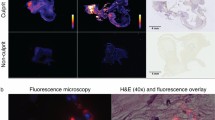
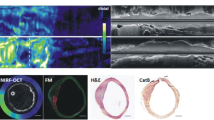
Abstract
Purpose
Unstable plaques may cause clinical events. Plaque destabilization results from the synergy between intraplaque angiogenesis and inflammation. Vascular endothelial growth factor (VEGF) and VEGF receptors (VEGFRs) are considered to be involved in these processes. We investigated the efficacy of the anti-VEGFR mimic single-chain VEGF (scVEGF) to map intra-plaque VEGFR expression and atherosclerotic plaque instability using near-infrared fluorescence (NIRF).
Procedures
Human carotid plaques were retrieved from 15 symptomatic and five asymptomatic patients. NIRF plaque imaging was performed pre-/post-incubation with scVEGF/Cy5.5. Biopsies taken from regions with high (hot spot) and low (cold spot) NIRF signals were examined for VEGF-A, VEGFR-1 and VEGFR-2 mRNA expression levels using real-time RT-PCR analysis. Immunohistochemistry for CD31 (endothelium), CD68 (macrophages) and αSMA (smooth muscle cells) was performed to evaluate plaque composition.
Results
NIRF imaging of 20 plaques revealed a heterogeneous distribution of scVEGF/Cy5.5 binding. After incubation NIRF activity increased from 3.9×10−5 ± 5.2×10−6 to 3.0×10−4 ± 2.2×10−5 and 5.8×10−5 ± 1.9×10−5 to 3.1×10−4 ± 1.9×10−5 photons/s/cm2/sr/illumination intensity on the intraluminal and extraluminal side, respectively (both p < 0.001). Real-time RT-PCR analysis showed a ~1.2- and ~16.4-fold increased mRNA expression of VEGFR-1 and VEGFR-2, respectively, in hot spots (vs. cold spots). Immunohistochemistry exhibited higher intraplaque capillary density in hot spots (vs. cold spots) (17.2 ± 3.7 vs. 5.4 ± 2.2 capillary/mm2; p = 0.037). Hot spots contained significantly reduced numbers of α-SMA-positive cells (vs. cold spots) (2.2 ± 0.7 % vs. 6.9 ± 1.5 %; p = 0.038). Finally, a ~2-fold increase of CD68+ infiltrating macrophages within hot spots (vs. cold spots) was observed (not significant, p = 0.17). Significant higher capillary density in hot spots (vs. cold spots) was observed in plaques from symptomatic patients but not in plaques from asymptomatic patients.
Conclusion
Our data support that scVEGF/Cy5.5 is a suitable indicator for plaque instability and a promising diagnostic tool for risk assessment in cardiovascular diseases.






Similar content being viewed by others
References
Mauriello A, Sangiorgi GM, Virmani R et al (2010) A pathobiologic link between risk factors profile and morphological markers of carotid instability. Atherosclerosis 208:572–580
Rothwell PM, Gibson R, Warlow CP (2000) Interrelation between plaque surface morphology and degree of stenosis on carotid angiograms and the risk of ischemic stroke in patients with symptomatic carotid stenosis. On behalf of the European Carotid Surgery Trialists' Collaborative Group. Stroke 31:615–621
Bjornheden T, Levin M, Evaldsson M, Wiklund O (1999) Evidence of hypoxic areas within the arterial wall in vivo. Arterioscler Thromb Vasc Biol 19:870–876
Ahmad S, Hewett PW, Wang P et al (2006) Direct evidence for endothelial vascular endothelial growth factor receptor-1 function in nitric oxide-mediated angiogenesis. Circ Res 99:715–722
Carmeliet P, Moons L, Luttun A et al (2001) Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 7:575–583
de Vries C, Escobedo JA, Ueno H et al (1992) The fms-like tyrosine kinase, a receptor for vascular endothelial growth factor. Science 255:989–991
Terman BI, Dougher-Vermazen M, Carrion ME et al (1992) Identification of the KDR tyrosine kinase as a receptor for vascular endothelial cell growth factor. Biochem Biophys Res Commun 187:1579–1586
Chang SH, Feng D, Nagy JA et al (2009) Vascular permeability and pathological angiogenesis in caveolin-1-null mice. Am J Pathol 175:1768–1776
O'Brien KD, Allen MD, McDonald TO et al (1993) Vascular cell adhesion molecule-1 is expressed in human coronary atherosclerotic plaques. Implications for the mode of progression of advanced coronary atherosclerosis. J Clin Invest 92:945–951
O'Brien KD, McDonald TO, Chait A et al (1996) Neovascular expression of E-selectin, intercellular adhesion molecule-1, and vascular cell adhesion molecule-1 in human atherosclerosis and their relation to intimal leukocyte content. Circulation 93:672–682
Post S, Peeters W, Busser E et al (2008) Balance between angiopoietin-1 and angiopoietin-2 is in favor of angiopoietin-2 in atherosclerotic plaques with high microvessel density. J Vasc Res 45:244–250
Bhardwaj S, Roy H, Heikura T, Yla-Herttuala S (2005) VEGF-A, VEGF-D and VEGF-D(DeltaNDeltaC) induced intimal hyperplasia in carotid arteries. Eur J Clin Invest 35:669–676
Celletti FL, Waugh JM, Amabile PG et al (2001) Vascular endothelial growth factor enhances atherosclerotic plaque progression. Nat Med 7:425–429
Holm PW, Slart RH, Zeebregts CJ et al (2009) Atherosclerotic plaque development and instability: a dual role for VEGF. Ann Med 41:257–264
Wallis de Vries BM, Hillebrands JL, van Dam GM et al (2009) Images in cardiovascular medicine. Multispectral near-infrared fluorescence molecular imaging of matrix metalloproteinases in a human carotid plaque using a matrix-degrading metalloproteinase-sensitive activatable fluorescent probe. Circulation 119:e534–e536
Hermus L, van Dam GM, Zeebregts CJ (2010) Advanced carotid plaque imaging. Eur J Vasc Endovasc Surg 39:125–133
Backer MV, Levashova Z, Patel V et al (2007) Molecular imaging of VEGF receptors in angiogenic vasculature with single-chain VEGF-based probes. Nat Med 13:504–509
Ahmed A, Dunk C, Kniss D, Wilkes M (1997) Role of VEGF receptor-1 (Flt-1) in mediating calcium-dependent nitric oxide release and limiting DNA synthesis in human trophoblast cells. Lab Invest 76:779–791
Bussolati B, Dunk C, Grohman M et al (2001) Vascular endothelial growth factor receptor-1 modulates vascular endothelial growth factor-mediated angiogenesis via nitric oxide. Am J Pathol 159:993–1008
Brown LF, Detmar M, Claffey K et al (1997) Vascular permeability factor/vascular endothelial growth factor: a multifunctional angiogenic cytokine. EXS 79:233–269
Feng D, Nagy JA, Brekken RA et al (2000) Ultrastructural localization of the vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) receptor-2 (FLK-1, KDR) in normal mouse kidney and in the hyperpermeable vessels induced by VPF/VEGF-expressing tumors and adenoviral vectors. J Histochem Cytochem 48:545–556
Feng D, Nagy JA, Hipp J et al (1997) Reinterpretation of endothelial cell gaps induced by vasoactive mediators in guinea-pig, mouse and rat: many are transcellular pores. J Physiol 504(Pt 3):747–761
Kolodgie FD, Gold HK, Burke AP et al (2003) Intraplaque hemorrhage and progression of coronary atheroma. N Engl J Med 349:2316–2325
Moreno PR, Purushothaman KR, Zias E et al (2006) Neovascularization in human atherosclerosis. Curr Mol Med 6:457–477
Shaalan WE, Cheng H, Gewertz B et al (2004) Degree of carotid plaque calcification in relation to symptomatic outcome and plaque inflammation. J Vasc Surg 40:262–269
Moura R, Tjwa M, Vandervoort P et al (2008) Thrombospondin-1 deficiency accelerates atherosclerotic plaque maturation in ApoE−/− mice. Circ Res 103:1181–1189
Conflict of Interest
None declared
Author information
Authors and Affiliations
Corresponding author
Additional information
M.K. Lam and S. Al-Ansari contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lam, M.K., Al-Ansari, S., van Dam, G.M. et al. Single-Chain VEGF/Cy5.5 Targeting VEGF Receptors to Indicate Atherosclerotic Plaque Instability. Mol Imaging Biol 15, 250–261 (2013). https://doi.org/10.1007/s11307-012-0594-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-012-0594-7