Abstract
Introduction
Glucose transporters and hexokinases determine the kinetics of 2-deoxy-2-[18F]fluoro-d-glucose (FDG). However, the genes controlling these proteins are not independent and may be modulated from other biological processes, e.g., like angiogenesis and proliferation. The impact of cell-proliferation-related genes on the FDG kinetics was assessed in colorectal tumors in this study.
Methods
Patients with primary colorectal tumors (n = 25) were examined with positron emission tomography and FDG within 2 days prior to surgery. Tissue specimens were obtained from the colorectal tumor and the normal colon by surgery and gene expression was assessed using gene arrays.
Results
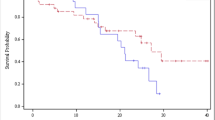
Overall, an increase of the expression of proliferation associated genes was observed by a factor of 2–5.3 for the colorectal tumors as compared with the normal colon. Correlation analysis revealed an impact of cdk2 on K1, thus directing to a modulation of the FDG uptake into the cells. The correlations were generally higher for the FDG influx as compared with the standardized uptake value (SUV). The influx was mainly correlated with proliferation inhibiting genes (cyclin G2, cdk inhibitor 1 C, cdk inhibitor 2B). It was possible to predict the expression of cyclin D2 using a multiple linear regression function and the parameters of the FDG kinetics with r = 0.67. Using a group based analysis it was possible to demonstrate, that tumors with an SUV >12 are associated with a high expression of cyclin D2 in the colorectal tumors. If the gene expression data for cyclin D1, cyclin G2, cdk2, cdk6 and cdk inhibtor 2B were used, the overall FDG uptake as measured by the SUV could be predicted with r = 0.75.
Conclusions
The results suggest that the FDG kinetics is modulated by proliferation associated genes. Especially K1, the parameter for the FDG transport into the cells, is modulated by cdk2. Tumors with a SUV exceeding 12 have usually a higher expression of cyclin D2. The parameters of the FDG kinetics can be used to predict the expression of proliferation associated genes individually.











Similar content being viewed by others
References
Strauss LG, Conti PS (1991) The applications of PET in clinical oncology. J Nucl Med 32:623–648
Higashi K, Clavo AC, Wahl RL (1993) Does FDG uptake measure proliferative activity of human cancer cells? In vitro comparison with DNA flow cytometry and tritiated thymidine uptake. J Nucl Med 34:414–419
Tateishi U, Yamaguchi U, Seki K, Terauchi T, Arai Y, Hasegawa T (2006) Glut-1 expression and enhanced metabolism are associated with tumour grade in bone and soft tissue sarcomas: a prospective evaluation by [18F]fluorodeoxyglucose positron emission tomography. Eur J Nucl Med Mol Imaging 33:683–691
Vesselle H, Schmidt RA, Pugsley JM et al (2000) Lung cancer proliferation correlates with [F-18]fluorodeoxyglucose uptake by positron emission tomography. Clin Cancer Res 6:3837–3844
Pugachev A, Ruan S, Carlin S et al (2005) Dependence of FDG uptake on tumour microenvironment. Int J Radiat Oncol Biol Phys 62:545–553
Strauss LG, Klippel S, Pan L, Schönleben K, Haberkorn U, Dimitrakopoulou-Strauss A (2007) Assessment of quantitative FDG PET data in primary colorectal tumours: which parameters are important with respect to tumour detection? Eur J Nucl Med Mol Imaging 34:868–877
Burt BM, Humm JL, Kooby DA et al (2001) Using positron emission tomography with [18 F]FDG to predict tumour behavior in experimental colorectal cancer. Neopasia 3:189–195
Dimitrakopoulou-Strauss A, Strauss LG, Schwarzbach M et al (2001) Dynamic PET 18F-FDG studies in patients with primary and recurrent soft-tissue sarcomas: impact on diagnosis and correlation with grading. J Nucl Med 42:713–720
Strauss LG, Dimitrakopoulou-Strauss A, Koczan D et al (2004) 18F-FDG kinetics and gene expression in giant cell tumours. J Nucl Med 45:1528–1535
Strauss LG, Koczan D, Klippel S et al (2008) Impact of angiogenesis-related gene expression on the tracer kinetics of 18F-FDG kinetics in colorectal tumours. J Nucl Med 49:1238–1244
Ohtake T, Kosaka N, Watanabe T, Yokoyama I, Moritan T, Masuo M et al (1991) Noninvasive method to obtain input function for measuring glucose utilization of thoracic and abdominal organs. J Nucl Med 32:1432–1438
Pan L, Mikolajczyk K, Strauss LG, Haberkorn U, Dimitrakopoulou-Strauss A (2007) Machine learning based parameter imaging and kinetic modeling of PET data [abstract]. J Nucl Med 48(suppl):158P
Dimitrakopoulou-Strauss A, Strauss LG, Mikolajczyk K, Burger C, Lehnert T, Bernd LG, Ewerbeck V (2003) On the fractal nature of dynamic positron emission tomography (PET) studies world. J Nucl Med 2:306–313
Strauss LG, Pan L, Koczan D et al (2007) Fusion of positron emission tomography (PET) and gene array data: a new approach for the correlative analysis of molecular biological and clinical data. IEEE Trans Med Imag 26:804–812
Wunder JS, Paulian G, Huyos AG, Heller G, Meyers PA, Healey JH (1998) The histological response to chemotherapy as a predictor of the oncological outcome of operative treatment of Ewing sarcoma. J Bone Joint Surg Am 80:1020–1033
Zhang J, Kang SK, Wang L, Touijer A, Hricak H (2009) Distribution of renal tumour growth rates determined by using serial volumetric CT measurements. Radiology 250:137–144
Goh V, Halligan S, Daley F, Wellsted DM, Guenther T, Bartram CT (2008) Colorectal tumour vascularity: quantitative assessment with multidetector CT—do tumour perfusion measurements reflect angiogenesis? Radiology 249:910–917
Schiepers C, Chen W, Dahlbom M, Cloughesy T, Hoh CK, Huang SC (2007) 18F-fluorothymidine kinetics of malignant brain tumours. Eur J Nucl Med Mol Imaging 34:1003–1111
Dimitrakopoulou-Strauss A, Strauss LG (2008) The role of 18 F-FLT in cancer imaging: does it really reflect proliferation? Eur J Nucl Med Mol Imaging 35:523–526
Riedl CC, Akhurst T, Larson S et al (2007) 18F-FDG PET scanning correlates with tissue markers of poor prognosis and predicts mortality for patients after liver resection for colorectal metastases. J Nucl Med 48:771–775
Buck AK, Halter G, Schirrmeister H et al (2003) Imaging proliferation in lung tumours with PET: 18F-FLT versus 18F-FDG. J Nucl Med 44:1426–1431
Henriksson E, Kjellén E, Baldetorp B et al (2009) Comparison of cisplatin sensitivity and the 18F fluoro-2-deoxy 2 glucose uptake with proliferation parameters and gene expression in squamous cell carcinoma cell lines of the head and neck. J Exp Clin Cancer Res 28:17. doi:10.1186/1756-9966-28-17
Sánchez Salmóna A, Garridoa M, Abdulkaderb I, Gudec F, Leónd L, Ruibala A (2009) The immunohistochemical expression of cyclin B1 is associated with higher maxSUV in 18F-FDG-PET in non-small cell lung cancer patients. Initial results. Rev Esp Med Nucl 28:63–65
Zhou YL, Deng CS (2005) Correlations of expressions of Glut1 and HIF-1alpha to cellular proliferation of colorectal adenocarcinoma. Ai Zheng 24:685–689
Sakamaki T, Casimiro MC, Ju X et al (2006) Cyclin D1 determines mitochondrial function in vivo. Mol Cell Biol 26:5449–5469
Lee SH, Heo JS, Han HJ (2007) Effect of hypoxia on 2-deoxyglucose uptake and cell cycle regulatory protein expression of mouse embryonic stem cells: involvement of Ca2+/PKC, MAPKs and HIF-1alpha. Cell Physiol Biochem 19:269–282
Strauss LG, Dimitrakopoulou-Strauss A, Haberkorn U (2003) Shortened PET data acquisition protocol for the quantification of 18F-FDG kinetics. J Nucl Med 44:1933–1939
Strauss LG, Pan L, Cheng C, Haberkorn U, Dimitrakopoulou-Strauss A (2009) Accuracy of shortened acquisition protocols for the quantitative assessment of the 2-tissue compartment method using dynamic PET F-18-deoxyglucose (FDG) studies. [abstract]. J Nucl Med 50(2):630
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Strauss, L.G., Koczan, D., Klippel, S. et al. Impact of Cell-Proliferation-Associated Gene Expression on 2-Deoxy-2-[18F]fluoro-d-Glucose (FDG) Kinetics as Measured by Dynamic Positron Emission Tomography (dPET) in Colorectal Tumors. Mol Imaging Biol 13, 1290–1300 (2011). https://doi.org/10.1007/s11307-010-0465-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-010-0465-z