Abstract
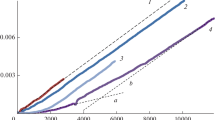
A kinetic study of the prooxidant effect of α-tocopherol was performed. The rates of allylic hydrogen abstraction from various unsaturated fatty acid esters (ethyl stearate 1, ethyl oleate 2, ethyl linoleate 3, ethyl linolenate 4, and ethyl arachidonate 5) by α-tocopheroxyl radical in toluene were determined, using a double-mixing stopped-flow spectrophotometer. The second-order rate constants (k p) obtained are <1 × 10−2 M−1 s−1 for 1, 1.90 × 10−2 M−1 s−1 for 2, 8.33 × 10−2 M−1 s−1 for 3, 1.92 × 10−1 M−1 s−1 for 4, and 2.43 × 10−1 M−1 s−1 for 5 at 25.0 °C. Fatty acid esters 3, 4, and 5 contain two, four, and six –CH2– hydrogen atoms activated by two π-electron systems (–C=C–CH2–C=C–). On the other hand, fatty acid ester 2 has four –CH2– hydrogen atoms activated by a single π-electron system (–CH2–C=C–CH2–). Thus, the rate constants, k abstr/H, given on an available hydrogen basis are k p/4 = 4.75 × 10−3 M−1 s−1 for 2, k p/2 = 4.16 × 10−2 M−1 s−1 for 3, k p/4 = 4.79 × 10−2 M−1 s−1 for 4, and k p/6 = 4.05 × 10−2 M−1 s−1 for 5. The k abstr/H values obtained for 3, 4, and 5 are similar to each other, and are by about one order of magnitude higher than that for 2. From these results, it is suggested that the prooxidant effect of α-tocopherol in edible oils, fats, and low-density lipoproteins may be induced by the above hydrogen abstraction reaction.





Similar content being viewed by others
Abbreviations
- ArOH:
-
2,6-Di-tert-butyl-4(4′-methoxyphenyl)phenol
- ArO·:
-
2,6-Di-tert-butyl-4(4′-methoxyphenyl)phenoxyl (aroxyl)
- AsH− :
-
Ascorbate mono anion
- As·− :
-
Ascorbate mono anion radical
- 5,7-Di-iPr-Toc·:
-
5,7-Di-isopropyl-tocopheroxyl
- ESR:
-
Electron spin resonance
- LDL:
-
Low-density lipoprotein
- LH:
-
Lipid (or fatty acid ethyl ester)
- LOOH:
-
Lipid hydroperoxide (or methyl linoleate hydroperoxide)
- LOO·:
-
Lipid peroxyl radical
- LO·:
-
Lipid alkoxyl radical
- NRP:
-
Non-radical products
- α-TocH:
-
α-Tocopherol
- α-Toc·:
-
α-Tocopheroxyl
References
Niki E (1987) Antioxidants in relation to lipid peroxidation. Chem Phys Lipids 44:227–253
Barclay LRC (1993) Model biomembranes: quantitative studies of peroxidation, antioxidant action, partitioning, and oxidative stress. Can J Chem 71:1–16
Jiang Q, Christen S, Shigenaga MK, Ames BN (2001) γ-Tocopherol, the major form of vitamin E in the US diet, deserves more attention. Am J Clin Nutr 74:714–722
Traber MG, Atkinson J (2007) Vitamin E, antioxidant and nothing more. Free Radic Biol Med 43:4–15
Mukai K, Tsuzuki N, Ouchi S, Fukuzawa K (1982) Electron spin resonance studies of chromanoxyl radicals derived from tocopherols. Chem Phys Lipids 30:337–345
Burton GW, Doba T, Gabe EJ, Hughes L, Lee FL, Prasad L, Ingold KU (1985) Autoxidation of biological molecules. 4. Maximizing the antioxidant activity of phenols. J Am Chem Soc 107:7053–7065
Cillard J, Cillard P, Cormier M, Girre L (1980) α-Tocopherol prooxidant effect in aqueous media: increased autoxidation rate of linoleic acid. J Am Oil Chem Soc 57:252–255
Terao J, Matsushita S (1986) The peroxidizing effect of α-tocopherol on autoxidation of methyl linoleate in bulk phase. Lipids 21:255–260
Bowry VW, Stocker R (1993) Tocopherol-mediated peroxidation. The prooxidant effect of vitamin E on the radical-initiated oxidation of human low-density lipoprotein. J Am Chem Soc 115:6029–6044
Mukai K, Noborio S, Nagaoka S (2005) Why is the order reversed? Peroxyl-scavenging activity and fats-and-oils protecting activity of vitamin E. Int J Chem Kinet 37:605–610 and references are cited therein
Kagan VE, Quinn PJ (eds) (2001) Coenzyme Q: molecular mechanisms in health and disease. CRC Press, USA
Mukai K, Sawada K, Kohno Y, Terao J (1993) Kinetic study of the prooxidant effect of tocopherol. Hydrogen abstraction from lipid hydroperoxides by tocopheroxyls in solution. Lipids 28:747–752
Nagaoka S, Sawada K, Fukumoto Y, Nagashima U, Katsumata S, Mukai K (1992) Mechanism of prooxidant reaction of vitamin E. Kinetic, spectroscopic, and ab initio study of proton-transfer reaction. J Phys Chem 96:6663–6668
Packer JE, Slater TF, Willson RL (1979) Direct observation of a free radical interaction between vitamin E and vitamin C. Nature 278:737–738
Mukai K, Itoh S, Morimoto H (1992) Stopped-flow kinetic study of vitamin E regeneration reaction with biological hydroquinones (reduced forms of ubiquinone, vitamin K, and tocopherolquinone) in solution. J Biol Chem 267:22277–22281
Bisby RH, Parker AW (1995) Reaction of ascorbate with the α-tocopheroxyl radical in micellar and bilayer membrane systems. Arch Biochem Biophys 317:170–178
Nagaoka S, Kakiuchi T, Ohara K, Mukai K (2007) Kinetics of the reaction by which natural vitamin E is regenerated by vitamin C. Chem Phys Lipids 146:26–32
Lucarini M, Pedulli GF, Cipollone M (1994) Bond dissociation enthalpy of α-tocopherol and other phenolic antioxidants. J Org Chem 59:5063–5070
Gregor W, Grabner G, Adelwhrer C, Rosenau T, Gille L (2005) Antioxidant properties of natural and synthetic chromanol derivatives: study by fast kinetics and electron spin resonance spectroscopy. J Org Chem 70:3472–3483
Mukai K, Ouchi A, Mitarai A, Ohara K, Matsuoka C (2009) Formation and decay dynamics of vitamin E radical in the antioxidant reaction of vitamin E. Bull Chem Soc Jpn 82:494–503 and references are cited therein
Remorova AA, Roginsky VA (1991) Rate constants for the reaction of α-tocopherol phenoxy radicals with unsaturated fatty acid esters, and the contribution of this reaction to the kinetics of inhibition of lipid oxidation. Kinet Catal 32:726–731
Watanabe A, Noguchi N, Fujisawa A, Kodama T, Tamura K, Cynshi O, Niki E (2000) Stability and reactivity of aroxyl radicals derived from a novel antioxidant BO-653 and related compounds. Effects of substituent and side chain in solution and membranes. J Am Chem Soc 122:5438–5442
Nagaoka S, Okauchi Y, Urano S, Nagashima U, Mukai K (1990) Kinetic and ab initio study of the prooxidant effect of vitamin E. Hydrogen abstraction from fatty acid esters and egg yolk lecithin. J Am Chem Soc 112:8921–8924
Mukai K, Morimoto H, Okauchi Y, Nagaoka S (1993) Kinetic study of reactions between tocopheroxyl radicals and fatty acids. Lipids 28:753–756
Mukai K, Watanabe Y, Uemoto Y, Ishizu K (1986) Stopped-flow investigation of antioxidant activity of tocopherol. Bull Chem Soc Jpn 59:3113–3116
Rieker A, Scheffler K (1965) Die Beteiligung von Phenylresten an der Aroxylmesomerie. Liebigs Ann Chem 689:78–92
Burton GW, Ingold KU (1986) Vitamin E: applications of the principles of physical organic chemistry to the exploration of its structure and function. Acc Chem Res 19:194–201
Esterbauer H, Ramos P (1995) Chemistry and pathophysiology of oxidation of LDL. Rev Physiol Biochem Pharmacol 127:31–64
Bowry VW, Ingold KU, Stocker R (1992) Vitamin E in human low-density lipoprotein. Biochem J 288:341–344
Vinson JA, Jang J, Yang J, Dabbagh Y, Liang X, Serry M, Proch J, Cai S (1999) Vitamins and especially flavonoids in common beverages are powerful in vitro antioxidants which enrich lower density lipoproteins and increase their oxidative resistance after ex vivo spiking in human plasma. J Agric Food Chem 47:2502–2504
Liu Z-Q, Ma L-P, Zhou B, Yang L, Liu Z-L (2000) Antioxidative effects of green tea polyphenols on free radical initiated and photosensitized peroxidation of human low density lipoprotein. Chem Phys Lipids 106:53–63
Laranjinha J, Cadenas E (1999) Redox cycles of caffeic acid, α-tocopherol, and ascorbate: implications for protection of low-density lipoproteins against oxidation. IUBMB Life 48:57–65
Ohara K, Ichimura Y, Tsukamoto K, Ogata M, Nagaoka S, Mukai K (2006) Kinetic study on the free radical-scavenging and vitamin E-regenerating actions of caffeic acid and its related compounds. Bull Chem Soc Jpn 79:1501–1508
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ouchi, A., Ishikura, M., Konishi, K. et al. Kinetic Study of the Prooxidant Effect of α-Tocopherol. Hydrogen Abstraction from Lipids by α-Tocopheroxyl Radical. Lipids 44, 935–943 (2009). https://doi.org/10.1007/s11745-009-3339-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-009-3339-x