wells, M. L., York, S. W., Preston, J. F., & Ingram, L. O. (2001). Biotechnology Progress, 17, 287–293.
Altschul, S. F., Madden, T. L., Schaffer, A. A., Zhang, J., Zhang, Z., Miller, W., et al. (1997). Nucleic Acids Research, 25(17), 3389–3402.
Acknowledgements
This research was supported by the National Nature Science Foundation of China (No.30450007)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hou-Rui, Z., Xiang-Xiang, Q., Silva, S.S. et al. Novel Isolates for Biological Detoxification of Lignocellulosic Hydrolysate. Appl Biochem Biotechnol 152, 199–212 (2009). https://doi.org/10.1007/s12010-008-8249-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8249-5