Abstract
Patterns of plant diversity along the altitudinal gradient of Tianshan in central Xinjiang, China were examined. Plant and environment characteristics were surveyed from higher, south of Bogeda peak, to lower, north of Guerbantonggute desert. There were a total of 341 vascular plant, 295 herbage, 41 shrub, and seven tree species in the sampled plots. The plant richness of vegetation types generally showed a unimodal pattern along altitude, with a bimodal change of plant species number at 100-m intervals of altitudinal samples. The two belts of higher plant richness were in transient areas between vegetation types, the first in areas from dry grass to forest, and the second from forest to sub-alpine grass and bush. The beta diversity varied with altitudinal changes, with herbaceous species accounting for most species, and thus had similar species turnover patterns to total species. Matching the change of richness of plant species to environmental factors along altitude and correlating these by redundancy analysis revealed that the environmental factors controlling species richness and its pattern were the combined effects of temperature, precipitation, soil water, and nutrition. Water was more important at low altitude, and temperature at high altitude, and soil chemical and physical characters at middle altitudes. This study provides insights into plant diversity conservation of Bogeda Natural Reserve Areas in Tianshan Mountain.
Similar content being viewed by others
Introduction
Biodiversity distributions are important to natural reserve area planning and management. Knowing species richness patterns is crucial to provide insights to environmental planners, nature reserve designers, ecologists, and botanists. It is widely thought that species richness decreases with decreasing growing-season temperatures (Odland and Birks 1999), and this has been applied to both altitudinal (Rahbek 1995) and latitudinal gradients (Connor and McCoy 1979; Murray 1997). However, this hypothesis has been questioned by some evidence (Rahbek 1995, 1997; Vetaas and Grytnes 2002; Bhattarai and Vetaas 2003; Sanchez-Gonzalez and Lopez-Mata 2005), the unimodal and multi-modal plant diversity along altitude were observed in recent researches (Rahbek 1997; Fleishman et al. 1998; Heaney 2001; Grytnes 2003).
The plant diversity along altitudinal gradients is critical in understanding patterns of vegetation distribution in varied environmental conditions and broad longitude and latitude geographical expansions (Juarez et al. 2007; Kazakis Ghosn et al. 2007). Vegetation types and species composition vary greatly along environmental gradients. The geographic site and altitude affect species richness, their effects on species diversity can be demonstrated from South to North Poles and from sea level to mountain top (Gaston 2000; Allen et al. 2002). There are orderly vegetation zones along latitude, the spatial extents from one zonal vegetation type to another are very great, making it difficult and costly to study the species diversity and vegetation characters. The altitudinal gradient is often claimed to mirror the latitudinal gradient (Rahbek 1995; Fosaa 2004), mountains provided the best available test system to understand what might drive latitude effects of biodiversity (Körner 2000).
The plant richness along ecological gradients has important implications for community analysis. Some case studies showed that plant richness along altitude in arid and semi-arid areas are unimodal (Peet 1978; Wilson et al. 1990). However, interactions between species and extreme environmental stress may cause skewed or non-unimodal responses (Oksanen and Minchin 2002).
This investigation focused on the plant diversity and its environment features in Bogeda Peak Reserve, Xinjiang, China. Considering its obvious vertical vegetation belts in landscapes, arid natural conditions in region, and unique geographical position in central Euro-Asia continent, Bogeda Peak Reserve is an ideal place for studying changes of biodiversity along altitudinal gradients. It has rich species and communities, heterogeneous habitats with different climatic and soil combinations, many endemic, endangered and rare species and unique ecosystems (Hai and Zhang 1990). There has been little study of vascular plant species diversity and biodiversity patterns in this region.
Several questions were addressed in this research, including: How did vascular plant species-richness change from low to high altitudes in this area? Was there a particular pattern of plant diversity with environmental factor gradient? Were the turnover rates of species different between vegetation types across the altitudinal gradient?
Methods
Research site
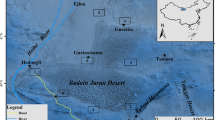
The geographic location of research was within 87°45′–88°05′E, 43°53′–44°30′N, in the North Slope of Bogeda Peak Reserve in Tianshan Mountain Ranges, Fukang County near Urumqi, the capital of Xinjiang Uigur Autonomous Region, China (Fig. 1). Tianshan Mountain begins at Pamiri Plateau; its length from west to east is ca. 2,500 km. About half of the mountain range is in China and the rest is in central Asian countries. The width of Tianshan Mountain is ca. 350 km, located between 41° and 44°N, Tianshan Mountain has many peaks higher than 4,000 m a.s.l., permanently covered with ice and snow and surrounded by desert, so they intercept water from the wet sea air current in the northwest. With increased elevation, the temperature decreases gradually and the climate is wetter, therefore there is more precipitation on the mountain at higher altitudes than the lower areas of the mountain, but precipitation in the highest areas was less than that in the upper–middle altitude belt. Modern glaciers are widely distributed in upper parts of the mountain ranges, with snow and ice generally around 3,800–5,000 m a.s.l. These glaciers are the main water sources for many rivers originating from Tianshan Mountain. There is forest and meadow vegetation in alpine and sub-alpine areas of the mountain range where there is sufficient rainfall and snow water.
Location of the research area and sample plots along the altitudinal gradient and the climatic stations. The right map shows rapid increase in steepness above 600 m a.s.l. from the northern plain to the southern mountain, and the topographic profile of the research areas in Fukang area of the middle section of Tianshan. The right map shows the locations of sample plots
Climate and soil
Changing climatic factors with altitude drives the formation of zonal vegetation and soil in mountain areas. There is typical zonal vegetation distributed in east and middle Tianshan Mountain. There are front terrains of mountain thrust into vast arid areas at modest slope, and these areas have typical climatic features of arid desert with annual precipitation 100–200 mm or less (Table 1). The precipitation is 500–650 mm (Table 1) in the middle of the mountain belt (1,500 m a.s.l.), and 700–800 mm (Table 1) in the alpine belt of the mountains (4,000 m a.s.l.). Hilly landscapes in higher elevations of the mountains are usually steep, wet and cold, with the average July temperature of no more than 16°C.
There are two meteorological stations within the research area with different elevations and contrasting climates (Table 1). The annual average temperature in lower Fukang City is much greater than the higher Tianchi Lake, but the differences in July and January temperatures in these two areas are similar. Other climatic factors including evaporation and sunlight hours are also significantly different. The evaporation in Fukang City is 1,811.9 mm year−1, and in Tianchi it is 1,371.0 mm year−1; and the sunlight hours are 3,099.4 and 3,027.7 h year−1 (Table 1), respectively (Zhou 1995).
There are brown calcic, desert calcic, desert forest, meadow, marsh, salty, crop cultured, and sandy soils in the research area. The brown calcic soils are mainly at altitude greater than 800 m a.s.l., desert calcic soils in plains area at altitude <800 m a.s.l.; meadow, marsh, salty and crop cultured soils are all in plains area at altitude <600 m a.s.l. (Jiang and Li 1990).
Field surveying and chemical analysis methods
A field survey of plant biodiversity and environmental factors was conducted along the transect from higher south of Bogeda peak to lower north of Guerbantonggute desert, the distance from south to north is about 80 km, relative altitudinal difference is about 5,000 m (Fig. 2). The change patterns of vegetation, plant species, soil, climate with altitude were studied and analyzed in detail, and the implication of this kind of gradient change with environmental changes was probed.
Fieldwork was carried out in summer, July 2001 and June–July 2002. Plants and soils were sampled along an altitudinal gradient from 450 to 3,400 m a.s.l. (Fig. 1). The sites were chosen randomly at 100-m intervals of altitude; with 2–3 plots surveyed at each altitude, plot dimensions of 20 × 20 m, with 87 plots sampled in total. The plots were divided into four subplots along two crossing diagonals. For each subplot, we recorded species, measured diameter at breast height, height, crown width of all trees of height ≥1.3 m, and number of individuals and height of trees <1.3 m. For the shrub survey, four 5 × 5 m subplots were sampled in each plot, the species and number of stems was recorded. We estimated density, cover, and measured height and crown width of grass by species in five 1 × 1 m subplots of each plot; if possible, species were recorded on site, otherwise, specimens were taken back for later identification. Three soil samples were taken from each surveyed plot. Wet and dry weights of all soils samples were used to calculate soil water content. Then we mixed the samples from one plot into one sample, the chemical elements and salt ions were analyzed in the Chemical Analysis Laboratory of the Institute of Xinjiang Ecology and Geography, Chinese Academy of Sciences. The pH was determined in a 1:2.5 (w:v) suspension of soil in water using a pH meter (EC-10; Hack). Organic C content was determined by the Tyurin method. Total nitrogen (N) (%) was ascertained by the micro-Kjeldahl procedure after digestion with concentrated H2SO4 and measurement of NH3 by the indophenols blue method on an autoanalyser, total potassium (K) (%) by NaOH fused flaming spectrum method, and total phosphorus (P) (%) HClO4–H2SO4 method. Electrical conductivity was determined by a saturated-paste method. Available N was extracted with 2 M KCl, available P with a 0.5 M NaHCO3 solution at pH 8.5 (Olsen’s method), and available K with 2 M ammonium acetate at pH 7.0.
Data analysis methods
The data were analyzed in two ways: (1) vegetation was classified into four vegetation types from the highest south to the lowest north; and (2) the characteristics of each vegetation type were analyzed using the field observation data. Analysis was according to altitude of 100-m intervals ordered from low to high elevation, the plant species and environmental patterns demonstrated using topographical variations.
Species richness, within community diversity, and between-community diversity was calculated for each altitude sample. The species richness was the number of species of plant life forms: tree, shrub and herbage. Paired plots diversity (β-diversity) was measured using the index of Wilson and Shmida (1984). Species turnover rate was calculated as the gain and loss of species between neighboring altitudes of 100-m intervals. The formula is:
where g(H) and l(H) are the number of species gained and lost, respectively, from altitude H − 1 to altitude H, and α(H) is the species richness at altitude H. This index is independent of species richness and sample size, and is directly related to degree of species turnover along the environmental gradient. Since sampling area can affect plant diversity (Rahbek 1995), we used two 20 × 20-m plots per 100-m altitudinal intervals from the plot set, so the sampling area at every 100-m elevation interval was 800 m2.
The relationships between plant richness and environmental factors along altitude gradients were analyzed by correlation analysis (SPSS 2000) and redundancy analysis (RDA; using CANOCO 4.5 software; Ter Braak and Šmilauer 2002). The significant environmental variables (P < 0.05) were selected after forward selection using an unrestricted Monte Carlo permutation test based on 9,999 random permutations.
Results
Vegetation character
The community types and the environmental factors at each altitude are presented in Table 2. Due to its unique geographic site, there are complete vertical vegetation zones of desert areas along altitude (Fig. 2).
From the highest to the lowest vegetation zones, there were large differences in plant species, both life form and physiognomy. The dominant species of meadow and grassland in 2,800–3,400 m a.s.l. were Jemerus spp., Sabina spp., and herbages. Forest was dominated by a spruce tree species Picea schrenkiana, it forms pure conifer forests, distributed in 1,700–2,800 m a.s.l. The grassland zone was below the forest, at 1,100–1,700 m a.s.l., with dominant plant species of sandy needle grass (Stipa glareosa P. Smirn), fescuegrass (Festuca spp. Linn.). Desert grassland had elevation of 800–1,100 m a.s.l. and was dominated by desert wormwood (Artemisia desertorum Spreng), songory reaumuria (Reaumuria soongorica), and saxoul (Haloxylon ammodendron). The desert vegetation was below 800 m a.s.l., with dominant species of songory reaumuria and saxoul.
The plant species richness in each altitudinal zone is presented in Fig. 3. The total number of species showed a pronounced maximum in the mid elevations from low to high elevation zonal types using this analysis, the number of herbaceous species had a similar trend to total species, but richness of shrub and tree species increased from low to high elevation. The total species richness at 400–1,100 m a.s.l. was about half that at 2,800–3,400 m a.s.l., indicating great biodiversity variations in the research region, and also possible physical environment changes in relatively small areas and short distances due to the large elevation differences.
Physiognomy changes with elevations were investigated, mainly through canopy height of dominant species. The dimensions of dominant plants were used to denote the physiognomy of the whole community. The canopy heights of vegetation types along elevations changed greatly. The meadow and grassland had the smallest canopies, herbaceous stratum average height was 10.92 ± 7.16 cm (mean ± SD), and average crown width was 7.39 ± 4.12 cm; the shrub stratum had average height of 10.00 ± 8.00 cm and average crown width 34.87 ± 14.53 cm. The forest had the highest canopy, average height of trees was 9.59 ± 5.31 m, average crown width 2.27 ± 1.08 m, and average diameter at breast height 14.49 ± 11.00 cm.
The species number changes of vegetation types with environmental factors
The plant species richness showed a unimodal-shape change from the lowest elevation desert bush with 86 species to desert grass 164, forest 208, and highest alpine meadow 166 (Table 2). The plant diversity pattern of vegetation types along altitude corresponded with precipitation, soil organic mass (%) and soil total N (%). But the mean annual temperature monotonously increased with elevation, which had distinctly different pattern from plant species, precipitation, soil organic mass and soil N content changes (Table 2).
Plant diversity characteristics with altitude change
There were 341 herbaceous, 41 shrub and seven tree species in the sample plots. The general trend in species richness along altitudinal gradient was that species numbers gradually increased up to 1,500 m a.s.l., decreased from 1,500 to 1,800 m a.s.l., moderately increased from 1,800 to 2,400 m a.s.l., then greatly increased at 2,500 m a.s.l., and decreased at 3,300 m a.s.l. when approaching the upper vegetation boundary (Fig. 4). The pattern of species number was bimodal, the most species were at elevations 1,500 and 2,700 m a.s.l., and there were some fluctuations of plant species number along the altitude gradient. The maximum number was 64 species in one sampled elevation area of 800 m2 and the least was ten species. The altitudes at which there were most species were typical transitions from one vegetation type to another, elevation 1,600 m a.s.l. was within the transition between meadow and grass to forest; elevation 2,700 m a.s.l. was from the transition between forest and grass land. Shrubs accounted for little of total species composition in the sample plots, there was only two shrub species in 2,700–3,000 m a.s.l., 17 in 1,700–2,700 m, 24 in 1,100–1,700 m a.s.l., and 23 in 450–1,100 m a.s.l. There were a total of seven tree species, the most widely distributed and abundant was spruce, distributed at altitudes of 1,600–2,700 m a.s.l., and mainly in shady aspects as pure spruce forest. The two other tree species that frequently occurred were poplar (Populus diversifolia Schrenk) and elm (Ulmus pumila L.), but the species abundances were less.
The β-diversity index along elevation revealed that the species turnover rates were rapid in some extreme climatic conditions (Fig. 5). Since there were few tree and shrub species, the tree β-diversity index had a very different change pattern to the herbage and total species. Generally, the total and herbaceous species turnover rates were greater at lower elevations than higher, especially in the desert and semi-desert vegetation belts. As elevations increased, the turnover rate decreased; and above the forest zone the rate increased again at alpine and sub-alpine grasslands.
Chemical properties of soil with altitude
In this study, the relevant environmental resources are energy, water and nutrition. The energy and precipitation are closely related to altitude, and water available for plant growth can be in the form of soil water. The nutrition is represented by soil chemical factors (Fig. 6).
Soil chemical and physical features change with altitude. Organic matter (a), pH (b), electrical conductivity (c), soil water content (d) are given as average ± standard deviation. The soil nutrition is in two groups, total amount in % N (e), K (g) and P (f), available amount in ppm N (e), P (f) and K (g)
Soil organic matter along altitudes was unimodal, with the highest value in the forest belt (Fig. 6a). Soil pH changed dramatically from low to high elevation, pH was greater at low than high altitudes. The electrical conductivity in forest zonal soil was greater than other community types. Water in soil was closely related to vegetation types and productivity, with high water content in forest soils (Fig. 6b–d). The elements, N, P, and K in soil can be measured in two ways, percentage (total) soil contents (Fig. 6e–g), and that available to plants in ppm in soil (Fig. 6h–j). There was no obvious pattern to P and K, but N amount changed with elevation, being unimodal with the highest amount in the forest belt (1,700–2,500 m a.s.l.).
The relationship of plant richness to environmental factors
The relationships between plant richness to soil and topographical factors were explored using RDA analysis. The elevations were divided into ranges according to the number of species (Fig. 7). The slope, available N, elevation and organic matter had close relationships with species richness; the desert and semi-desert plant communities had low species richness (10–20 species) and negative correlations with elevation, and positive correlations with pH. The plots with greater than 60 plant species had weaker relationships with soil nutrition and water content.
RDA analysis of species richness with environmental factors, the numbers refer to the ranges of species, Rich plant species richness. The environmental factors are: Ele elevation, Slope, Aspect, pH in soil, Soil W water content in soil, Org Matter organic matter, N., K. refer to N, K % content and Quick N, K refer to available N, K (ppm) content in soil. The gray and black arrows refer to species richness and environmental factors, respectively
Discussion
The research area is in the central area of the Euro-Asia continent, and there is little rainfall. In such climatic conditions, the zonal vegetation should be mainly desert and desert shrub. However, the Tianshan Mountain Range intercepts water from Pacific and Atlantic air currents at high altitude, and has diverse landscapes, which have increased biodiversity more than expected in this area. Thus it is an ideal area for studying biodiversity patterns and dynamics in natural and disturbed ecosystems. There are many protecting plants: Populus diversifolia Schrenk, Haloxylon ammodendron Bge., Haloxylon persicum Bge. Ex Boiss; and medical plants: Corydalis glaucescens Rgl., Glycyrrhiza aspera Pall, Glycyrrhiza uralensis Fisch, Cynomorium songaricum Rupr., Cistanche salsa C.A. Mey in the study area. It is a very unique area of flora, especially for biodiversity conservation, and these plants have been listed in the China Plant Red Data Book (Fu and Jin 1992).
The plant species richness had a unimodal pattern when the sample plots were grouped into vegetation types by an ordinal altitude; the forest had the highest plant diversity and semi-desert grassland the next highest. This pattern is consistent with unimodal pattern theory. The canopy height of forest was ten times that of grass and bush vegetation, and using this as an indicator of community productivity indicated that the forest had very high production relative to other communities. We found that the community richness had a close relationship with temperature, precipitation, and soil N and organic matter.
Other studies indicated that species richness would be greatest at intermediate altitudes in the transition between two zonal vegetation types (Lomolino 2001). If we did not divide plots into vegetation groups, but analyzed the data by altitudinal order, the species richness of plants along the altitude gradient is inconsistent with the hypothesis that species richness decreases and unimodal with altitude (Woodward 1987; Körner 1995; Fosaa 2004). The plant species richness appeared to fluctuate, and plant biodiversity change along altitude had patterns which correlated with climatic and soil factors. The richness of plant species increased with altitude from the lowest to highest points (450 and 1,500 m a.s.l., respectively), declined in the forest belt of 1,600–2,700 m a.s.l., then increased in sub-alpine grass belts, and finally decreased in alpine meadow. Both of the peaks of plant biodiversity occurred in transition areas, one from dry grass to forest, and the other from forest to sub-alpine grass and bush. It is likely that elevation ranges with high richness are transition zones in which the effects of temperature, soil water and local environmental factors (aspect, slope, organic matter, water content, and soil nutrition) combine with contrasting spatial patterns (some increase with elevation while others decrease), resulting in a greater species interchange between communities. It is the elevation equivalent of the well-known “ecotone effect” (Lomolino 2001; Mark and Wilbur 2002; Sanchez-Gonzalez and Lopez-Mata 2005). Grim (1979) and Huston and DeAngelis (1994) found species richness was lower where resources were lowest, was higher at intermediate resources levels, and decreased gradually as resource levels increased. One of the most conspicuous features of the altitudinal gradient along the northern slope of middle Tianshan is the extreme aridity in the lowlands and low temperatures at high elevations; this indicates that species richness should be greatest at intermediate altitudes, as observed in North American deserts (Whittaker and Niering 1975). This hypothesis comes from the favorability concept, in which areas with stressful environmental characteristics and/or extreme unpredictability have very low plant diversity. Such a species diversity pattern is supported in the present research, both poor and rich resources areas have low species numbers because the resource supply is close to the minimal threshold required by most of the species to survive. Tilman (1982) suggested that habitats rich in resources will be rapidly dominated by a low number of competitively superior species. Hence, higher diversity occurred in belts with intermediate resource levels, in which most species could coexist. Such unimodal diversity has been widely reported in plant and animals (Tilman and Pacala 1993).
Due to the various topographies in the middle section of Tianshan Range, the combinations of different physical environment factors create diverse habitats in the research area. Of all the physical factors, elevation was the key one, as it greatly controls or influences the other factors. With increasing or decreasing elevation, the physical environmental features varied and showed some regular patterns. The climatic factors have different changing patterns: temperature varied definitely with elevation in a consistent way, with increased elevation the temperature was lower. This trend was reversed at some elevations, with temperature inversion phenomena in the research area. Precipitation pattern was unimodal-shape with increasing altitude, the most precipitation was at 2,600 m a.s.l., but precipitation decreased with increased altitude above 2,600 m a.s.l., at a lesser rate than with decreased altitude below 2,600 m a.s.l.
The water content, soil organic matter and soil nutrients were important factors affecting plant biodiversity patterns. The soil organic matter contents in each elevation (Fig. 6a) were <5% at 400–1,300 m a.s.l., 5–15% at 1,300–1,600 m a.s.l., 15–25% at 1,700–2,800 m a.s.l., and 8–15% beyond 2,800 m a.s.l.; this was in a unimodal pattern with the maximum in the forest zone, probably due to high productivity of the forest, and that it has more soil organic matter than other vegetation types. Another reason was that soil organic matter decays slowly at low temperature, resulting in high accumulation of humus (Sanchez-Gonzalez and Lopez-Mata 2005). At high elevation, oxygen deficiency can combine with low temperatures and produce even lower decomposition rates.
The pH tended to be lower at higher elevations, with values <6.9. The soil was alkaline below 1,500 m a.s.l. and acidic above 1,500 m a.s.l. Thus plants adapted to desert environments can live in the low altitude areas. The soil water content had a similar pattern to soil organic matter, a unimodal change with increased elevations. The highest soil water content was in the middle of the forest zone, almost the same elevation as the highest organic matter, demonstrating that decomposition was slow at high soil water contents. The soil electrical conductivity was unimodal with the maximum in the middle of the altitudinal gradient apart from an abnormal point at low elevation. We found that both the K and P contents irregularly changed, the total K (%) and available K (ppm) content decreased with increased altitude, with r = −0.64 (P < 0.01) and −0.387 (P < 0.05), respectively (Table 3). The total P (%) and available P (ppm) content increased with altitude, with r = 0.421 and −0.09, respectively. The N resource was the most important factor affecting plant biodiversity and was consistently related to altitude. There was very little soil N in low elevation desert plains, but was much higher in middle elevations where the spruce forest was dominant, and decreased a little in sub-alpine and alpine meadows.
The resource patterns along the altitudes rapidly changed from one zone to another, and there were large differences within each environmental factor. The annual precipitation in desert grassland was 227.2 mm, much less than forests with 533.1 mm (Table 1). Annual mean temperature in semi-desert grassland of 6.3°C was much higher than 0.28°C in the forest zone (Table 2). These extreme zones were not the sites of the most plant diversity. The coexistence of a greater number of species was influenced by local variations in the relative quantity of nutrients, soil physical characteristics, topography and biotic factors.
The β-diversity showed species turnover rates varied greatly with altitude (Fig. 5). The species turnover rates were higher at 400–1,700 m a.s.l., the highest zone was 600 m a.s.l. in the range of desert vegetation, and at 1,800 m a.s.l. in the transition zone from grass to forest vegetation. The dominant plant species in desert and semi-desert bush and grassland were suitable for alkali soils and dry climates. These plants include Haloxylon ammodendron (C.A. Mey.) Bge., Kalidium foliatum (Pall.) Moq., Halostachys caspica (M.B.) C.A. Mey., Suaeda physophora Pall., et al. shrubs, and Suaeda Forsk. Ex Scop., and Petrosimonia sibirica (Pall.) Bge., et al. herbages within 400–700 m a.s.l. These plants disappeared at greater altitudes, and plants adapted to semi-desert appeared within 700–1,100 m a.s.l., they are Atraphaxis replicata Lam. et al. The species within 1,100–1,700 m a.s.l. were diverse with no typical dominant species, but some special species such as Rosa platyacantha Schrenk., Berberis heteropoda Schrenk., Festuca ovina L., and Peganum harmala L. et al. The species turnover rates in the forest zone of 1,700–2,800 m a.s.l. were less than at 400–1,700 m a.s.l., but there was a peak at 1,800 m a.s.l. This was caused by declining grassland species and increasing forest species. The dominant tree species were Apicea schrenkian, shrubs and herbaceous species include Lonicera hispida Pall. and Calamagrostis arundinacea (L.) Roth et al. The β-diversity in meadows above 2,800 m a.s.l. had large fluctuations, until 3,000 m a.s.l. it was similar to the forest zone. Above 3,000 m a.s.l., the turnover rate was the least of the whole altitude range, then it increased dramatically. This may be that vegetation reached its upper limiting zone, as most species cannot tolerate the harsh low temperature environment.
The herbaceous cover had a close positive relationship with species richness (r = 0.708, P < 0.01, 2-tailed). The change of herbaceous species can represent the plant richness dynamics and is a good indicator of plant species diversity. The lowest values of herbaceous cover and shrub density were in desert grass and bush types (400–700 m a.s.l.). The vegetation is sparse in this zone because of lack of water, and temperature and nutrition were not limiting factors. The cover value of herbaceous species was low in the forest zone where the dominant species were spruce trees. With both trees and shrubs, the cover value was high, and the diversity and vegetation cover was negatively related. Therefore total cover was not an index of plant diversity, but net primary productivity (Fig. 8a). The density of shrub and tree species had a relatively steady pattern, since they appeared along the altitudinal gradient (Fig. 8b).
Learning altitudinal species-richness patterns and their relationships with environmental factors are very important to resolve the problems of biodiversity conservation and its changes with global change and human disturbances (Grytnes 2003). Middle Tianshan Mountain Range, at the center of the Euro-Asia continent, has complicated topography and climatic conditions, and is a sensitive region to global climatic change. The diverse landscape and micro-habitats make it ideal for experiments on the effects of global change and human disturbance to vegetation, therefore further investigation on the climatic change on plant diversity will be necessary. This study had just one survey time per site, thus flowering plants of other seasons such as spring or autumn would be missed, any species appearing with seasonal changes should be monitored in future research in this area.
Conclusions
This study found that the diversity of plant species at the middle of Tianshan Mountain Range increased significantly from low elevations to the transition zone between semi-desert grass to forest at 1,500 m a.s.l., decreased modestly in the forest zone, then increased at the transition from forest to alpine meadow at 2,700–2,900 m a.s.l., and finally decreased near the upper limit of vegetation.
The vegetation changes along altitude were in an ordered pattern, with the number of species in a unimodal pattern. The total number of species had a multi-modal shape from high elevation to low elevation, the herbaceous species number followed a similar trend to total species, but the numbers of shrub and tree species increased from high elevation to low elevation and decreased a little in lowest desert vegetation types. The total number of species at 450–1,100 m a.s.l. was about half that at 3,400–2,700 m a.s.l., showing great variation in biodiversity in the research region, and also the possible physical environment changes in a relatively small area from large differences in elevation. This is similar to the change in number of species across latitude from north to south in the Northern Hemisphere. The relationships between species abundance and environmental factors were weak. There were strong relationships among vegetation canopy height and climatic and soil factors, and these relationships were usually positive. We conclude that climatic and soil factors changed in a regular way along altitudinal gradient, and biodiversity was irregular, and canopy height of plants was positive correlated with elevation. The canopy average and maximum heights were significantly correlated with precipitation, soil N and soil organic matter.
The β-diversity decreased gradually with increasing elevation, with higher values at the low elevations, lowest values in forest and meadow zones in the upper section of the elevation gradient. Few species are adapted to live in the full spectrum of variation in resource conditions, at some extreme altitudes the species turnover rates were close to 100%. Differences in climatic, topographic and edaphic factors along the elevation gradient can explain the species richness and diversity pattern.
Matching the change in richness of plant species to environmental factors along altitudinal gradient, and correlating these using RDA, we found that the main environmental factors controlling species richness and its pattern depended on combined effects of temperature, precipitation, soil water and soil nutrition. Water was important at low altitude, temperature at high altitude, and soil chemical and physical characters at middle altitudes. The research results provided some insights on plant biodiversity patterns and conservation of Bogeda Natural Reserve Areas.
References
Allen AP, Brown JH, Gillooly JF (2002) Global biodiversity, biochemical kinetics, and the energetic-equivalence rule. Science 297:1545–1548
Bhattarai KR, Vetaas OR (2003) Variation in plant species richness of different life forms along a subtropical elevation gradient in the Himalayas, east Nepal. Glob Ecol Biogeogr 12:327–340
Connor EF, McCoy ED (1979) The statistics and biology of the species–area relationship. Am Nat 113:791–833
Fleishman E, Austin GT, Weiss AD (1998) An empirical test of Rapoport’s rule: elevational gradients in montane butterfly communities. Ecology 79:2483–2493
Fosaa AM (2004) Biodiversity patterns of vascular plant species in mountain vegetation in the Faroe Islands. Divers Distrib 10:217–223
Fu LG, Jin JM (1992) China plant red data book-rare and endangered plants, volume 1. Science Press, Beijing
Gaston KJ (2000) Global patterns in biodiversity. Nature 405:220–227
Grim JP (1979) Plant strategies and vegetation processes. Wiley, New York
Grytnes JA (2003) Species-richness patterns of vascular plants along seven altitudinal transects in Norway. Ecography 26:291–300
Hai Y, Zhang L (1990) Plant name lists in Fukang Desert Ecosystem Station and its nearby area (in Chinese). Arid Zone Res 7(Supp.):44–48
Heaney RH (2001) Small mammal diversity along elevational gradients in the Philippines: an assessment of patterns and hypotheses. Glob Ecol Biogeogr 10:15–39
Huston M, DeAngelis DL (1994) Competition and coexistence: the effects of resources transport and supply rates. Am Nat 144:954–977
Jiang H, Li S (1990) Soil characteristics in Fukang arid ecosystem station and its neighbor area (in Chinese). Arid Zone Res 7(Supp.):6–13
Juarez A, Ortega-Baes P, Suhring S, Martin W, Galındez G (2007) Spatial patterns of dicot diversity in Argentina. Biodivers Conserv 16:1669–1677
Kazakis Ghosn GD, Vogiatzakis IN, Papanastasis VP (2007) Vascular plant diversity and climate change in the alpine zone of the Lefka Ori, Crete. Biodivers Conserv 16:1603–1615
Körner C (1995) Alpine plant diversity: a global survey and functional interpretation. In: Chapin SF, Korner C (eds) Arctic and alpine biodiversity, pattern, causes and ecosystem consequences. Ecolog Studies 113:45–60
Körner C (2000) Why are there global gradients in species richness? Mountains might hold the answer. Trends Ecol Evol 15:513–514
Lomolino VM (2001) Elevation gradients of species-density: historical and prospective view. Global Ecol Biogeogr 10:3–13
Mark CG, Wilbur HM (2002) Ecology of ecotones: interactions between Salamanders on a complex environmental gradient. Ecology 83(8):2112–2123
Murray DF (1997) Regional and local vascular plant diversity in the Arctic. Opera Bot 132:9–18
Odland A, Birks HJB (1999) The altitudinal gradient of vascular plant richness in Aurland, western Norway. Ecography 22:548–566
Oksanen J, Minchin PR (2002) Continuum theory revisited: what shape are species responses along ecological gradients? Ecol Model 157:119–129
Peet RK (1978) Forest vegetation of the Colorado Front Range. Vegetatio 37:65–78
Rahbek C (1995) The elevational gradient of species richness: a uniform pattern? Ecography 18:200–205
Rahbek C (1997) The relationship among area, elevation, and regional species richness in Neotropical birds. Am Nat 149:875–902
Sanchez-Gonzalez A, Lopez-Mata L (2005) Plant species richness and diversity along an altitudinal gradient in the Sierra Nevada, Mexico. Divers Distrib 11:567–575
SPSS (2000) Advanced statistical analysis using SPSS (v10.0). Chicago, Illinois
Ter Braak CJF, Šmilauer P (2002) CANOCO reference manual and CanoDraw for Windows user’s guide: software for canonical community ordination (version 4.5). Microcomputer, Ithaca, New York
Tilman D (1982) Resource competition and community structure. Princeton University Press, New Jersey
Tilman D, Pacala S (1993) The maintenance of species richness in plant communities. In: Ricklefs RE, Schluter D (eds) Species diversity in ecological communities. Historical and geographical perspectives. The University of Chicago Press, Chicago, pp 13–25
Vetaas OR, Grytnes JA (2002) Distribution of vascular plants species richness and endemic richness along the Himalayan elevation gradient in Nepal. Glob Ecol Biogeogr 11:291–301
Whittaker RH, Niering RH (1975) Vegetation of the Santa Catalina Mountains, Arizona. V. Biomass, production and diversity along the elevation gradient. Ecology 56:771–790
Wilson MV, Shmida A (1984) Measuring beta diversity with presence–absence data. J Ecol 72:1055–1064
Wilson JB, Lee WG, Mark AF (1990) Species diversity in relation to ultramafic substrate and to altitude in southwestern New Zealand. Plant Ecol 86:15–20
Woodward FI (1987) Climate and plant distribution. Cambridge studies in ecology. Cambridge University Press, Cambridge
Zhang X (1959) Geographic distribution of forests in East Tianshan areas. In: Xinjiang Integrated Survey Team of Chinese Academy of Sciences, Geography Institute of Russian Academy of Sciences, USSR (eds) The nature condition of Xinjiang Uigur autonomous region. Collected Publications Science Press, Beijing, China, pp 201–226
Zhou X (1995) Vertical climatic difference in the middle section of northern slope of Tianshan mountains. Arid Land Geogr 18(2):52–60
Acknowledgments
The study was financially supported by a major project of National Natural Science Foundation (Grant No. 30590382). Many thanks to Mr Jin Jiang, Drs Yuanming Zhang, Haibao Ren, Ting Wang, Shixin Wu, Professor Borong Pan for their help in the field surveying. I am grateful to two anonymous reviewers for helpful critiques, suggestions and comments to significantly improve earlier versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Nomenclatures: the scientific name for plants follows Flora of China (Compiling Committee of Flora of China).
About this article
Cite this article
Sang, W. Plant diversity patterns and their relationships with soil and climatic factors along an altitudinal gradient in the middle Tianshan Mountain area, Xinjiang, China. Ecol Res 24, 303–314 (2009). https://doi.org/10.1007/s11284-008-0507-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-008-0507-z