Abstract
Background
Oral anticoagulation (OA) is a common treatment with a known risk of fatal or major bleeding, but also minor bleeding symptoms and menorrhagia can cause substantial discomfort and necessitate medical or surgical interventions. The extent of these side effects is however not previously reported. The objective of this study is to assess the frequency of minor bleeding symptoms and menorrhagia attributed to OA treatment.
Methods
Ninety fertile women between 15 and 49 years-of-age on OA treatment completed an inquiry at the anticoagulation clinics of Malmö, Lund and Gothenburg, Sweden.
Results
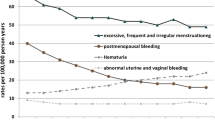
The frequency of minor bleeding symptoms was significantly increased during OA treatment (P < 0.05) except for hematuria. The incidence of bleeding after tooth extraction (>3 h) increased from 3.0 to 45.2%, easy bruising 17.8–75.6%, epistaxis 11.1–23.6%, gingival bleeding 22.2–48.3% and hematuria 10.0–15.6% (Table 1). Hematemesis was reported in 5.6% prior to as compared to 14.4% during OA treatment, blood in the feces in 8.9 and 18.9%, respectively. Mean duration of menses increased from 5.6 to 6.1 days (P < 0.01) and reported menorrhagia from 44.2 to 70.8% (P < 0.001). Eighteen percent were treated for menorrhagia before and 29.9% during OA treatment (P < 0.01).
Conclusions
OA treatment is known to confer increased risk of fatal or major bleeding. This study shows that fertile women on OA also experience significantly increased minor bleeding symptoms including menorrhagia that may considerably impair quality of life.
Similar content being viewed by others
Abbreviations
- OA:
-
Oral anticoagulation
References
Johansson H (1999) Anticoagulant therapy can be safer: an active follow-up and written care programs reduce the number of complications [in Swedish]. Läkartidningen 96:3388–3390
Palareti G, Leali N, Coccheri S, Poggi M, Manotti C, Dángelo A, Pengo V, Reba N, Moia M, Ciavarella N, Devoto G, Merrettini M, Musolesi S (1994) Bleeding complications of oral anticoagulant treatment: an inception-cohort, prospective collaborative study (ISOCOAT). Lancet 343:687-691
Själander A, Engström G, Berntorp E, Svensson P (2003) Risk of hemorrhagic stroke in patients with oral anticoagulation compared to the general population. J Intern Med 254:434-438
van Eijkeren M, Christiaens G, Haspels A, Sixma J (1990) Measured menstrual blood loss in women with a bleeding disorder using oral anticoagulant therapy. Am J Obstet Gynecol 62:1261-1263
Olsson BS et al (2003) SPORTIF III investigators stroke prevention with the oral direct thrombin inhibitor ximegalatran compared with warfarin in patients with non-valvular atrial fibrillation (SPORTIF III): randomized controlled trial. Lancet 362:1691-1698
Wallentin L, Wilcox RG, Weaver WD, Emanuelsson H, Goodvin A, Nystrom P, Bylock A (2003) Oral ximegalatran for secondary prophylaxis after myocardial infarction: the ESTEEM randomized controlled trial. Lancet 362:789-797
Schulman S, Wahlander K, Lundstrom T, Clason SB, Eriksson H (2003) THRIVE III Investigators. Secondary prevention of venous thromboembolism with the oral direct thrombin inhibitor ximegalatran. N Eng J Med 349:1713–1721
Acknowledgments
This study was supported by grants from Research funds from Malmö University Hospital, Lund University and Region Skåne.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Själander, A., Friberg, B., Svensson, P. et al. Menorrhagia and minor bleeding symptoms in women on oral anticoagulation. J Thromb Thrombolysis 24, 39–41 (2007). https://doi.org/10.1007/s11239-006-0003-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-006-0003-7