Abstract
Objectives
Expression of anti-Saccharomyces cerevisiae antibodies (ASCA) identifies patients and individuals at risk for Crohn’s disease and has also been reported in 40–60% of celiac disease (CD) cases, suggesting a role of host response to enteric microbiota in the development of inflammatory lesions. In this prospective study in patients with suspicion of CD, we evaluate the frequency and association of ASCA to serological responses for other host microbial targets formally associated with Crohn’s disease, including the Pseudomonas fluorescens associated sequence I2 and a Bacteroides caccae TonB-linked outer membrane protein, OmpW.
Methods
Small bowel mucosal biopsies were taken from 242 patients with suspicion of CD, their sera were tested for antibodies to tissue transglutaminase (tTG), ASCA, I2, and OmpW. Eighty adult healthy blood donors were used as controls.
Results
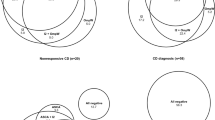
The diagnosis of CD was confirmed on biopsy in 134 cases. The occurrence of ASCA and I2 positivity was significantly higher in adult CD patients than in patients with non-CD disease. Anti-I2 levels in the sera were significantly higher in adult CD patients than in non-CD disease or the controls and anti-OmpW levels in CD and non-CD patients when compared to controls. Positive seroreactivity to OmpW seemed to increase with age. Of the CD patients, 90% were seropositive for at least one microbial antigen tested.
Conclusions
This study demonstrates a mosaic of disease-related serological responses to microbial antigens in patients with CD. Immune responses to commensal enteric bacteria may play a role in the small intestine mucosal damage in CD.



Similar content being viewed by others
References
Walker-Smith JA, Guandalini S, Schmitz J, et al. Revised criteria for diagnosis of coeliac disease. Arch Dis Child. 1990;65:909–11.
Mäki M, Holm K, Koskimies S, Hällström O, Visakorpi JK. Normal small bowel biopsy followed by coeliac disease. Arch Dis Child. 1990;65:1137–41.
Marsh MN. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity (“celiac sprue”). Gastroenterology. 1992;102:330–54.
Dietrich W, Ehnis T, Bauer M, Donner P, Volta U, Riecken EO, Schuppan D. Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat Med. 1997;3:797–801.
Troncone R, Maurano F, Rossi M, Micillo M, Greco L, Auricchio R, Salerno G, Salvatore F, Sacchetti L. IgA antibodies to tissue transglutaminase: an effective diagnostic test for celiac disease. J. Pediatr. 1999;134:166–71.
Salmi TT, Collin P, Maki M, Laurila K, Partanen J, Huhtala H, Király R, Lorand L, Reunala T, Mäki M, Kaukinen K. Immunoglobulin a autoantibodies against transglutaminase 2 in the small intestinal mucosa predict forthcoming celiac disease. Aliment Pharmacol Ther. 2006 1;24:541–52.
Sjöberg K, Eriksson S, Tenngart B, Bodil Roth E, Leffler H, Stenberg P. Factor XIII and tissue transglutaminase antibodies in coeliac and inflammatory bowel disease. Autoimmunity. 2002;35:357.
Main J, McKenzie H, Yeaman GR. Antibody to Saccharomyces cerevisiae (baker’s yeast) in patients with Crohn’s disease. BMJ. 1988;297:1105–6.
McKenzie H, Main J, Pennington CR, Kerr MA, Robson D, Pennington CR, Parratt D. Antibody to selected strains of Saccharomyces cerevisiae (baker’s and brewer’s yeast) and Candida albicans in Crohn’s disease. Gut. 1990;31:536–8.
Vasiliauskas EA, Kam LY, Karp LC, Gaiennie J, Yang H, Targan SR. Marker antibody expression stratifies Crohn’s disease into immunologically homogeneous subgroups with distinct clinical characteristics. Gut. 2000;47:487–96.
Dubinsky MC, Ofman JJ, Urman M, Targan SR, Seidman EG. Clinical utility of serodiagnostic testing in suspected pediatric inflammatory bowel disease. Am J Gastroenterol. 2001;96:758–65.
Arnott ID, Landers CJ, Nimmo EJ, Drummond HE, Smith BK, Targan SR, Satsangi J. Sero-reactivity to microbial components in Crohn’s disease is associated with disease severity and progression, but not NOD2/CARD15 genotype. Am J Gastroenterol. 2004;99:2376–84.
Iltanen S, Tervo L, Halttunen T, Wei B, Braun J, Rantala I, Honkanen T, Kronenberg M, Cheroutre H, Turovskaya O, Autio V, Ashorn M. Elevated serum anti-I2 and anti-OmpW antibody levels in children with IBD. Inflamm Bowel Dis. 2006;12:389–94.
Halfvarson J, Standaert-Vitse A, Järnerot G, Sendid B, Jouault T, Bodin L, Duhamel A, Colombel JF, Tysk C, Poulain D. Anti-Saccharomyces cerevisiae antibodies in twins with inflammatory bowel disease. Gut. 2005;54:1237–43.
Damoiseaux J, Bouten B, Linders A, Austen J, Roozendaal C, Russel MG, Forget PP, Tervaert JW. Diagnostic value of anti-Saccharomyces cerevisiae and antineutrophil cytoplasmic antibodies for inflammatory bowel disease: high prevalence in patients with celiac disease. J Clin Immunol. 2002;22(5):281–8. Sep.
Granito A, Zauli D, Muratori P, Muratori L, Grassi A, Bortolotti R, Petrolini N, Veronesi L, Gionchetti P, Bianchi FB, Volta U. Anti-Saccharomyces cerevisiae and perinuclear antineutrophil cytoplasmic antibodies in coeliac disease before and after gluten-free diet. Aliment Pharmacol Ther. 2005;21:881–7.
Ahmad T, Marshall SE, Jewell D. Genetics of inflammatory bowel disease: the role of the HLA complex. World J Gastroenterol. 2006;12:3628–35.
Rioux JD, Daly MJ, Silverberg MS, Steinhart AH, McLeod RS, Griffiths AM, Green T, Brettin TS, Stone V, Bull SB, Bitton A, Williams CN, Greenberg GR, Cohen Z, Lander ES, Hudson TJ, Siminovitch KA. Genetic variation in the 5q31 cytokine gene cluster confers susceptibility to Crohn disease. Nat Genet. 2001;29:223–8. Oct.
Amundsen SS, Adamovic S, Hellqvist A, Nilsson S, Gudjónsdóttir AH, Ascher H, Ek J, Larsson K, Wahlström J, Lie BA, Sollid LM, Naluai AT: A comprehensive screen for SNP associations on chromosome region 5q31–33 in Swedish/Norwegian celiac disease families. Eur J Hum Genet. 2007;15:980–7.
Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature. 2007;447:661–78.
Sartor RB. Induction of mucosal immune responses by bacteria and bacterial components. Curr Opin Gastroenterol. 2001;17:555–61.
Landers CJ, Cohavy O, Misra R, Yang H, Lin YC, Braun J, Targan SR. Selected loss of tolerance evidenced by Crohn’s disease associated immune responses to auto- and microbial antigens. Gastroenterology. 2002;123:689–99.
Sutton CL, Kim J, Yamane A, Dalwadi H, Wei B, Landers C, Targan SR, Braun J. Identification of a novel bacterial sequence associated with Crohn’s disease. Gastroenterology. 2000;119:23–31.
Wei B, Dalwadi H, Gordon LK, Landers C, Bruckner D, Targan SR, Braun J. Molecular cloning of a Bacteroides caccae TonB-linked outer membrane protein identified by an inflammatory bowel disease marker antibody. Infect Immun. 2001;69:6044–54.
Polymeros D, Bogdanos DP, Day R, Arioli D, Vergani D, Forbes A. Does cross-reactivity between mycobacterium avium paratuberculosis and human intestinal antigens characterize Crohn’s disease? Gastroenterology. 2006;131:85–96.
Papp M, Altorjay I, Norman GL, Shums Z, Palatka K, Vitalis Z, Foldi I, Lakos G, Tumpek J, Udvardy ML, Harsfalvi J, Fischer S, Lakatos L, Kovacs A, Bene L, Molnar T, Tulassay Z, Miheller Veres G, Papp J, Lakatos PL, Hungarian IBD Study Group. Seroreactivity to microbial components in Crohn’s disease is associated with ileal involvement, noninflammatory disease behavior and NOD2/CARD15 genotype, but not with risk for surgery in a Hungarian cohort of IBD patients. Inflamm Bowel Dis. 2007;13:984–92.
Clancy R, Ren Z, Turton J, Pang G, Wettstein A. Molecular evidence for Mycobacterium avium subspecies paratuberculosis (MAP) in Crohn’s disease correlates with enhanced TNF-alpha secretion. Dig Liver Dis. 2007;39:445–51.
Garcia-Lafuente A, Antolin M, Guarner F, Crespo E, Salas A, Forcada P, Laguarda M, Gavaldá J, Baena JA, Vilaseca J, Malagelada JR. Incrimination of anaerobic bacteria in the induction of experimental colitis. Am J Physiol. 1997;272:G10–G15.
Ley RE, Peterson DA, Gordon JI. Ecological and evolutionary forces shaping microbial diversity in the human intestine. Cell. 2006;124:837–48.
Sulkanen S, Collin P, Laurila K, Mäki M. IgA and IgG-class antihuman umbilical cord antibody tests in adult coeliac disease. Scand J Gastroenterol. 1998;33:251–4.
Kuitunen P, Kosnai I, Savilahti E. Morphometric study of the jejunal mucosa in various childhood enteropathies with special reference to intraepithelial lymphocytes. J Pediatr Gastroenterol Nutr. 1982;1:525–31.
Korponay-Szabo IR, Laurila K, Szondy Z, Halttunen T, Szalai Z, Dahlbom I, Rantala I, Kovács JB, Fésüs L, Mäki M. Missing endomysial and reticulin binding of coeliac antibodies in transglutaminase 2 knockout tissues. Gut. 2003;52:199–204.
Reese GE, Constantinides VA, Simillis C, Darzi AW, Orchard TR, Fazio VW, Tekkis PP. Diagnostic precision of anti-Saccharomyces cerevisiae antibodies and perinuclear antineutrophil cytoplasmic antibodies in inflammatory bowel disease. Am J Gastroenterol. 2006;101:2410–22.
Giaffer MH, Clark A, Holdsworth CD. Antibodies to Saccharomyces cerevisiae in patients with Crohn’s disease and their possible pathogenic importance. Gut. 1992;33:1071–5.
Mallant-Hent RCh, Mary B, von Blomberg E, Yüksel Z, Wahab PJ, Gundy C, Meyer GA, Mulder CJ. Disappearance of anti-Saccharomyces cerevisiae antibodies in coeliac disease during a gluten-free diet. Eur J Gastroenterol Hepatol. 2006;18:75–8.
Desplat-Jego S, Johanet C, Escande A, Goetz J, Fabien N, Olsson N, Ballot E, Sarles J, Baudon JJ, Grimaud JC, Veyrac M, Chamouard P, Humbel RL. Update on Anti-Saccharomyces cerevisiae antibodies, anti-nuclear associated anti-neutrophil antibodies and antibodies to exocrine pancreas detected by indirect immunofluorescence as biomarkers in chronic inflammatory bowel diseases, Results of a multicenter study. World J Gastroenterol. 2007;13(16):2312–8. Apr 28.
Toumi D, Mankai A, Belhadj R, Ghedira-Besbes L, Jeddi M, Ghedira I. Anti-Saccharomyces cerevisiae antibodies in coeliac disease. Scand J Gastroenterol. 2007;42(7):821–6. Jul..
Peeters M, Geypens B, Claus D, Nevens H, Ghoos Y, Verbeke G, Baert F, Vermeire S, Vlietinck R, Rutgeerts P. Clustering of increased small intestinal permeability in families in Crohn’s disease. Gasteroenterolog. 1997;113:802–7.
Clemente MG, De Virgiliis S, Kang JS, Macatagney R, Musu MP, Di Pierro MR, Drago S, Congia M, Fasano A. Early effects of gliadin on enterocyte intracellular signalling involved in intestinal barrier function. Gut. 2003;52:218–23.
Drago S, El Asmar R, Di Pierro M, Grazia Clemente M, Tripathi A, Sapone A, Thakar M, Iacono G, Carroccio A, D’Agate C, Not T, Zampini L, Catassi C, Fasano A. Gliadin, zonulin and gut permeability: effects on celiac and non-celiac intestinal mucosa and intestinal cell lines. Scand J Gastroenterol. 2006;41:408–19.
Sendid B, Quinton JF, Charrier G, Goulet O, Cortot A, Grandbastien B, Poulain D, Colombel JF. Anti-Saccharomyces cerevisiae mannan antibodies in familial Crohn’s disease. Am J Gastroenterol. 1998;93:1306–10.
Sutton CL, Yang H, Li Z, Rotter JI, Targan SR, Braun J. Familial expression of anti-Saccharomyces cerevisiae mannan antibodies in affected and unaffected relative of patients with Crohn’s disease. Gut. 2000;46:58–63.
Vermeire S, Peeters M, Vlietinck R, Joossens S, Den Hond E, Bulteel V, Bossuyt X, Geypens B, Rutgeerts P. Anti-Saccharomyces cerevisiae antibodies (ASCA), phenotypes of IBD, and intestinal permeability: a study in IBD families. Inflamm Bowel Dis. 2001;7:8–15.
Kaila B, Orr K, Bernstein CN. The anti-Saccharomyces cerevisiae antibody assay in a province wide practice: accurate in identifying cases of Crohn’s disease and predicting inflammatory disease. Can J Gastroenterol. 2005;19:717–21.
van Elburg RM, Uil JJ, Mulder CJ, Heymans H. Intestinal permeability in patients with coeliac disease and relatives of patients with coeliac disease. Gut. 1993;34:354–7.
Sander GR, Cummins AG, Henshall T, Powell BC. Rapid disruption of intestinal barrier function by gliadin involves altered expression of apical junctional proteins. FEBS Lett. 2005;579:4851–5.
Ciccocioppo R, Finamore A Ara C, Di Sabatino A, Mengheri E, Corazza GR. Altered expression, localization, and phosphorylation of epithelial junctional proteins in celiac disease. Am J Clin Pathol 2006;125:502–11.
Granito A, Muratori L, Muratori P, Guidi M, Lenzi M, Bianchi FB, Volta U. Anti-Saccaharomyces cerevisiae antibodies (ASCA) in coeliac disease. Gut. 2006;55:296.
Dubinsky MC, Lin YC, Dutridge D, Picornell Y, Landers CJ, Farrior S, Wrobel I, Quiros A, Vasiliauskas EA, Grill B, Israel D, Bahar R, Christie D, Wahbeh G, Silber G, Dallazadeh S, Shah P, Thomas D, Kelts D, Hershberg RM, Elson CO, Targan SR, Taylor KD, Rotter JI, Yang H, Western Regional Pediatric IBD Research Alliance. : Serum immune responses predict rapid disease progression among children with Crohn’s disease: immune responses predict disease progression. Am J Gastroenterol. 2006;101:360–7.
Mow WS, Vasiliauskas EA, Lin YC, Fleshner PR, Papadakis KA, Taylor KD, Landers CJ, Abreu-Martin MT, Rotter JI, Yang H, Targan SR. Association of antibody responses to microbial antigens and complications of small bowel Crohn’s disease. Gastroenterology. 2004;126:414–24.
Ferrante M, Henckaerts L, Joossens M, Pierik M, Joossens S, Dotan N, Norman GL, Altstock RT, Van Steen K, Rutgeerts P, Van Assche G, Vermeire S. New serological markers in inflammatory bowel disease are associated with complicated disease behaviour. Gut. 2007;56:1394–403.
Acknowledgements
This study was supported by grants from the Paediatric Research Foundation, the Competitive Research Funding of the Pirkanmaa Hospital District, the Finnish Gastroenterology Association (MA), the Mary and Georg C Ehrnrooth Foundation and, and NIH PO1-DK 46763 (J.B.). We would like to thank Leena Ripsaluoma, Marja-Leena Koskinen, and Mikko Hurme for excellent assistance in preparing this study
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ashorn, S., Raukola, H., Välineva, T. et al. Elevated Serum Anti-Saccharomyces cerevisiae, Anti-I2 and Anti-OmpW Antibody Levels in Patients with Suspicion of Celiac Disease. J Clin Immunol 28, 486–494 (2008). https://doi.org/10.1007/s10875-008-9200-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-008-9200-9