Abstract
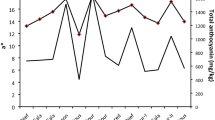
Phenolic compounds are generally believed to be key components of the oxidative defenses of plants against pathogens and herbivores. However, phenolic oxidation in the gut fluids of insect herbivores has rarely been demonstrated, and some phenolics could act as antioxidants rather than prooxidants. We compared the overall activities of the phenolic compounds in red oak (Quercus rubra) and sugar maple (Acer saccharum) leaves in the midgut fluids of two caterpillar species, Malacosoma disstria (phenolic-sensitive) and Orgyia leucostigma (phenolic-tolerant). Three hypotheses were examined: (1) ingested sugar maple leaves produce higher levels of semiquinone radicals (from phenolic oxidation) in caterpillar midgut fluids than do red oak leaves; (2) O. leucostigma maintains lower levels of phenolic oxidation in its midgut fluids than does M. disstria; and (3) phenolic compounds in tree leaves have overall prooxidant activities in the midgut fluids of caterpillars. Sugar maple leaves had significantly lower ascorbate:phenolic ratios than did red oak leaves, suggesting that phenolics in maple would oxidize more readily than those in oak. As expected, semiquinone radicals were at higher steady-state levels in the midgut fluids of both caterpillar species when they fed on sugar maple than on red oak, consistent with the first hypothesis. Higher semiquinone radical levels were also found in M. disstria than in O. leucostigma, consistent with the second hypothesis. Finally, semiquinone radical formation was positively associated with two markers of oxidation (protein carbonyls and total peroxides). These results suggest that the complex mixtures of phenolics in red oak and sugar maple leaves have overall prooxidant activities in the midgut fluids of M. disstria and O. leucostigma caterpillars. We conclude that the oxidative defenses of trees vary substantially between species, with those in sugar maple leaves being especially active, even in phenolic-tolerant herbivore species.





Similar content being viewed by others
References
S. Ahmad (1992) ArticleTitleBiochemical defence of pro-oxidant plant allelochemicals by herbivorous insects Biochem. Syst. Ecol. 20 269–296
H. M. Appel (1993) ArticleTitlePhenolics in ecological interactions: The importance of oxidation J. Chem. Ecol. 19 1521–1552
H. M. Appel J. C. >Schultz (1994) ArticleTitleOak tannins reduce effectiveness of Thuricide (Bacillus thuringiensis) in gypsy moth (Lepidoptera: Lymantriidae) J. Econ. Entomol. 87 1736–1742
Baker, W. L. 1972. Eastern Forest Insects. USDA Miscellaneous Publication no. 1175, Washington, DC..
R. V. Barbehenn (2003) ArticleTitleAntioxidants in grasshoppers: Higher levels defend the midgut tissues of a polyphagous species than a graminivorous species J. Chem. Ecol. 29 683–702
R. V. Barbehenn M. M. Martin (1992) ArticleTitleThe protective role of the peritrophic membrane in the tannin-tolerant larvae of Orgyia leucostigma (Lepidoptera) J. Insect Physiol. 38 973–980
R. V. Barbehenn M. M. Martin (1994) ArticleTitleTannin sensitivity in Malacosoma disstria: Roles of the peritrophic envelope and midgut oxidation J. Chem. Ecol. 20 1985–2001
R. V. Barbehenn M. M. Martin A. E. Hagerman (1996) ArticleTitleReassessment of the roles of the peritrophic envelope and hydrolysis in protecting polyphagous grasshoppers from ingested hydrolyzable tannins J. Chem. Ecol. 22 1911–1929
R. V. Barbehenn S. L. Bumgarner E. F. Roosen M. M. Martin (2001) ArticleTitleAntioxidant defenses in caterpillars: Role of the ascorbate-recycling system in the midgut lumen J. Insect Physiol. 47 349–357
R. V. Barbehenn A. C. Walker F. Uddin (2003) ArticleTitleAntioxidants in the midgut fluids of a tannin-tolerant and a tannin-sensitive caterpillar: Effects of seasonal changes in tree leaves J. Chem. Ecol. 29 1099–1116
R. V. Barbehenn U. Poopat B. Spencer (2003) ArticleTitleSemiquinone and ascorbyl radicals in the gut fluids of caterpillars measured with EPR spectrometry Insect Biochem. Mol. Biol. 33 125–130
J. L. Bi G. W. Felton (1995) ArticleTitleFoliar oxidative stress and insect herbivory: Primary compounds, secondary metabolites, and reactive oxygen species as components of induced resistance J. Chem. Ecol. 21 1511–1530
J. L. Bi J. B. Murphy G. W. Felton (1997) ArticleTitleAntinutritive and oxidative components as mechanisms of induced resistance in cotton to Helicoverpa zea J. Chem. Ecol. 23 97–117
L. Bravo (1998) ArticleTitlePolyphenols: Chemistry, dietary sources, metabolism, and nutritional significance Nutr. Rev. 56 317–333
G. Buettner (1993) ArticleTitleThe pecking order of free radicals and antioxidants: Lipid peroxidation, α-tocopherol, and ascorbate Arch. Biochem. Biophys. 300 535–543
E. Cadenas (1995) Mechanisms of oxygen activation and reactive oxygen species detoxification S. Ahmad (Eds) Oxidative Stress and Antioxidant Defenses in Biology Chapman and Hall New York 1–61
A. T. Canada E. Giannella T. D. Nguyen R. P. Mason (1990) ArticleTitleThe production of reactive oxygen species by dietary flavonols Free Radic. Biol. Med. 9 441–449
S. S. Duffey M. J. Stout (1996) ArticleTitleAntinutritive and toxic components of plant defense against insects Arch. Insect Biochem. Physiol. 32 3–37
G. W. Felton (1996) ArticleTitleNutritive quality of plant protein: Sources of variation and insect herbivore responses Arch. Insect Biochem. Physiol. 32 107–130
G. W. Felton K. K. Donato R. J. Vecchio ParticleDel S. S. Duffey (1989) ArticleTitleActivation of plant polyphenol oxidases by insect feeding damage reduces the nutritive quality of foliage J. Chem. Ecol. 15 2667–2694
G. W. Felton K. K. Donato R. M. Broadway S. S. Duffey (1992) ArticleTitleImpact of oxidized plant phenolics on the nutritional quality of dietary protein to a noctuid herbivore J. Insect Physiol. 38 277–285
G. Galati O. Sabzevari J. X. Wilson P. J. O’brien (2002) ArticleTitleProoxidant activity and cellular effects of the phenoxyl radicals of dietary flavonoids and other polyphenolics Toxicology 177 91–104
T. W. Gant R. Ramakrishna R. P. Mason G. M. Cohen (1988) ArticleTitleRedox cycling and sulphydryl arylation; their relative importance in the mechanism of quinone cytotoxicity to isolated hepatocytes Chem.-Biol. Interact. 65 157–173
H. D. Graham (1992) ArticleTitleStabilization of the Prussian blue color in the determination of polyphenols J. Agric. Food Chem. 40 801–805
A. E. Hagerman K. M. Riedl G. A. Jones K. N. Sovik N. T. Ritchard P. W. Hartzfeld T. L. Riechel (1998) ArticleTitleHigh molecular weight plant polyphenolics (tannins) as biological antioxidants J. Agric. Food Chem. 46 188–189
A. E. Hagerman R. T. Dean M. J. Davies (2003) ArticleTitleRadical chemistry of epigallocatechin gallate and its relevance to protein damage Arch. Biochem. Biophys. 414 115–120
B. Halliwell J. M. C. Gutteridge (1999) Free Radicals in Biology and Medicine Oxford University Press Oxford
J. B. Harborne (1985) Phenolics and plant defence C. F. Sumere ParticleVan P. J. Lea (Eds) The Biochemistry of Plant Phenolics Oxford University Press New York 393–408
K. Hoover K. T. Kishida L. A. Digiorgio J. Workman S. A. Alaniz B. D. Hammock S. S. Duffey (1998) ArticleTitleInhibition of baculoviral disease by plant-mediated peroxidase activity and free radical generation J. Chem. Ecol. 24 1949–2001
K. S. Johnson R. V. Barbehenn (1999) ArticleTitleOxygen levels in the gut lumens of herbivorous insects J. Insect Physiol. 46 897–903
K. S. Johnson G. W. Felton (2001) ArticleTitlePlant phenolics as dietary antioxidants for herbivorous insects: A test with genetically modified tobacco J. Chem. Ecol. 27 2579–2597
D. N. Karowe (1989) ArticleTitleDifferential effect of tannic acid on two tree-feeding Lepidoptera: Implications for theories of plant anti-herbivore chemistry Oecologia 80 507–512
R. A. Larson (1995) Antioxidant mechanisms of secondary natural products S. Ahmad (Eds) Oxidant-Induced Stress and Antioxidant Defenses in Biology Chapman and Hall NewYork 210–237
J. Lykkesfeldt S. Loft H. E. Poulsen (1995) ArticleTitleDetermination of ascorbic acid and dehydroascorbic acid in plasma by high-performance liquid chromatography with coulometric detection—Are they reliable biomarkers of oxidative stress? Anal. Biochem. 229 329–335
D. Metadiewa A. K. Jaiswal N. Cenas E. Dickancaite N. Segura-Auilar (1999) ArticleTitleQuercetin may act as a cytotoxic prooxidant after its metabolic activation to semiquinone and quinoidal product Free Radic. Biol. Med. 26 107–116
R. W. Nicol J. T. Arnason B. Helson M. M. Abou-Zaid (1997) ArticleTitleEffect of host and non-host trees on the growth and development of the forest tent caterpillar, Malacosoma disstria Hübner (Lepidoptera: Lasiocampidae) Can. Entomol. 129 995–1003
J. Nourooz-Zadeh J. Tajaddini-Sarmadi S. P. Wolff (1994) ArticleTitleMeasurement of plasma hydroperoxide concentrations by the ferrous oxidation–xylenol orange assay in conjunction with triphenylphosphine Anal. Biochem. 220 403–409
S. Ossipova V. Ossipov E. Haukioja J. Loponen K. Pihlaja (2001) ArticleTitleProanthocyanidins of mountain birch leaves: Quantification and properties Phytochem. Anal. 12 128–133
R. S. Pardini (1995) ArticleTitleToxicity of oxygen from naturally occurring redox-active pro-oxidants Arch. Insect Biochem. Physiol. 29 101–118
M. P. Price L. G. Butler (1977) ArticleTitleRapid visual estimation and spectrophotometric determination of tannin content of Sorghum grain J. Agric. Food Chem. 25 1268–1273
G. J. Quinlan J. M. C. Gutteridge (2000) Carbonyl assay for oxidative damage to proteins N. Taniguchi J. M. C. Gutteridge (Eds) Experimental Protocols for Reactive Oxygen and Nitrogen Species Oxford University Press Oxford 257–258
A. Z. Reznick L. Packer (1994) ArticleTitleOxidative damage to proteins: Spectrophotometric method for carbonyl assay Methods Enzymol. 233 357–371
Y. Sakihama M. F. Cohen S. C. Grace H. Yamasaki (2002) ArticleTitlePlant phenolic antioxidant and prooxidant activities: Phenolics-induced oxidative damage mediated by metals in plants Toxicology 177 67–80
J.-P. Salminen K. Lempa (2002) ArticleTitleEffects of hydrolyzable tannins on a herbivorous insect: Fate of individual tannins in insect digestive tract Chemocology 12 203–211
J.-P. Salminen V. Ossipov J. Loponen E. Haukioja K. Pihlaja (1999) ArticleTitleCharacterization of hydrolyzable tannins from leaves of Betula pubescens by high-performance liquid chromatography–mass spectrometry J. Chromatrogr., A 864 283–291
J.-P. Salminen T. Roslin M. Karonen J. Sinkkonen K. Pihlaja P. Pulkkinen (2004) ArticleTitleSeasonal variation in the content of hydrolysable tannins, flavonoid glycosides, and proanthocyanidins in oak leaves J. Chem. Ecol. 30 1675–1693
SAS Institute. 2000. The SAS System for Windows. Version 8e. SAS Institute, Cary, NC, USA
Stehr, W. F. and Cook, E. F. 1968. A revision of the genus Malacosoma Hübner in North America (Lepidoptera: Lasiocampidae): Systematics, biology, immatures, and parasites. Smithsonian Inst., U.S. Nat. Mus. Bull. no. 276
C. M. Stoscheck (1990) ArticleTitleIncreased uniformity in the response of the Coomassie blue G protein assay to different proteins Anal. Biochem. 184 111–116
N. Sugihara T. Arakawa M. Ohnishi K. Furuno (1999) ArticleTitleAnti- and pro-oxidative effects of flavonoids on metal-induced lipid hydroperoxide-dependent lipid peroxidation in cultured hepatocytes loaded with α-linolenic acid Free Radic. Biol. Med. 27 1313–1323
C. B. Summers G. W. Felton (1994) ArticleTitleProoxidant effects of phenolic acids on the generalist herbivore Helicoverpa zea (Lepidoptera: Noctuiidae): Potential mode of action for phenolic compounds in plant anti-herbivore chemistry Insect Biochem. Mol. Biol. 24 943–953
R. L. Thiboldeaux R. L. Lindroth J. W. Tracy (1998) ArticleTitleEffects of juglone (5-hydroxy-1,4-naphthoquinone) on midgut morphology and glutathione status in Saturniid moth larvae Comp. Biochem. Physiol. 120 481–487
R. H. Thompson (1964) Structure and reactivity of phenolic compounds J. B. Harborne (Eds) Biochemistry of Phenolic Compounds Academic Press New York 1–32
J. A. Vinson J. Proch P. Bose (2001) ArticleTitleDetermination of quantity and quality of polyphenol antioxidants in foods and beverages Methods Enzymol. 335 103–114
J. Wang C. P. Constabel (2004) ArticleTitlePolyphenol oxidase overexpression in transgenic Populus leaves enhances resistance to forest tent caterpillar (Malacosoma disstria) herbivory Planta 220 87–96
L. Wilkinson (2000) SYSTAT: The System for Statistics SYSTAT, Inc. Evanston, IL
J. Zheng M. Cho A. D. Jones B. D. Hammock (1997) ArticleTitleEvidence of quinone metabolites of naphthalene covalently bound to sulfur nucleophiles of proteins of murine clara cells after exposure to napthalene Chem. Res. Toxicol. 10 1008–1014
Acknowledgments
We thank Michael M. Martin for suggesting revisions to our manuscript, Juha-Pekka Salminen for analyzing phenolic compounds with HPLC-DAD, Bob McCron for providing insect eggs, and David Borneman, Brian Klatt, and Marvin Pettway for permission to use trees at Kuebler-Langford Park, the Matthaei Botanical Gardens, and the University of Michigan, respectively. This work was supported by NSF grant IBN-9974583 to RVB.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barbehenn, R., Cheek, S., Gasperut, A. et al. Phenolic Compounds in Red Oak and Sugar Maple Leaves Have Prooxidant Activities in the Midgut Fluids of Malacosoma disstria and Orgyia leucostigma Caterpillars. J Chem Ecol 31, 969–988 (2005). https://doi.org/10.1007/s10886-005-4242-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-005-4242-4