ABSTRACT
OBJECTIVE
To perform a systematic review and meta-analysis of clinical trials evaluating the efficacy and safety of midodrine in orthostatic hypotension (OH).
METHODS
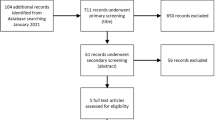
We searched major databases and related conference proceedings through June 30, 2012. Two reviewers independently selected studies and extracted data. Random-effects meta-analysis was used to pool the outcome measures across studies.
RESULTS
Seven trials were included in the efficacy analysis (enrolling 325 patients, mean age 53 years) and two additional trials were included in the safety analysis. Compared to placebo, the mean change in systolic blood pressure was 4.9 mmHg (p = 0.65) and the mean change in mean arterial pressure from supine to standing was −1.7 mmHg (p = 0.45). The change in standing systolic blood pressure before and after giving midodrine was 21.5 mmHg (p < 0.001). A significant improvement was seen in patients’ and investigators’ global assessment symptoms scale (a mean difference of 0.70 [95 % CI 0.30–1.09; p < 0.001] and 0.80 [95 % CI 0.76–0.85; p < 0.001], respectively). There was a significant increase in risk of piloerection, scalp pruritis, urinary hesitancy/retention, supine hypertension and scalp paresthesia after giving midodrine. The quality of evidence was limited by imprecision, heterogeneity and increased risk of bias.
CONCLUSION
There is insufficient and low quality evidence to support the use of midodrine for OH.




Similar content being viewed by others
REFERENCES
Freeman R, Wieling W, Axelrod FB, et al. Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Auton Neurosci. 2011;161:46–8.
Low PA. Update on the evaluation, pathogenesis, and management of neurogenic orthostatic hypotension: introduction. Neurology. 1995;45:S4–5.
Low PA, Singer W. Management of neurogenic orthostatic hypotension: an update. Lancet Neurol. 2008;7:451–8.
Somberg JC. The midodrine withdrawal. Am J Ther. 2010;17:445.
Dhruva SS, Redberg RF. Accelerated approval and possible withdrawal of midodrine. JAMA. 2010;304:2172–3.
Mitka M. FDA takes slow road toward withdrawal of drug approved with fast-track process. JAMA. 2011;305:982–4.
Mitka M. Trials to address efficacy of midodrine 18 years after it gains FDA approval. JAMA. 2012;307:1124–7.
Somberg JC. The midodrine withdrawal. Am J Ther. 2010;17:445.
Cohen J. A coefficient of agreement for nominal scales. Educ Psychol Meas. 1960;20:37–46.
Higgins JP, Green S, Eds. Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011 Available from www.cochrane-handbook.org 2011.
Atkins D, Best D, Briss PA, et al. Grading quality of evidence and strength of recommendations. BMJ. 2004;328:1490.
Guyatt GH, Oxman AD, Kunz R, Vist GE, Falck-Ytter Y, Schunemann HJ. What is “quality of evidence” and why is it important to clinicians? BMJ. 2008;336:995–8.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60.
Fouad-Tarazi FM, Okabe M, Goren H. Alpha sympathomimetic treatment of autonomic insufficiency with orthostatic hypotension. Am J Med. 1995;99:604–10.
Jankovic J, Gilden JL, Hiner BC, et al. Neurogenic orthostatic hypotension: a double-blind, placebo-controlled study with midodrine. Am J Med. 1993;95:38–48.
Low PA, Gilden JL, Freeman R, Sheng KN, McElligott MA. Efficacy of midodrine vs. placebo in neurogenic orthostatic hypotension. A randomized, double-blind multicenter study. Midodrine Study Group. JAMA. 1997;277:1046–51.
Schrage WG, Eisenach JH, Dinenno FA, et al. Effects of midodrine on exercise-induced hypotension and blood pressure recovery in autonomic failure. J Appl Physiol. 2004;97:1978–84.
Wecht JM, Rosado-Rivera D, Handrakis JP, Radulovic M, Bauman WA. Effects of midodrine hydrochloride on blood pressure and cerebral blood flow during orthostasis in persons with chronic tetraplegia. Arch Phys Med Rehabil. 2010;91:1429–35.
Wright RA, Kaufmann HC, Perera R, et al. A double-blind, dose-response study of midodrine in neurogenic orthostatic hypotension. Neurology. 1998;51:120–4.
Axelrod FB, Krey L, Glickstein JS, Weider Allison J, Friedman D. Preliminary observations on the use of midodrine in treating orthostatic hypotension in familial dysautonomia. J Auton Nerv Syst. 1995;55:29–35.
Hoeldtke RD, Horvath GG, Bryner KD, Hobbs GR. Treatment of orthostatic hypotension with midodrine and octreotide. J Clin Endocrinol Metab. 1998;83:339–43.
Kaufmann H, Brannan T, Krakoff L, Yahr MD, Mandeli J. Treatment of orthostatic hypotension due to autonomic failure with a peripheral alpha-adrenergic agonist (midodrine). Neurology. 1988;38:951–6.
Logan IC, Witham MD. Efficacy of treatments for orthostatic hypotension: a systematic review. Age Ageing. 2012;41:587–94.
Pathak A, Raoul V, Montastruc JL, Senard JM. Adverse drug reactions related to drugs used in orthostatic hypotension: a prospective and systematic pharmacovigilance study in France. Eur J Clin Pharmacol. 2005;61:471–4.
Sterne JA, Gavaghan D, Egger M. Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. J Clin Epidemiol. 2000;53:1119–29.
Acknowledgments
We would like to thank Dr. Christian Jeng-Singh (Mayo Clinic, Rochester, MN) for his help in the translation of the German articles.
Conflict of Interest
The authors declare that they do not have a conflict of interest.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ajay K. Parsaik and Balwinder Singh contributed equally to this manuscript.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Online Appendix
(PDF 71 kb)
Rights and permissions
About this article
Cite this article
Parsaik, A.K., Singh, B., Altayar, O. et al. Midodrine for Orthostatic Hypotension: A Systematic Review and Meta-Analysis of Clinical Trials. J GEN INTERN MED 28, 1496–1503 (2013). https://doi.org/10.1007/s11606-013-2520-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11606-013-2520-3