Abstract
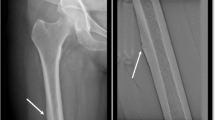
Atypical femoral shaft fractures are associated with the extended usage of nitrogen-containing bisphosphonates as therapy for osteoporosis. For such fractures, the positron emission tomography (PET) procedure, coupled with computerized tomography (CT), provides a potential imaging modality for defining aspects of the pathogenesis, site specificity, and possible prodromal abnormalities prior to fracture. PET-CT may assess the radiokinetic variables K1 (a putative marker for skeletal blood flow) and Ki (a putative marker for skeletal bone formation), and when combined with PET imaging modalities and CT skeletal site localization, may define the site of such radiokinetic findings. Further studies into the clinical usage of PET-CT in patients with atypical femoral shaft fractures are warranted.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
• Shane E, Burr D, Ebeling PR et al: Atypical subtrochanteric and diaphyseal femoral fractures: Report of a task force of the American Society for Bone and Mineral Research. J Bone Min Res 2011, 25: 2267–2294. This multi-authored and well-referenced report is the primary source in 2011 for information on the pathogenesis, clinical presentation, diagnostic procedures utilized, and potential therapeutic modalities of AFSF.
Abrahamsen B, Eiken P, Eastell R. Subtrochanteric and diaphyseal femur fractures in patients treated with alendronate: a register-based national cohort study. J Bone Min Res. 2009;24:1095–102.
Black DM, Kelly MP, Genant HK, et al. Bisphosphonates and fractures of the subtrochanteric or diaphyseal femur. N Engl J Med. 2010;362:1761–71.
Schilcher J, Michaelsson K, Aspenberg P. Bisphosphonate use and atypical fractures of the femoral shaft. N Engl J Med. 2011;364:1728–37.
Park-Wyllie LY, Mamdani MM, Juurlink DN, et al. Bisphosphonate use and the risk of subtrochanteric or femoral shaft fractures in older women. JAMA. 2011;305:783–9.
Hawkins RA, Choi Y, Huang S-C, et al. Evaluation of the skeletal kinetics of fluorine-18-fluoride ion with PET. J Nucl Med. 1992;33:633–42.
Messa C, Goodman WG, Hoh CK, et al. Bone metabolic activity measured with positron emission tomography and [18F] fluoride ion in renal osteodystrophy: correlation with bone histomorphometry. J Clin Endocrinol Metab. 1993;77:949–55.
Piert M, Zittel TT, Becker GA, et al. Assessment of porcine bone metabolism by dynamic [18F] ion PET: correlation with bone histomorphometry. J Nucl Med. 2001;42:1091–100.
Frost ML, Cook GJR, Blake GM, et al. The relationship between regional bone turnover measured using 18F-fluoride positron emission tomography and changes in BMD is equivalent to that seen for biochemical markers of bone turnover. J Clin Densitometry. 2007;10:46–54.
Frost ML, Fogelman I, Blake GM, et al. Association between global markers of bone formation and direct measurement of spinal bone formation in osteoporosis. J Bone Mineral Res. 2004;19:1797–804.
Frost ML, Blake GM, Park-Holohan S, et al. Long term precision of 18F-fluoride PET skeletal kinetic studies in the assessment of bone metabolism. J Nucl Med. 2008;49:700–7.
•• Doot RK, Muzi M, Peterson LM et al: Kinetic analysis of 18F-fluoride PET images of breast cancer bone metastases. J Nucl Med 2010, 51: 521–527. This paper, along with Frost et al. [17••], is the best and most up-to-date assessment of the radiokinetics of 18 F-fluoride PET for usage in metabolic bone disease.
Cook GJR, Lodge MA, Blake GM, et al. Differences in skeletal kinetics between vertebral and humeral bone measured by 18F-fluoride positron emission tomography in postmenopausal women. J Bone Mineral Res. 2000;15:763–9.
Frost ML, Blake GM, Cook GJR, et al. Differences in regional bone perfusion and turnover between lumbar spine and distal humerus: 18F-fluoride PET study of treatment-naïve and treated postmenopausal women. Bone. 2009;45:942–8.
Frost ML, Cook GJR, Blake GM, et al. Prospective study of risedronate on regional bone metabolism and blood flow at the lumbar spine measured by 18F-fluoride positron emission tomography. J Bone Mineral Res. 2003;18:2215–22.
Uchida K, Nakajima H, Miyazaki T, et al. Effects of alendronate on bone metabolism in glucocorticoid-induced osteoporosis measured by 18F-fluoride PET: a prospective study. J Nucl Med. 2009;50:1808–14.
•• Frost ML, Siddique M, Blake GM et al: Differential effects of teriparatide on regional bone formation using 18F-fluoride positron emission tomography. J Bone Mineral Res 2011, 26: 1002–1011. This paper, along with Doot et al. [12••], is the best and most up-to-date assessment of the radiokinetics of 18 F-fluoride PET for usage in metabolic bone disease.
Li J, Miller MA, Hutchins GD, et al. Imaging bone microdamage in vivo with positron emission tomography. Bone. 2005;35:819–24.
Ito M, Wakao N, Hida T, et al. Analysis of hip geometry by clinical CT for the assessment of hip fracture risk in elderly Japanese women. Bone. 2010;46:453–7.
Ito M, Nakamura T, Fukunaga M, et al. Effect of eldecalcitrol, an active vitamin D analog, on hip fracture and biomechanical properties: 3D assessment by clinical CT. Bone. 2011;49:328–34.
Taddei F, Martelli S, Reggiani B, et al. Finite-element modeling of bones from CT data: sensitivity to geometry and material uncertainties. IEEE Transactions on Biomechanical Engineering. 2006;53:2194–200.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chesnut, C.H., Chesnut, C.H. Can PET-CT Imaging and Radiokinetic Analyses Provide Useful Clinical Information on Atypical Femoral Shaft Fracture in Osteoporotic Patients?. Curr Osteoporos Rep 10, 42–47 (2012). https://doi.org/10.1007/s11914-011-0088-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11914-011-0088-6