Abstract
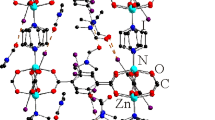
The synthesis and crystal structures of three new metal organic frameworks of type [Zn(L-2H)] n (1), {[ZnLCl2](CH3CN)0.5(DMF)0.5(H2O)0.5} n (2) and {[CdL(DMF)(NO3)2]·DMF} n (3), all based on the dipyridyl-derivatised macrocycle, dipyridyldibenzotetraaza[14]annulene (L), are reported along with the X-ray structure of the protonated metal-free ligand as its perchlorate salt, [(HL)(ClO4)] n (4). In [Zn(L-2H)] n , the zinc ion occupies the macrocyclic cavity, being bound to the N4-donor set of the macrocyclic ring in its doubly deprotonated form. Each zinc atom is also axially bound by a pyridyl moiety from an adjacent complex, resulting in formation of an infinite one-dimensional chain of the ‘herringbone’ type in which pairs of macrocyclic complexes interact via face-to-face π–π stacking interactions. In contrast, the zinc ion in {[ZnLCl2](CH3CN)0.5(DMF)0.5(H2O)0.5} n does not occupy the macrocyclic cavity but is bound to a pyridyl nitrogen from two ligands such that it acts as a bridge between macrocyclic units and results in the generation of a one-dimensional chain. Two chloro ligands also bind to each zinc centre to yield a distorted tetrahedral coordination geometry. Offset π–π stacking occurs between adjacent chains involving alternate macrocycles in each chain, giving rise to a zig-zag arrangement. Pairs of interacting chains pass through the above-mentioned chains to generate further π–π stacking to yield an overall three-dimensional structure that contains large ellipsoidal-shaped channels. In {[CdL(DMF)(NO3)2]·DMF} n the cadmium ion again does not occupy the macrocyclic cavity but acts as a bridge between macrocycles to once again afford a linear chain structure. Each cadmium is bound to two pyridyl groups (arising from different molecules of L), two nitrato ligands and one oxygen-bound dimethylformamide molecule to yield a distorted pentagonal bipyramidal coordination geometry. The protonated ligand, [(HL)(ClO4)] n , adopts a linear chain structure in which one pyridyl group is protonated and interacts intermolecularly via a hydrogen bond with the non-protonated pyridyl group of an adjacent macrocyclic unit to yield a hydrogen-bonded linear chain structure.








Similar content being viewed by others
References
Zhang, J.-P., Huang, X.-C., Chen, X.-M.: Supramolecular isomerism in coordination polymers. Chem. Soc. Rev. 38, 2385–2396 (2009)
Spokoyny, A.M., Kim, D., Sumrein, A., Mirkin, C.A.: Infinite coordination polymer nano- and microparticle structures. Chem. Soc. Rev. 38, 1218–1227 (2009)
Morsali, A., Masoomi, M.Y.: Structures and properties of mercury(II) coordination polymers. Coord. Chem. Rev. 253, 1882–1905 (2009)
Lin, W., Rieter, J.W., Taylor, K.M.L.: Modular synthesis of functional nanoscale coordination polymers. Angew. Chem. Int. Ed. 48, 650–658 (2009)
Biradha, K., Ramanan, A., Vittal, J.J.: Coordination polymers versus metal-organic frameworks. Cryst. Growth Des. 9, 2969–2970 (2009)
Murray, L.J., Dinca, M., Long, J.R.: Hydrogen storage in metal-organic frameworks. Chem. Soc. Rev. 38, 1294–1314 (2009)
Robson, R.: Design and its limitations in the construction of bi- and poly-nuclear coordination complexes and coordination polymers (aka MOFs): a personal view. Dalton Trans. 38, 5113–5131 (2008)
Tanaka, D., Kitagawa, S.: Template effects in porous coordination polymers. Chem. Mater. 20, 922–931 (2008)
Friese, V.A., Kurth, D.G.: Soluble dynamic coordination polymers as a paradigm for materials science. Coord. Chem. Rev. 252, 199–211 (2008)
Kitagawa, S., Matsuda, R.: Chemistry of coordination space of porous coordination polymers. Coord. Chem. Rev. 251, 2490–2509 (2007)
Batten, S.R., Neville, S.M., Turner, D.R.: Coordination polymers: design analysis and application. RSC Publishing, Cambridge (2009)
Czaja, A.U., Trukhan, N., Müller, U.: Chem. Soc. Rev. 38, 1284–1293 (2009)
Lee, J.-Y., Farha, O.K., Roberts, J., Scheidt, K.A., Nguyen, S.-B.T., Hupp, J.T.: Metal-organic framework materials as catalysts. Chem. Soc. Rev. 38, 1450–1459 (2009)
Biradha, K., Sarkar, M., Rajput, L.: Crystal engineering of coordination polymers using 4,4′-bipyridine as a bond between transition metal atoms. Chem. Commun. 40, 4169–4179 (2006)
Suh, M.P., Moon, H.R.: Coordination polymer open frameworks constructed of macrocyclic complexes. Adv. Inorg. Chem. 59, 39–79 (2007)
Abrahams, B.F., Hoskins, B.F., Robson, R.: A new type of infinite 3D polymeric network containing 4-connected, peripherally-linked metalloporphyrin building blocks. J. Am. Chem. Soc. 113, 3606–3609 (1991)
Abrahams, B.F., Hoskins, B.F., Michail, D.M., Robson, R.: Assembly of porphyrin building blocks into network structures with large channels. Nature 369, 727–729 (1994)
Lin, K.-J.: SMTP-1: The first functionalized metalloporphyrin molecular sieves with large channels. Angew. Chem. Int. Ed. 38, 2730 (1999)
Pan, L., Noll, B.C., Wang, X.: Self-assembly of free-base tetrapyridylporphyrin units by metal ion coordination. Chem. Comm. 157–158 (1999)
Sharma, C.V.K., Broker, G.A., Huddleston, J.G., Baldwin, J.W., Metzger, R.M., Rogers, R.D.: Design strategies for solid-state supramolecular arrays containing both mixed-metalated and freebase porphrins. J. Am. Chem. Soc. 121, 1137 (1999)
Hagrman, D., Hagrman, P.J., Zubieta, J.: Solid-state coordination chemistry: the self-assembly of microporous organic-inorganic hybrid frameworks constructed from tetrapyridylporphyrin and bimetallic oxide chains or oxide clusters. Angew. Chem. Int. Ed. 38, 3165 (1999)
Reichardt, C., Scheibelein, W.: Synthesis with aliphatic dialdehyde, XXVII. A non-template synthesis for the preparation of metal-free 1,4,8,11-tetraaza[14]annulene derivatives. Z. Naturforsch. 33, 1012–1015 (1978)
Bruker, : SMART, SAINT and XPREP. Area detector control and data integration and reduction software. Bruker Analytical X-ray Instruments Inc, Madison (1995)
Molecular Structure Corporation (1997–1998). teXsan for Windows: Single structure analysis software, MSC, 3200 Research forest drive, The Woodlands, USA
Farrugia, L.J.: WinGX-32: system of programs for solving, refining and analysing single crystal X-ray diffraction data for small molecules. J. Appl. Cryst. 32, 837 (1999)
Hall, S.R., du Boulay, D.J., Olthof-Hazekamp, R. (eds.): Xtal3.6 system. University of Western Australia, Perth (1999)
Sheldrick, G.M.: SADABS, empirical absorption correction program for area detector data. University of Göttingen, Germany (1996)
Altomare, A., Burla, M.C., Camalli, M., Cascarano, G.L., Giocavazzo, C., Guagliardi, A., Moliterni, A.G.C., Polidori, G., Spagna, S.: SIR97: a new tool for crystal structure determination and refinement. J. Appl. Crystallogr. 32, 115–119 (1999)
Sheldrick, G.M.: SHELX97 programs for crystal structure analysis. University of Göttingen, Germany (1998)
Sheldrick, G.M.: SHELXTL-Plus Graphical interface for crystal structure solution and refinement. Bruker Analytical X-ray Instrument Inc., Maddison (1998)
Bruker, : Gemini: twinning solution program suite. Bruker Analytical X-Ray Instruments Inc., Madison (1999)
Farrugia, L.J.: WinGX suite for small-molecule single-crystal crystallography. J. Appl. Cryst. 30, 565 (1999)
Acknowledgment
We thank the Australian Research Council for support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Andrew Parkin—deceased.
Y. Mulyana, C. J. Kepert, J. McMurtrie, P. Turner, G. Wei and J. G. Wilson dedicate this manuscript to Prof. Len Lindoy in celebration of his 75th birthday.
Rights and permissions
About this article
Cite this article
Mulyana, Y., Lindoy, L.F., Kepert, C.J. et al. New metal organic frameworks incorporating the ditopic macrocyclic ligand dipyridyldibenzotetraaza[14]annulene. J Incl Phenom Macrocycl Chem 71, 455–462 (2011). https://doi.org/10.1007/s10847-011-0007-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-011-0007-6