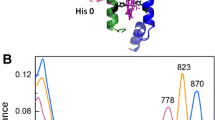
Abstract
Biohybrid antennas built upon chromophore–polypeptide conjugates show promise for the design of efficient light-capturing modules for specific purposes. Three new designs, each of which employs analogs of the β-polypeptide from Rhodobacter sphaeroides, have been investigated. In the first design, amino acids at seven different positions on the polypeptide were individually substituted with cysteine, to which a synthetic chromophore (bacteriochlorin or Oregon Green) was covalently attached. The polypeptide positions are at –2, –6, –10, –14, –17, –21, and –34 relative to the 0-position of the histidine that coordinates bacteriochlorophyll a (BChl a). All chromophore–polypeptides readily formed LH1-type complexes upon combination with the α-polypeptide and BChl a. Efficient energy transfer occurs from the attached chromophore to the circular array of 875 nm absorbing BChl a molecules (denoted B875). In the second design, use of two attachment sites (positions –10 and –21) on the polypeptide affords (1) double the density of chromophores per polypeptide and (2) a highly efficient energy-transfer relay from the chromophore at –21 to that at –10 and on to B875. In the third design, three spectrally distinct bacteriochlorin–polypeptides were prepared (each attached to cysteine at the –14 position) and combined in an ~1:1:1 mixture to form a heterogeneous mixture of LH1-type complexes with increased solar coverage and nearly quantitative energy transfer from each bacteriochlorin to B875. Collectively, the results illustrate the great latitude of the biohybrid approach for the design of diverse light-harvesting systems.







Similar content being viewed by others
Abbreviations
- BChl a :
-
Bacteriochlorophyll a
- HFA:
-
Hexafluoroacetone trihydrate
- NIR:
-
Near infrared
- OGR:
-
Oregon Green
- PGly:
-
Propargyl glycine
References
Aratani N, Osuka A (2010) Synthetic strategies toward multiporphyrinic arrays. In: Kadish KM, Smith KM, Guilard R (eds) Handbook of porphyrin science, vol 1. World Scientific Publishing Co, Singapore, pp 1–132
Balaban TS, Tamiaki H, Holzwarth AR (2005) Chlorins programmed for self-assembly. Top Curr Chem 258:1–38
Biemann K (1992) Mass spectrometry of peptides and proteins. Annu Rev Biochem 61:977–1010
Bullough PA, Qian P, Hunter CN (2009) Reaction center-light-harvesting core complexes of purple bacteria. In: Hunter CN, Daldal F, Thurnauer MC, Beatty JT (eds) The purple phototropic bacteria. Springer, Dordrecht, pp 155–179
Chang MC, Callahan PM, Parkes-Loach PS, Cotton TM, Loach PA (1990) Spectroscopic characterization of the light-harvesting complex of Rhodospirillum rubrum and its structural subunit. Biochemistry 29:421–429
Chen C-Y, Sun E, Fan D, Taniguchi M, McDowell BE, Yang E, Diers DF, Bocian D, Holten D, Lindsey JS (2012) Synthesis and photophysical properties of metallobacteriochlorins. Inorg Chem 51:9443–9464
Chen Y-F, Chang CA, Lin Y-H, Tsay Y-G (2013) Determination of accurate protein monoisotopic mass with the most abundant mass measurable using high-resolution mass spectrometry. Anal Biochem 440:108–113
Chen C-Y, Bocian DF, Lindsey JS (2014) Synthesis of 24 bacteriochlorin isotopologues, each containing a symmetrical pair of 13C or 15N atoms in the inner core of the macrocycle. J Org Chem 79:1001–1016
Conroy MJ, Westerhuis WHJ, Parkes-Loach PS, Loach PA, Hunter CN, Williamson MP (2000) The solution structure of Rhodobacter sphaeroides LH1β reveals two helical domains separated by a more flexible region: structural consequences for the LH1 complex. J Mol Biol 298:83–94
da Graca Miguel M, Eidelman O, Ollivon M, Walter A (1989) Temperature dependence of the vesicle–micelle transition of egg phosphatidylcholine and octyl glucoside. Biochemistry 28:8921–8928
Davis CM, Bustamante PL, Todd JB, Parkes-Loach PS, McGlynn P, Olsen JD, McMaster L, Hunter CN, Loach PA (1997) Evaluation of structure–function relationships in the core light-harvesting complex of photosynthetic bacteria by reconstitution with mutant polypeptides. Biochemistry 36:3671–3679
Delmotte C, Delmas A (1999) Synthesis and fluorescence properties of Oregon Green 514 labeled peptides. Bioorg Med Chem Lett 9:2989–2994
Du H, Fuh RCA, Li J, Corkan LA, Lindsey JS (1998) PhotochemCAD. A computer-aided design and research tool in photochemistry and photobiology. Photochem Photobiol 68:141–142
Engelhardt H, Baumeister W, Saxton WO (1983) Electron microscopy of photosynthetic membranes containing bacteriochlorophyll b. Arch Microbiol 135:169–175
Fowler GJS, Sockalingum GD, Robert B, Hunter CN (1994) Blue shifts in bacteriochlorophyll absorbance correlate with changed hydrogen bonding patterns in light-harvesting 2 mutants of Rhodobacter sphaeroides with alterations at α-Tyr-44 and α-Tyr-45. Biochem J 299:695–700
Gabrielsen M, Gardiner AT, Cogdell RJ (2009) Peripheral complexes of purple bacteria. In: Hunter CN, Daldal F, Thurnauer MC, Beatty JT (eds) The purple phototropic bacteria. Springer, Dordrecht, pp 135–153
Harris MA, Parkes-Loach PS, Springer JW, Jiang J, Martin EC, Qian P, Jiao J, Niedzwiedzki DM, Kirmaier C, Olsen JD, Bocian DF, Holten D, Hunter CN, Lindsey JS, Loach PA (2013) Integration of multiple chromophores with native photosynthetic antennas to enhance solar energy capture and delivery. Chem Sci 4:3924–3933
Harvey PD (2003) Recent advances in free and metalated multiporphyrin assemblies and arrays; a photophysical behavior and energy transfer perspective. In: Kadish KM, Smith KM, Guilard R (eds) The porphyrin handbook, vol 18. Academic Press, San Diego, pp 63–250
Harvey PD, Stern C, Guilard R (2011) Bio-inspired molecular devices based on systems found in photosynthetic bacteria. In: Kadish KM, Smith KM, Guilard R (eds) Handbook of porphyrin science, vol 11. World Scientific Publishing Co, Singapore, pp 1–179
Hu X, Schulten K (1998) Model for the light-harvesting complex I (B875) of Rhodobacter sphaeroides. Biophys J 75:683–694
Iida K, Dewa T, Nango M (2009) Assembly of bacterial light-harvesting complexes on solid substrates. In: Hunter CN, Daldal F, Thurnauer MC, Beatty JT (eds) The purple phototropic bacteria. Springer, Dordrecht, pp 861–875
Kikuchi J, Asakura T, Loach PA, Parkes-Loach PS, Shimada K, Hunter CN, Conroy MJ, Williamson MP (1999) A light-harvesting antenna protein retains its folded conformation in the absence of protein–lipid and protein–pigment interactions. Biopolymers 49:361–372
Kobayashi M, Akiyama M, Kano H, Kise H (2006) An overview of chlorophylls and bacteriochlorophylls: biochemistry, biophysics, functions and applications. In: Grimm B, Porra RJ, Rüdiger W, Scheer H (eds) Chlorophylls and bacteriochlorophylls: biochemistry, biophysics, functions and applications. Springer, Dordrecht, pp 79–94
Koepke J, Hu X, Muenke C, Schulten K, Michel H (1996) The crystal structure of the light-harvesting complex II (B800–850) from Rhodospirillum molischianum. Structure 4:581–597
Lin HJ, Szmacinski H, Lakowicz JR (1999) Lifetime-based pH sensors: indicators for acidic environments. Anal Biochem 269:162–167
Lindsey JS, Mass O, Chen CY (2011) Tapping the near-infrared spectral region with bacteriochlorin arrays. New J Chem 35:511–516
Loach PA, Parkes-Loach PS (1995) Structure-function relationships in core light-harvesting complexes (LH1) as determined by characterization of the structural subunit and by reconstitution experiments. In: Blankenship RE, Madigan MT, Bauer CE (eds) Advances in photosynthesis: anoxygenic photosynthetic bacteria. Kluwer Academic Publishers, Dordrecht, pp 437–471
Loach PA, Parkes-Loach PS (2009) Structure-function relationships in bacterial light-harvesting complexes investigated by reconstitution techniques. In: Hunter CN, Daldal F, Thurnauer MC, Beatty JT (eds) The purple phototropic bacteria. Springer, Dordrecht, pp 181–198
Mauzerall D, Greenbaum NL (1989) The absolute size of a photosynthetic unit. Biochim Biophys Acta 974:119–140
McDermott G, Prince SM, Freer AA, Hawthornthwaite-Lawless AM, Papiz MZ, Cogdell RJ, Isaacs NW (1995) Crystal structure of an integral membrane light-harvesting complex from photosynthetic bacteria. Nature 374:517–521
Mchedlov-Petrossyan NO, Vodolazkaya NA, Gurina YA, Sun WC, Gee KR (2010) Medium effects on the prototropic equilibria of fluorescein fluoro derivatives in true and organized solution. J Phys Chem B 114:4551–4564
Meadows KA, Iida K, Kazuichi T, Recchia PA, Heller BA, Antonio B, Nango M, Loach PA (1995) Enzymatic and chemical cleavage of the core light-harvesting polypeptides of photosynthetic bacteria: determination of the minimal polypeptide size and structure required for subunit and light-harvesting complex formation. Biochemistry 34:1559–1574
Meadows KA, Parkes-Loach PS, Kehoe JW, Loach PA (1998) Reconstitution of light-harvesting complexes of photosynthetic bacteria using chemically synthesized polypeptides. 1. Minimal requirements for subunit formation. Biochemistry 37:3411–3417
Miller KR (1979) Structure of a bacterial photosynthetic membrane. Proc Natl Acad Sci USA 76:6415–6419
Olsen JD, Sockalingum GD, Robert B, Hunter CN (1994) Modification of a hydrogen bond to a bacteriochlorophyll a molecule in the light-harvesting 1 antenna of Rhodobacter sphaeroides. Proc Natl Acad Sci USA 91:7124–7128
Olsen JD, Sturgis JN, Westerhuis WHJ, Fowler GJS, Hunter CN, Robert B (1997) Site-directed modification of the ligands to the bacteriochlorophylls of the light-harvesting LH1 and LH2 complexes of Rhodobacter sphaeroides. Biochemistry 36:12625–12632
Orte A, Crovetto L, Talavera EM, Boens N, Alvarez-Pez JM (2005a) Absorption and emission study of 2′-7′-difluorofluorescein and its excited-state buffer-mediated proton exchange reactions. J Phys Chem A 109:734–747
Orte A, Bermejo R, Talavera EM, Crovetto L, Alvarez-Pez JM (2005b) 2′-7′-Difluorofluorescein excited-state proton reactions: correlation between time-resolved emission and steady-state fluorescence intensity. J Phys Chem A 109:2840–2846
Orte A, Talavera EM, Maçanita AL, Orte JC, Alvarez-Pez JM (2005c) Three-state 2′-7′-difluorofluorescein excited-state proton transfer reactions in moderately acidic and very acidic media. J Phys Chem A 109:8705–8718
Papiz MZ, Prince SM, Howard T, Cogdell RJ, Isaacs NW (2003) The structure and thermal motion of the B800–850 LH2 complex from Rps. acidophila at 2.0 Å resolution and 100 K: new structural features and functionally relevant motions. J Mol Biol 326:1523–1538
Parkes-Loach PS, Sprinkle JR, Loach PA (1988) Reconstitution of the B873 light-harvesting complex of Rhodospirillum rubrum from the separately isolated α- and β-polypeptides and bacteriochlorophyll a. Biochemistry 27:2718–2727
Parkes-Loach PS, Majeed AP, Law CJ, Loach PA (2004) Interactions stabilizing the structure of the core light-harvesting complex (LH1) of photosynthetic bacteria and its subunit (B820). Biochemistry 43:7003–7016
Qian P, Hunter CN, Bullough PA (2005) The 8.5 Å projection structure of the core RC–LH1–PufX dimer of Rhodobacter sphaeroides. J Mol Biol 349:948–960
Qian P, Papiz MZ, Jackson PJ, Brindley AA, Ng IW, Olsen JD, Dickman MJ, Bullough PA, Hunter CN (2013) Three-dimensional structure of the Rhodobacter sphaeroides RC–LH1–PufX complex: dimerization and quinone channels promoted by PufX. Biochemistry 52:7575–7585
Reddy KR, Jiang J, Krayer M, Harris MA, Springer JW, Yang E, Jiao J, Niedzwiedzki DM, Pandithavidana D, Parkes-Loach PS, Kirmaier C, Loach PA, Bocian DF, Holten D, Lindsey JS (2013) Palette of lipophilic bioconjugatable bacteriochlorins for construction of biohybrid light-harvesting architectures. Chem Sci 4:2036–2053
Roszak AW, Howard TD, Southall J, Gardiner AT, Law CJ, Isaacs NW, Cogdell RJ (2003) Crystal structure of the RC-LH1 core complex from Rhodopseudomonas palustris. Science 302:1969–1972
Rusinova E, Tretyachenko-Ladokhina V, Vele OE, Senear DF, Ross JBA (2002) Alexa and Oregon Green dyes as fluorescence anisotropy probes for measuring protein–protein and protein–nucleic acid interactions. Anal Biochem 308:18–25
Scheer H (2006) An overview of chlorophylls and bacteriochlorophylls: biochemistry, biophysics, functions and applications. In: Grimm B, Porra RJ, Rüdiger W, Scheer H (eds) Chlorophylls and bacteriochlorophylls: biochemistry, biophysics, functions and applications. Springer, Dordrecht, pp 1–26
Sener MK, Schulten K (2009) From atomic-level structure to supramolecular organization in the photosynthetic unit of purple bacteria. In: Hunter CN, Daldal F, Thurnauer MC, Beatty JT (eds) The purple phototrophic bacteria. Springer, Dordrecht, pp 275–294
Sorgen PL, Cahill SM, Krueger-Koplin RD, Krueger-Koplin ST, Schenck CC, Girvin ME (2002) Structure of the Rhodobacter sphaeroides light harvesting 1 β subunit in detergent micelles. Biochemistry 41:31–41
Springer JW, Parkes-Loach PS, Reddy KR, Krayer M, Jiao J, Lee GM, Niedzwiedzki DM, Harris MA, Kirmaier C, Bocian DF, Lindsey JS, Holten D, Loach PA (2012) Biohybrid photosynthetic antenna complexes for enhanced light-harvesting. J Am Chem Soc 134:4589–4599
Stark W, Kühlbrandt W, Wildhaber I, Wehrli E, Mühlethaler K (1984) The structure of the photoreceptor unit of Rhodopseudomonas viridis. EMBO J 3:777–783
Sturgis JN, Olsen JD, Robert B, Hunter CN (1997) Functions of conserved tryptophan residues of the core light-harvesting complex of Rhodobacter sphaeroides. Biochemistry 36:2772–2778
Sun WC, Gee KR, Klaubert DH, Haugland RP (1997) Synthesis of fluorinated fluoresceins. J Org Chem 62:6469–6475
Taniguchi M, Du H, Lindsey JS (2013) Enumeration of virtual libraries of combinatorial modular macrocyclic (bracelet, necklace) architectures and their linear counterparts. J Chem Inf Model 53:2203–2216
Taniguchi M, Henry S, Cogdell RJ, Lindsey JS (2014) Statistical considerations on the formation of circular photosynthetic light-harvesting complexes from Rhodopseudomonas palustris. Photosynth Res. doi:10.1007/s11120-014-9975-x
Todd JB, Recchia PA, Parkes-Loach PS, Olsen JD, Fowler GJS, McGlynn P, Hunter CN, Loach PA (1999) Minimal requirements for in vitro reconstitution of the structural subunit of light-harvesting complexes of photosynthetic bacteria. Photosynth Res 62:85–98
Wang Z-Y, Shimonaga M, Muraoka Y, Kobayashi M, Nozawa T (2001) Methionine oxidation and its effect on the stability of a reconstituted subunit of the light-harvesting complex from Rhodospirillum rubrum. Eur J Biochem 268:3375–3382
Wang Z, Muraoka Y, Shimonaga M, Kobayashi M, Nozawa T (2002) Selective detection and assignment of the solution NMR signals of bacteriochlorophyll a in a reconstituted subunit of a light-harvesting complex. J Am Chem Soc 124:1072–1078
Wang Z-Y, Gokan K, Kobayashi M, Nozawa T (2005) Solution structures of the core light-harvesting α and β polypeptides from Rhodospirillum rubrum: implications for the pigment–protein and protein–protein interactions. J Mol Biol 347:465–477
Yang J, Yoon MC, Yoo H, Kim P, Kim D (2012) Excitation energy transfer in multiporphyrin arrays with cyclic architectures: towards artificial light-harvesting antenna complexes. Chem Soc Rev 41:4808–4826
Acknowledgments
This research was carried out as part of the Photosynthetic Antenna Research Center (PARC), an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences under Award Number DE-SC0001035. The authors thank Dr. Klaus Schulten for providing the structural coordinates for the LH1-model used in Fig. 1b, c and Dr. Neil Hunter for Rb. sphaeroides puc 705BA.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Harris, M.A., Jiang, J., Niedzwiedzki, D.M. et al. Versatile design of biohybrid light-harvesting architectures to tune location, density, and spectral coverage of attached synthetic chromophores for enhanced energy capture. Photosynth Res 121, 35–48 (2014). https://doi.org/10.1007/s11120-014-9993-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-014-9993-8