Abstract
Purpose
Recently, it was reported that OCTN1 transporter (SLC22A4) is associated with rheumatoid arthritis (RA) and Crohn’s disease. Additionally, we reported that OCTN1 is expressed in hematopoietic cells, preferentially in erythroid cells. Accordingly, we assessed the physiological role of OCTN1 by examining the effect of knockdown of OCTN1 in blood cells using siRNA method.
Materials and Methods
Vector-based short hairpin RNA (shRNA) was used to establish K562 cell line which shows stably decreased expression of OCTN1. The characteristic of knockdown of OCTN1 in K562 cells was investigated by cell proliferation, cell differentiation, and uptake of ergothioneine that is a good substrate of OCTN1.
Results
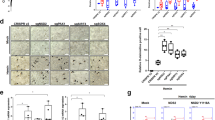
Several clones of K562 cells exhibited significantly reduced expression of OCTN1 mRNA and protein. They also showed a decreased growth rate and butyrate-dependent differentiation to erythrocytes compared with control-vector transfected cells. In addition, uptake of [3H]ergothioneine by K562 cells suggested that Na+-dependent and high-affinity transporter which is similar to the characteristics of OCTN1 is functional. Moreover, uptake of ergothioneine by K562 cells which exhibit decreased-expression of OCTN1 was decreased in comparison with wild type K562 cells.
Conclusions
It was suggested that OCTN1 is involved in the transport of physiological compounds that are important for cell proliferation and erythroid differentiation.





Similar content being viewed by others
Abbreviations
- ET:
-
ergothioneine
- FBS:
-
fetal bovine serum
- G3PDH:
-
glyceraldehyde-3-phosphate dehydrogenase
- NMG:
-
N-methyl-d-glucamine
- OCTN:
-
organic cation/carnitine transporter
- PBS:
-
phosphate-buffered saline
- PCR:
-
polymerase chain reaction
- RA:
-
rheumatoid arthritis
References
I. Tamai, H. Yabuuchi, J. Nezu, Y. Sai, A. Oku, M. Shimane, and A. Tsuji. Cloning and characterization of a novel human pH-dependent organic cation transporter, OCTN1. FEBS Lett. 419:107–111 (1997).
H. Yabuuchi, I. Tamai, J. Nezu, K. Sakamoto, A. Oku, M. Shimane, Y. Sai, and A. Tsuji. Novel membrane transporter OCTN1 mediates multispecific, bidirectional, and pH-dependent transport of organic cations. J. Pharmacol. Exp. Ther. 289:768–773 (1999).
I. Tamai, R. Ohashi, J. Nezu, H. Yabuuchi, A. Oku, M. Shimane, Y. Sai, and A. Tsuji. Molecular and functional identification of sodium ion-dependent, high affinity human carnitine transporter OCTN2. J. Biol. Chem. 273:20378–20382 (1998).
I. Tamai, R. Ohashi, J. Nezu, Y. Sai, D. Kobayashi, A. Oku, M. Shimane, and A. Tsuji. Molecular and functional characterization of organic cation/carnitine transporter family in mice. J. Biol. Chem. 275:40064–40072 (2000).
J. Nezu, I. Tamai, A. Oku, R. Ohashi, H. Yabuuchi, N. Hashimoto, H. Nikaido, Y. Sai, A. Koizumi, Y. Shoji, G. Takada, T. Matsuishi, M. Yoshino, H. Kato, T. Ohura, G. Tsujimoto, J. Hayakawa, M. Shimane, and A. Tsuji. Primary systemic carnitine deficiency is caused by mutations in a gene encoding sodium ion-dependent carnitine transporter. Nat. Genet. 21:91–94 (1999).
I. Tamai, T. Nakanishi, D. Kobayashi, K. China, Y. Kosugi, J. Nezu, Y. Sai, and A. Tsuji. Involvement of OCTN1 (SLC22A4) in pH-dependent transport of organic cations. Mol. Pharm. 1:57–66 (2004).
D. Kobayashi, S. Aizawa, T. Maeda, I. Tsuboi, H. Yabuuchi, J. Nezu, A. Tsuji, and I. Tamai. Expression of organic cation transporter OCTN1 in hematopoietic cells during erythroid differentiation. Exp. Hematol. 32:1156–1162 (2004).
S. Tokuhiro, R. Yamada, X. Chang, A. Suzuki, Y. Kochi, T. Sawada, M. Suzuki, M. Nagasaki, M. Ohtsuki, M. Ono, H. Furukawa, M. Nagashima, S. Yoshino, A. Mabuchi, A. Sekine, S. Saito, A. Takahashi, T. Tsunoda, Y. Nakamura, and M. Yamamoto. An intronic SNP in a RUNX1 binding site of SLC22A4, encoding an organic cation transporter, is associated with rheumatoid arthritis. Nat. Genet. 35:341–348 (2003).
V. Peltekova, R. F. Wintle, L. A. Rubin, C. L. Amos, Q. Huang, X. Gu, B. Newman, M. V. Oene, D. Cescon, G. Greenberg, A. M. Griffiths, P. H. St George-Hyslop, and K. A. Siminovitch. Functional variants of OCTN cation transporter genes are associated with Crohn disease. Nat. Genet. 36:471–475 (2004).
K. Yamazaki, M. Takazae, T. Tanaka, T. Ichimori, S. Saito, A. Iida, Y. Onouchi, A. Hata, and Y. Nakamura. Association analysis of SLC22A4, SLC22A5 and DLG5 in Japanese patients with Crohn disease. J. Hum. Genet. 49:664–668 (2004).
D. Gründemann, S. Harlfinger, S. Golz, A. Geerts, A. Lazar, R. Berkels, N. Jung, A. Rubbert, and E. Schomig. Discovery of ergothioneine transporter. Proc. Natl. Sci. U.S.A. 102:5256–5261 (2005).
D. Taubert, G. Grimberg, N. Jung, A. Rubbert, and E. Schomig. Function role of the 503F variant of the organic cation transporter OCTN1 in Crohn’s disease. Gut 54:1505–1506 (2005).
T. Mayumi, K. Okamoto, K. Yoshida, Y. Kawai, H. Kawano, T. Hama, and K. Tanaka. Studies on Ergothioneine. VIII. Preventive effects of ergothioneine on cadmium-induced teratogenesis. Chem. Pharm. Bull. 30:2141–2146 (1982).
P. E. Hartman. Ergothioneine as antioxidant. Methods Enzymol. 186:310–318 (1990).
Y. Shima, T. Maeda, S. Aizawa, I. Tsuboi, D. Kobayashi, R. Kato, and I. Tamai. L-arginine import via cationic amino acid transporter CAT1 is essential for both differentiation and proliferation of erythrocytes. Blood 107:1352–1356 (2006).
C. P. Perkins, V. Mar, J. R. Shutter, J. del Castillo, D. M. Danilenko, E. S. Medlock, I. L. Ponting, M. Graham, K. L. Stark, Y. Zuo, J. M. Cunningham, and R. A. Bosselman. Anemia and perinatal death result from loss of the murine ecotropic retrovirus receptor mCAT-1. Genes Dev. 11:914–925 (1997).
M. Jordan, A. Schalhorn, and F. M. Wurm. Transfecting mammalian cells: optimization of critical parameters affecting calcium-phosphate precipitate formation. Nucleic Acids Res. 24:596–601 (1996).
T. Nozawa, M. Nakajima, I. Tamai, K. Noda, J. Nezu, Y. Sai, A. Tsuji, and T. Yokoi. Genetic polymorphisms of human organic anion transporters OATP-C (SLC21A6) and OATP-B (SLC21A9): Allele frequencies in the Japanese population and functional analysis. J. Pharmacol. Exp. Ther. 302:804–813 (2002).
C. Zhang, Z. Ao, A. Seth, and S. F. Schlossman. A mitochondrial membrane protein defined by a novel monoclonal antibody is preferentially detected in apoptotic cells. J. Immunol. 157:3980–3987 (1996).
O. H. Lowry, N. J. Rosebrough, A. L. Farr, and R. J. Randall. Protein measurement with the folin phenol reagent. J. Biol. Chem. 193:265–275 (1951).
K. Yamaoka, Y. Tanigawa, T. Nakagawa, and A. Uno. pharmacokinetic analysis programa (multi) for microcomputer. J. Pharmacobiodyn. 4:879–885 (1981).
Y. Kawasaki, Y. Kato, Y. Sai, and A. Tsuji. Functional characterization of human organic cation transporter OCTN1 single nucleotide polymorphisms in the Japanese population. J. Pharm. Sci. 93:2920–2926 (2004).
T. Konishi, N. Tamaki, F. Tunemori, N. Masumitu, H. Okumura, and T. Hama. Studies on ergothioneine (II) origin of ergothioneine in the animal organs. Vitamins 46:127–129 (1972).
J.-H. Jang, O. I. Aruoma, L. S. Jen, H. Y. Chung, and Y. J. Surh. Ergothioneine resucues PC12 cells from β-amyloid-induced apoptotic death. Free Radic. Biol. Med. 36:288–299 (2004).
T. Maeda, T. Wakasawa, Y. Shima, I. Tsuboi, S. Aizawa, and I. Tamai. Biol. Pharm. Bull. 29:234–239 (2006).
K. Oishi, S. Hofmann, G. A. Diaz, B. Tartania, D. Manwani, L. Ng, R. Young, H. Vlassara, Y. A. Loannou, D. Forrest, and B. D. Gelb. Targeted disruption of Slc19a2, the gene encoding the high-affinity thiamin transporter Thtr-1, causes diabetes mellitus, sensorineural deafness and megaloblastosis in mice. Hum. Mol. Genet. 11:2951–2960 (2002).
T. Maeda, M. Hirayama, D. Kobayashi, K. Miyazawa, and I. Tamai. Mechanism of the regulation of OCTN1 transporter (SLC22A4) by rheumatoid arthritis-associated transcriptional factor RUNX1 and inflammatory cytokines. Drug Metab. Dispos. 35:394–401 (2007).
Acknowledgments
This investigation was supported in part by a Grant in Aid for Scientific Research from The Ministry of Education, Culture, Sports, Science, and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakamura, T., Sugiura, S., Kobayashi, D. et al. Decreased Proliferation and Erythroid Differentiation of K562 Cells by siRNA-induced Depression of OCTN1 (SLC22A4) Transporter Gene. Pharm Res 24, 1628–1635 (2007). https://doi.org/10.1007/s11095-007-9290-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-007-9290-8