Abstract
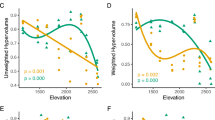
Phenotypic plasticity is a common feature of plant invaders, but little is known about variation in plasticity among invading populations. Variation in plasticity of ecologically important traits could facilitate the evolution of greater plasticity and invasiveness. We examined plasticity among invasive populations of Microstegium vimineum (Japanese stiltgrass), a widespread and often dominant grass of forests in the eastern U.S. with two separate experiments. First, we exposed seven Microstegium populations to a drought treatment in growth chambers and monitored growth and physiological responses. Then, we established a greenhouse experiment using a subset of the populations; two that exhibited the most divergent responses and one intermediate population. In the greenhouse, we manipulated drought and shade and evaluated biomass production and specific leaf area (SLA). Microstegium exhibited plasticity for biomass production and SLA in the greenhouse experiment, and populations significantly varied in the degree of plasticity under drought and shade treatments. Two populations significantly increased biomass production under favorable conditions, unlike the third population. The most productive populations also responded to shade stress via greater SLA, possibly allowing for greater utilization of available light, while the third population did not. These results show that Microstegium can exhibit plastic responses to environmental conditions. Moreover, variation for plasticity among populations provides the potential for further evolution of plasticity. Future studies should focus on the relative importance of plasticity for the success of Microstegium and other plant invaders and evaluate post-introduction evolution of plasticity.



Similar content being viewed by others
References
Agrawal AA (2001) Ecology—phenotypic plasticity in the interactions and evolution of species. Science 294:321–326
Baker HG (1965) Characteristics and modes of origin of weeds. In: Baker HG, Stebbins GL (eds) The genetics of colonizing species. Academic Press, New York, p 588
Barden LS (1987) Invasion of Microstegium vimineum (Poaceae), an exotic, annual, shade-tolerant, C-4 grass, into a North Carolina floodplain. Am Midland Nat 118:40–45
Bossdorf O, Auge H, Lafuma L, Rogers WE, Siemann E, Prati D (2005) Phenotypic and genetic differentiation between native and introduced plant populations. Oecologia 144:1–11
Bossdorf O, Lipowsky A, Prati D (2008) Selection of preadapted populations allowed Senecio inaequidens to invade Central Europe. Divers Distrib 14:676–685
Burns JH, Winn AA (2006) A comparison of plastic responses to competition by invasive and non-invasive congeners in the Commelinaceae. Biol Invasions 8:797–807
Cheplick GP (2006) A modular approach to biomass allocation in an invasive annual (Microstegium vimineum; Poaceae). Am J Botany 93:539–545
Claridge K, Franklin SB (2002) Compensation and plasticity in an invasive plant species. Biol Invasions 4:339–347
Cole PG, Weltzin JF (2004) Environmental correlates of the distribution and abundance of Microstegium vimineum, in east Tennessee. Southeastern Nat 3:545–562
Cole PG, Weltzin JF (2005) Light limitation creates patchy distribution of an invasive grass in eastern deciduous forests. Biol Invasions 7:477–488
Conner JK (2003) Artificial selection: a powerful tool for ecologists. Ecology 84:1650–1660
Crooks JA (2005) Lag times and exotic species: the ecology and management of biological invasions in slow-motion. Ecoscience 12:316–329
Ellstrand NC, Schierenbeck KA (2000) Hybridization as a stimulus for the evolution of invasiveness in plants? Proc Natl Acad Sci USA 97:7043–7050
Fairbrothers DE, Gray JR (1972) Microstegium vimineum (Trin.) A. Camus (Gramineae) in the United States. J Torrey Bot Soc 99:97–100
Feng YL, Wang JF, Sang WG (2007) Biomass allocation, morphology and photosynthesis of invasive and noninvasive exotic species grown at four irradiance levels. Acta Oecol 31:40–47
Flory SL (2009) Management of Microstegium vimineum invasions and recovery of resident plant communities. Restor Ecol (in press)
Flory SL, Clay K (2009) Invasive plant removal method determines native plant community responses. J Appl Ecol 46:434–442
Flory SL, Rudgers JA, Clay K (2007) Experimental light treatments affect invasion success and the impact of Microstegium vimineum on the resident community. Nat Areas J 27:124–132
Gibson DJ, Spyreas G, Benedict J (2002) Life history of Microstegium vimineum (Poaceae), an invasive grass in southern Illinois. J Torrey Bot Soc 129:207–219
Grotkopp E, Rejmanek M (2007) High seedling relative growth rate and specific leaf area are traits of invasive species: phylogenetically independent contrasts of woody angiosperms. Am J Bot 94:526–532
Hamilton MA, Murray BR, Cadotte MW, Hose GC, Baker AC, Harris CJ, Licari D (2005) Life-history correlates of plant invasiveness at regional and continental scales. Ecol Lett 8:1066–1074
Higgins SI, Richardson DM, Cowling RM, Trinder-Smith TH (1999) Predicting the landscape-scale distribution of alien plants and their threat to plant diversity. Conserv Biol 13:303–313
Kolar CS, Lodge DM (2001) Progress in invasion biology: predicting invaders. Trends Ecol Evol 16:199–204
Lake JC, Leishman MR (2004) Invasion success of exotic in natural ecosystems: the role of disturbance, plant attributes and freedom from herbivores. Biol Conserv 117:215–226
Lavergne S, Molofsky J (2007) Increased genetic variation and evolutionary potential drive the success of an invasive grass. Proc Natl Acad Sci USA 104:3883–3888
Leger EA, Rice KJ (2003) Invasive California poppies (Eschscholzia californica Cham.) grow larger than native individuals under reduced competition. Ecol Lett 6:257–264
Leishman MR, Haslehurst T, Ares A, Baruch Z (2007) Leaf trait relationships of native and invasive plants: community- and global-scale comparisons. New Phytol 176:635–643
Mack RN, Simberloff D, Lonsdale WM, Evans H, Clout M, Bazzaz FA (2000) Biotic invasions: causes, epidemiology, global consequences, and control. Ecol Appl 10:689–710
Maron JL, Vila M, Bommarco R, Elmendorf S, Beardsley P (2004) Rapid evolution of an invasive plant. Ecol Monogr 74:261–280
McNulty SG, Aber JD (2001) US national climate change assessment on forest ecosystems: an introduction. Bioscience 51:720–722
Muth NZ, Pigliucci M (2007) Implementation of a novel framework for assessing species plasticity in biological invasions: responses of Centaurea and Crepis to phosphorus and water availability. J Ecol 95:1001–1013
NOAA and NCDC (2008) National Oceanic and Atmospheric Administration, National Climate Data Center. http://www.ncdc.noaa.gov/oa/ncdc.html
Nussey DH, Postma E, Gienapp P, Visser ME (2005) Selection on heritable phenotypic plasticity in a wild bird population. Science 310:304–306
Oswalt CM, Oswalt SN, Clatterbuck WK (2007) Effects of Microstegium vimineum (Trin.) A. Camus on native woody species density and diversity in a productive mixed-hardwood forest in Tennessee. For Ecol Manag 242:727–732
Pigliucci M (2001) Phenotypic plasticity: beyond nature and nurture. The Johns Hopkins University Press, Baltimore
Redman DE (1995) Distribution and habitat types for Nepal Microstegium [(Microstegium vimineum (Trin.) Camus] in Maryland and the District of Columbia. Castenea 60: 270–275
Rejmanek M (2000) Invasive plants: approaches and predictions. Aust Ecol 25:497–506
Richards CL, Bossdorf O, Muth NZ, Gurevitch J, Pigliucci M (2006) Jack of all trades, master of some? On the role of phenotypic plasticity in plant invasions. Ecol Lett 9:981–993
Roach DA, Wulff RD (1987) Maternal effects in plants. Annu Rev Ecol Syst 18:209–235
SAS Institute Inc (2002) Cary, NC, USA
Schlichting CD (1986) The evolution of phenotypic plasticity in plants. Annu Rev Ecol Syst 17:667–693
USDA and NRCS (2005) The PLANTS Database. Data compiled from various sources by Mark W. Skinner. Version 3.5. National Plant Data Center, Baton Rouge, LA 70874-4490 USA
Valladares F, Sanchez-Gomez D, Zavala MA (2006) Quantitative estimation of phenotypic plasticity: bridging the gap between the evolutionary concept and its ecological applications. J Ecol 94:1103–1116
van Kleunen M, Fischer M (2008) Adaptive rather than non-adaptive evolution of Mimulus guttatus in its invasive range. Basic Appl Ecol 9:213–223
Whitcraft CR, Talley DM, Crooks JA, Boland J, Gaskin J (2007) Invasion of tamarisk (Tamarix spp.) in a southern California salt marsh. Biol Invasions 9:875–879
Winter K, Schmitt MR, Edwards GE (1982) Microstegium vimineum, a shade-adapted C-4 grass. Plant Sci Lett 24:311–318
Zedler PH, Black C (2004). Exotic plant invasions in an endemic-rich habitat: the spread of an introduced Australian grass, Agrostis avenacea J. F. Gmel., in California vernal pools. Aust Ecol 29:537–546
Zou J, Rogers WE, Siemann E (2007) Differences in morphological and physiological traits between native and invasive populations of Sapium sebiferum. Funct Ecol 21:721–730
Acknowledgments
We thank Jennifer Rudgers, Lynda Delph, Heather Reynolds, Susan Cook, David Civitello, and Angie Shelton whose comments greatly improved the manuscript. We also thank Chris Herlihy who provided logistical support and advice on data collection and interpretation. Financial support was provided by the USDA Forest Service Hoosier National Forest, The Nature Conservancy, and the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Droste, T., Flory, S.L. & Clay, K. Variation for phenotypic plasticity among populations of an invasive exotic grass. Plant Ecol 207, 297–306 (2010). https://doi.org/10.1007/s11258-009-9673-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-009-9673-5