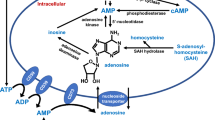
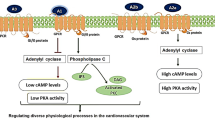
Abstract
Adenosine is an endogenous metabolite that has an anti-inflammatory effect across the vasculature. Extracellular adenosine activates 4 G-protein coupled receptors (A1, A3, A2A, and A2B) whose expression varies in different cells and tissues, including the vasculature and blood cells. Higher levels of adenosine are generated during stress, inflammation, and upon tissue damage. Some of the adenosine receptors (AR), such as the A2BAR, are further up-regulated following such stresses. This review discusses the role of adenosine and adenosine receptors in the development of atherosclerosis and some of the risk factors associated with this pathology. These include adenosine receptor-regulated changes in atherosclerosis, blood pressure, thrombosis, and myocardial infarction. Potential therapeutic applications are reviewed, as well as reasons for phenotypic differences occasionally observed between receptor knockout and pharmacological inhibition via drug administration.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Drury AN, Szent-Gyorgyi A. The physiological activity of adenine compounds with especial reference to their action upon the mammalian heart. J Physiol. 1929;68:213–37.
• Koupenova M, Johnston-Cox H, Vezeridis A, Gavras H, Yang D, Zannis V, et al. A2b adenosine receptor regulates hyperlipidemia and atherosclerosis. Circulation. 2012;125:354–63. The protective role of the anti-inflammatory A2B adenosine receptor in atherosclerosis is described. Activation of this receptor by a scpecific agonist reduces lesion formation and decreases plasma cholesterol and triglycerides levels while mice are still on high-fat, high-cholesterol diet..
Yang D, Koupenova M, McCrann DJ, Kopeikina KJ, Kagan HM, Schreiber BM, et al. The A2b adenosine receptor protects against vascular injury. Proc Natl Acad Sci U S A. 2008;105:792–6.
St Hilaire C, Koupenova M, Carroll SH, Smith BD, Ravid K. TNF-α upregulates the A2B adenosine receptor gene: The role of NAD(P)H oxidase 4. Biochem Biophys Res Commun. 2008;375:292–6.
Yang D, Zhang Y, Nguyen HG, Koupenova M, Chauhan AK, Makitalo M, et al. The A2B adenosine receptor protects against inflammation and excessive vascular adhesion. J Clin Invest. 2006;116:1913–23.
Kerbaul F, Benard F, Giorgi R, Youlet B, Carrega L, Zouher I, et al. Adenosine A2A receptor hyperexpression in patients with severe SIRS after cardiopulmonary bypass. J Investig Med. 2008;56:864–71.
Delyani JA, Van Wylen DG. Endocardial and epicardial interstitial purines and lactate during graded ischemia. Am J Physiol. 1994;266:H1019–26.
Reiss AB, Cronstein BN. Regulation of foam cells by adenosine. Arterioscler Thromb Vasc Biol. 2012;32:879–86.
Fredholm BB. Adenosine, an endogenous distress signal, modulates tissue damage and repair. Cell Death Differ. 2007;14:1315–23.
Fredholm BB, Arslan G, Halldner L, Kull B, Schulte G, Wasserman W. Structure and function of adenosine receptors and their genes. Naunyn Schmiedebergs Arch Pharmacol. 2000;362:364–74.
Moriyama K, Sitkovsky MV. Adenosine A2A receptor is involved in cell surface expression of A2B receptor. J Biol Chem. 2010;285:39271–88.
Bailey A, Ledent C, Kelly M, Hourani SM, Kitchen I. Changes in spinal delta and kappa opioid systems in mice deficient in the A2A receptor gene. J Neurosci. 2002;22:9210–20.
Li AC, Glass CK. The macrophage foam cell as a target for therapeutic intervention. Nat Med. 2002;8:1235–42.
Li AC, Glass CK. PPAR- and LXR-dependent pathways controlling lipid metabolism and the development of atherosclerosis. J Lipid Res. 2004;45:2161–73.
Aikawa M, Sugiyama S, Hill CC, Voglic SJ, Rabkin E, Fukumoto Y, et al. Lipid lowering reduces oxidative stress and endothelial cell activation in rabbit atheroma. Circulation. 2002;106:1390–6.
Stoll G, Bendszus M. Inflammation and atherosclerosis: novel insights into plaque formation and destabilization. Stroke. 2006;37:1923–32.
Boullier A, Bird DA, Chang MK, Dennis EA, Friedman P, Gillotre-Taylor K, et al. Scavenger receptors, oxidized LDL, and atherosclerosis. Ann N Y Acad Sci. 2001;947:214–22. discussion 22–3.
Gessi S, Fogli E, Sacchetto V, Merighi S, Varani K, Preti D, et al. Adenosine modulates HIF-1{α}, VEGF, IL-8, and foam cell formation in a human model of hypoxic foam cells. Arterioscler Thromb Vasc Biol. 2010;30:90–7.
Ohta A, Sitkovsky M. Role of G-protein-coupled adenosine receptors in downregulation of inflammation and protection from tissue damage. Nature. 2001;414:916–20.
Okusa MD, Linden J, Huang L, Rieger JM, Macdonald TL, Huynh LP. A(2A) adenosine receptor-mediated inhibition of renal injury and neutrophil adhesion. Am J Physiol Renal Physiol. 2000;279:F809–18.
Lee HT, Xu H, Nasr SH, Schnermann J, Emala CW. A1 adenosine receptor knockout mice exhibit increased renal injury following ischemia and reperfusion. Am J Physiol Renal Physiol. 2004;286:F298–306.
Hasko G, Kuhel DG, Chen JF, Schwarzschild MA, Deitch EA, Mabley JG, et al. Adenosine inhibits IL-12 and TNF-α production via adenosine A2a receptor-dependent and independent mechanisms. FASEB J. 2000;14:2065–74.
Bowlin TL, Borcherding DR, Edwards III CK, McWhinney CD. Adenosine A3 receptor agonists inhibit murine macrophage tumor necrosis factor-alpha production in vitro and in vivo. Cell Mol Biol (Noisy-le-grand). 1997;43:345–9.
Sajjadi FG, Takabayashi K, Foster AC, Domingo RC, Firestein GS. Inhibition of TNF-alpha expression by adenosine: role of A3 adenosine receptors. J Immunol. 1996;156:3435–42.
Salvatore CA, Tilley SL, Latour AM, Fletcher DS, Koller BH, Jacobson MA. Disruption of the A(3) adenosine receptor gene in mice and its effect on stimulated inflammatory cells. J Biol Chem. 2000;275:4429–34.
Reiss AB, Rahman MM, Chan ES, Montesinos MC, Awadallah NW, Cronstein BN. Adenosine A2A receptor occupancy stimulates expression of proteins involved in reverse cholesterol transport and inhibits foam cell formation in macrophages. J Leukoc Biol. 2004;76:727–34.
Chen JF, Huang Z, Ma J, Zhu J, Moratalla R, Standaert D, et al. A(2A) adenosine receptor deficiency attenuates brain injury induced by transient focal ischemia in mice. J Neurosci. 1999;19:9192–200.
• Bingham TC, Fisher EA, Parathath S, Reiss AB, Chan ES, Cronstein BN. A2A adenosine receptor stimulation decreases foam cell formation by enhancing ABCA1-dependent cholesterol efflux. J Leukoc Biol. 2010;87:683–90. This article further supports a potential effect of the A2A adenosine receptor on processes associated with atherosclerosis by affecting foam cell formationthrough regulating cholesterol efflux in macrophages.
St Hilaire C, Carroll SH, Chen H, Ravid K. Mechanisms of induction of adenosine receptor genes and its functional significance. J Cell Physiol. 2009;218:35–44.
Nakashima Y, Plump AS, Raines EW, Breslow JL, Ross R. ApoE-deficient mice develop lesions of all phases of atherosclerosis throughout the arterial tree. Arterioscler Thromb. 1994;14:133–40.
Piedrahita JA, Zhang SH, Hagaman JR, Oliver PM, Maeda N. Generation of mice carrying a mutant apolipoprotein E gene inactivated by gene targeting in embryonic stem cells. Proc Natl Acad Sci U S A. 1992;89:4471–5.
Plump AS, Smith JD, Hayek T, Aalto-Setala K, Walsh A, Verstuyft JG, et al. Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell. 1992;71:343–53.
Marathe S, Choi Y, Leventhal AR, Tabas I. Sphingomyelinase converts lipoproteins from apolipoprotein E knockout mice into potent inducers of macrophage foam cell formation. Arterioscler Thromb Vasc Biol. 2000;20:2607–13.
Jones MR, Zhao Z, Sullivan CP, Schreiber BM, Stone PJ, Toselli PA, et al. A(3) adenosine receptor deficiency does not influence atherogenesis. J Cell Biochem. 2004;92:1034–43.
• Wang H, Zhang W, Zhu C, Bucher C, Blazar BR, Zhang C, et al. Inactivation of the adenosine A2A receptor protects apolipoprotein E-deficient mice from atherosclerosis. Arterioscler Thromb Vasc Biol. 2009;29:1046–52. Elimination of the anti-inflammatory A2A adenosine receptor led to a suprising reduction in atherosclerosis, contrary to an expected phenotype based on an effect of this receptor on inflammation, and contrary to the protective effect of this receptor in a restensosis model.
• Wang H, Zhang W, Tang R, Zhu C, Bucher C, Blazar BR, et al. Adenosine receptor A2A deficiency in leukocytes increases arterial neointima formation in apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2010;30:915–22. This study was the first to highlight a role for A2A adenosine receptor in restenosis.
Chen H, Koupenova M, Yang D, Sume SS, Trackman PC, Ravid K. Regulation of MMP-9 expression by the A2b adenosine receptor and its dependency on TNF-alpha signaling. Exp Hematol. 2011;39:525–30.
Adair TH. Growth regulation of the vascular system: an emerging role for adenosine. Am J Physiol Regul Integr Comp Physiol. 2005;289:R283–96.
Ledent C, Vaugeois JM, Schiffmann SN, Pedrazzini T, El Yacoubi M, Vanderhaeghen JJ, et al. Aggressiveness, hypoalgesia, and high blood pressure in mice lacking the adenosine A2a receptor. Nature. 1997;388:674–8.
Varani K, Portaluppi F, Merighi S, Ongini E, Belardinelli L, Borea PA. Caffeine alters A2A adenosine receptors and their function in human platelets. Circulation. 1999;99:2499–502.
Yang D, Chen H, Koupenova M, Carroll SH, Eliades A, Freedman JE, et al. A new role for the A2b adenosine receptor in regulating platelet function. J Thromb Haemost. 2010;8:817–27.
Zhao Z, Makaritsis K, Francis CE, Gavras H, Ravid K. A role for the A3 adenosine receptor in determining tissue levels of cAMP and blood pressure: studies in knock-out mice. Biochim Biophys Acta. 2000;1500:280–90.
Eltzschig HK, Eckle T. Ischemia and reperfusion–from mechanism to translation. Nat Med. 2011;17:1391–401.
Ogawa S, Gerlach H, Esposito C, Pasagian-Macaulay A, Brett J, Stern D. Hypoxia modulates the barrier and coagulant function of cultured bovine endothelium. Increased monolayer permeability and induction of procoagulant properties. J Clin Invest. 1990;85:1090–8.
Ogawa S, Koga S, Kuwabara K, Brett J, Morrow B, Morris SA, et al. Hypoxia-induced increased permeability of endothelial monolayers occurs through lowering of cellular cAMP levels. Am J Physiol. 1992;262:C546–54.
Patel RA, Glover DK, Broisat A, Kabul HK, Ruiz M, Goodman NC, et al. Reduction in myocardial infarct size at 48 hours after brief intravenous infusion of ATL-146e, a highly selective adenosine A2A receptor agonist. Am J Physiol Heart Circ Physiol. 2009;297:H637–42.
Jordan JE, Zhao ZQ, Vinten-Johansen J. The role of neutrophils in myocardial ischemia-reperfusion injury. Cardiovasc Res. 1999;43:860–78.
Auchampach JA, Gross GJ. Adenosine A1 receptors, KATP channels, and ischemic preconditioning in dogs. Am J Physiol. 1993;264:H1327–36.
Ge ZD, Peart JN, Kreckler LM, Wan TC, Jacobson MA, Gross GJ, et al. Cl-IB-MECA [2-chloro-N6-(3-iodobenzyl)adenosine-5'-N-methylcarboxamide]reduces ischemia/reperfusion injury in mice by activating the A3 adenosine receptor. J Pharmacol Exp Ther. 2006;319:1200–10.
Eckle T, Krahn T, Grenz A, Kohler D, Mittelbronn M, Ledent C, et al. Cardioprotection by ecto-5'-nucleotidase (CD73) and A2B adenosine receptors. Circulation. 2007;115:1581–90.
Thim T, Hagensen MK, Bentzon JF, Falk E. From vulnerable plaque to atherothrombosis. J Intern Med. 2008;263:506–16.
Slager CJ, Wentzel JJ, Gijsen FJ, Thury A, van der Wal AC, Schaar JA, et al. The role of shear stress in the destabilization of vulnerable plaques and related therapeutic implications. Nat Clin Pract Cardiovasc Med. 2005;2:456–64.
Biaggioni I. Contrasting excitatory and inhibitory effects of adenosine in blood pressure regulation. Hypertension. 1992;20:457–65.
Ho WY, Lu PJ, Hsiao M, Hwang HR, Tseng YC, Yen MH, et al. Adenosine modulates cardiovascular functions through activation of extracellular signal-regulated kinases 1 and 2 and endothelial nitric oxide synthase in the nucleus tractus solitarii of rats. Circulation. 2008;117:773–80.
Shen FM, Su DF. The effect of adenosine on blood pressure variability in sinoaortic denervated rats is mediated by adenosine A2a-Receptor. J Cardiovasc Pharmacol. 2000;36:681–6.
Feng MG, Navar LG. Afferent arteriolar vasodilator effect of adenosine predominantly involves adenosine A2B receptor activation. Am J Physiol Renal Physiol. 2010;299:F310–5.
Li J, Fenton RA, Wheeler HB, Powell CC, Peyton BD, Cutler BS, et al. Adenosine A2a receptors increase arterial endothelial cell nitric oxide. J Surg Res. 1998;80:357–64.
Andersen H, Jaff MG, Hogh D, Vanhoutte P, Hansen PB. Adenosine elicits an eNOS-independent reduction in arterial blood pressure in conscious mice that involves adenosine A2A receptors. Acta Physiol (Oxf). 2011;203:197–207.
Xu Z, Park SS, Mueller RA, Bagnell RC, Patterson C, Boysen PG. Adenosine produces nitric oxide and prevents mitochondrial oxidant damage in rat cardiomyocytes. Cardiovasc Res. 2005;65:803–12.
Yang JN, Chen JF, Fredholm BB. Physiological roles of A1 and A2A adenosine receptors in regulating heart rate, body temperature, and locomotion as revealed using knockout mice and caffeine. Am J Physiol Heart Circ Physiol. 2009;296:H1141–9.
Giaime P, Carrega L, Fenouillet E, Mercier L, Gerolami V, Ruf J, et al. Relationship between A2A adenosine receptor expression and intradialytic hypotension during hemodialysis. J Investig Med. 2006;54:473–7.
Sun D, Samuelson LC, Yang T, Huang Y, Paliege A, Saunders T, et al. Mediation of tubuloglomerular feedback by adenosine: evidence from mice lacking adenosine 1 receptors. Proc Natl Acad Sci U S A. 2001;98:9983–8.
Belardinelli L, Lerman BB. Electrophysiological basis for the use of adenosine in the diagnosis and treatment of cardiac arrhythmias. Br Heart J. 1990;63:3–4.
Brown RD, Thoren P, Steege A, Mrowka R, Sallstrom J, Skott O, et al. Influence of the adenosine A1 receptor on blood pressure regulation and renin release. Am J Physiol Regul Integr Comp Physiol. 2006;290:R1324–9.
Johansson B, Halldner L, Dunwiddie TV, Masino SA, Poelchen W, Gimenez-Llort L, et al. Hyperalgesia, anxiety, and decreased hypoxic neuroprotection in mice lacking the adenosine A1 receptor. Proc Natl Acad Sci U S A. 2001;98:9407–12.
Gao X, Patzak A, Sendeski M, Scheffer PG, Teerlink T, Sallstrom J, et al. Adenosine A-receptor deficiency diminishes afferent arteriolar and blood pressure responses during nitric oxide inhibition and angiotensin II treatment. Am J Physiol Regul Integr Comp Physiol. 2011;301:R1669–81.
Feldman AM, Cheksis-Feiner E, Hamad E, Chan T. Adenosine receptor subtypes and the heart failure phenotype: translating lessons from mice to man. Trans Am Clin Climatol Assoc. 2011;122:198–214.
Tang Z, Diamond MA, Chen JM, Holly TA, Bonow RO, Dasgupta A, et al. Polymorphisms in adenosine receptor genes are associated with infarct size in patients with ischemic cardiomyopathy. Clin Pharmacol Ther. 2007;82:435–40.
Lieu HD, Shryock JC, von Mering GO, Gordi T, Blackburn B, Olmsted AW, et al. Regadenoson, a selective A2A adenosine receptor agonist, causes dose-dependent increases in coronary blood flow velocity in humans. J Nucl Cardiol. 2007;14:514–20.
Bothe GW, Bolivar VJ, Vedder MJ, Geistfeld JG. Genetic and behavioral differences among 5 inbred mouse strains commonly used in the production of transgenic and knockout mice. Genes Brain Behav. 2004;3:149–57.
Mekada K, Abe K, Murakami A, Nakamura S, Nakata H, Moriwaki K, et al. Genetic differences among C57BL/6 substrains. Exp Anim. 2009;58:141–9.
Huang TT, Naeemuddin M, Elchuri S, Yamaguchi M, Kozy HM, Carlson EJ, et al. Genetic modifiers of the phenotype of mice deficient in mitochondrial superoxide dismutase. Hum Mol Genet. 2006;15:1187–94.
Bryant CD, Zhang NN, Sokoloff G, Fanselow MS, Ennes HS, Palmer AA, et al. Behavioral differences among C57BL/6 substrains: implications for transgenic and knockout studies. J Neurogenet. 2008;22:315–31.
Liu L, Nutter LM, Law N, McKerlie C. Sperm freezing and in vitro fertilization in 3 substrains of C57BL/6 mice. J Am Assoc Lab Anim Sci. 2009;48:39–43.
Moayeri M, Crown D, Newman ZL, Okugawa S, Eckhaus M, Cataisson C, et al. Inflammasome sensor Nlrp1b-dependent resistance to anthrax is mediated by caspase-1, IL-1 signaling and neutrophil recruitment. PLoS Pathog. 2010;6:e1001222.
Acknowledgments
The authors apologize to all whose work they could not cite due to limited space required by the journal. This work was supported by National Heart, Lung, and Blood Institute grant HL93149, and by the Boston Nutrition Obesity Research Center (DK046200) Pilot Grant to Katya Ravid; Milka Koupenova was supported by postdoctoral Training Grant from NIH (HL007224); Hillary A. Johnston-Cox was supported by pre-doctoral Training Grant from NIH (HL007969).
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koupenova, M., Johnston-Cox, H. & Ravid, K. Regulation of Atherosclerosis and Associated Risk Factors by Adenosine and Adenosine Receptors. Curr Atheroscler Rep 14, 460–468 (2012). https://doi.org/10.1007/s11883-012-0263-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11883-012-0263-y