Abstract
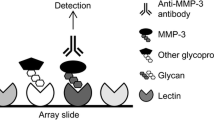
The expressions of terminal sugars in synovial and plasma fibronectins were studied in relation to rheumatoid arthritis (RA) progression defined according to the early, established and late radiological changes in the patients’ hands. The relative amounts of sialic acid and fucose were analyzed by lectin-ELISA using appropriate sialic acid-linked α2-3 (Maackia amurensis) and α2-6 (Sambucus nigra) lectins as well as fucose-linked α1-6 (Aleuria aurantia), α1-2 (Ulex europaeus), and α1-3 (Tetragonolobus purpureus). In the early RA group, the synovial fibronectin reactivities were the lowest with the all lectins used. In the established and late groups, relative sialylation and fucosylation significantly increased. However, sialylation negligibly decreased, whereas fucosylation remained at nearly the same level in the late group. Moreover, the expression of α1-6-linked fucose was found to be related to disease activity. In contrast, plasma fibronectin reactivity with lectins showed different dynamic alterations. In the early RA group, the reactivity of fibronectin with the lectins used was similar to that of healthy individuals, whereas it increased significantly in the established RA group compared with the early and normal plasma groups. In the late RA group it decreased to a level similar to that of the normal group. The lower expressions of terminal sugars in synovial fibronectin were mainly associated with the early degenerative processes of RA. In conclusion, such alterations may be applicable as a stage-specific marker for diagnosis and therapy of RA patients. The higher expression of terminal sugars in fibronectin could be associated with repair and adaptation processes in longstanding disease.

Similar content being viewed by others
Abbreviations
- RA:
-
rheumatoid arthritis
- FN:
-
fibronectin
- MAA:
-
lectin from Maackia amurensis
- SNA:
-
lectin from Sambucus nigra
- AAA:
-
lectin from Aleuria aurantia
- LTA:
-
lectin from Tetragonolobus purpureus
- UEA:
-
lectin from Ulex europaeus
References
Arnett, F.C., Edworthy, S.M., Bloch, D.A., McShane, D.J., Fries, J.F., Cooper, N.S., Healey, L.A., Kaplan, S.R., Liang, M.H., Luthra, H.S., et al.: The American rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 31, 315–324 (1988)
Audette, G.F., Vandonselaar, M., Delbaere, L.T.J.: The 2.2 Å resolution structure of the O(H) blood-group-specific lectin I from Ulex europaeus. J. Mol. Biol. 304, 423–433 (2000)
Axford, J.S.: Glycosylation and rheumatic disease. Biochim. Biophys. Acta. 1455, 219–229 (1999)
Barilla, M.L., Carsons, S.E.: Fibronectin fragments and their role in inflammatory arthritis. Semin. Arthritis Rheum. 29, 252–265 (2000)
Becker, D.J., Lowe, J.B.: Fucose, biosynthesis and biological function in mammals. Glycobiology 13, 41–53 (2003)
Carsons, S.: Enhanced expression of a peanut agglutinin reactive O linked oligosaccharide on fibronectins from the synovial fluid of patients with rheumatic disease, quantitation, domain localization, and functional significance. J. Rheumatol. 29, 896–902 (2002)
Carsons, S., Lavietes, B.B., Slomiany, A., Diamond, H.S., Berkowitz, E.: Carbohydrate heterogeneity of fibronectins. J. Clin. Invest. 80, 1342–1349 (1987)
Chen, H., Gu, D.N., Burton-Wurster, N., MacLeod, J.N.: Absence of the I-10 protein segment mediates restricted dimerization of the cartilage-specific fibronectin isoform. J. Biol. Chem. 277, 20095–20103 (2002)
Clemmensen, I., Andersen, B.: Different molecular forms of fibronectin in rheumatoid synovial fluid. Arthritis Rheum. 25, 25–31 (1982)
Durand, G., Seta, N.: Protein glycosylation and diseases: blood and urinary oligosaccharides as markers for diagnosis and therapeutic monitoring. Clin. Chem. 46, 795–805 (2000)
Ferens-Sieczkowska, M., Kossowska, B., Gancarz, R., Dudzik, D., Knas, M., Popko, J., Zwierz, K.: Fucosylation in synovial fluid as a novel clinical marker for differentiating joint diseases – a preliminary study. Clin. Exp. Rheumatol. 25, 134–137 (2007)
Fukuda, M., Levery, S.B., Hakomori, S.: Carbohydrate structure of hamster plasma fibronectin. Evidence for chemical diversity between cellular and plasma fibronectins. J. Biol. Chem. 257, 6856–6860 (1982)
Hassell, A.B., Davis, M.J., Fowler, P.D., Clarke, S., Fisher, J., Shadforth, M.F., Jones, P.W., Dawes, P.T.: The relationship between serial measures of disease activity and outcome in rheumatoid arthritis. Q. J. Med. 86, 601–607 (1993)
Helenius, A., Aebi, M.: Intracellular function of N-linked glycans. Science 291, 2364–2369 (2001)
Hirnle, L., Kątnik-Prastowska, I.: Amniotic fibronectin fragmentation and domain and sialyl- and fucosyl-glycotope expression associated with pregnancy complicated by intrauterine infection. Clin. Chem. Lab. Med. 45, 208–214 (2007)
Homandberg, G.A.: Potential regulation of cartilage metabolism in osteoarthritis by fibronectin fragments. Front. Biosci. 4, d713–d730 (1999)
Homandberg, G.A.: Cartilage damage by matrix degradation products, fibronectin fragments. Clin. Orthop. Relat. Res. 391, S100–S107 (2001)
Ingham, K. http://home.comcast.net/~kennethingham/newsite/index.htm (2006)
Kątnik, I., Jadach, J., Krotkiewski, H., Gerber, J.: Investigating the glycosylation of normal and ovarian cancer haptoglobins using digoxigenin-labeled lectins. Glycosyl. Dis. 1, 97–104 (1994)
Kątnik, I., Goodarzi, M.T., Turner, G.A.: An improved ELISA for the determination of sialyl Lewisx structures on purified glycoconjugates. Glycoconj. J. 13, 1043–1047 (1996)
Kątnik-Prastowska, I., Kratz, E.M., Faundez, R., Chełmońska-Soyta, A.: Lower expression of α2-3-sialylated fibronectin glycoform and appearance of asialo-FN glycoform associate with high concentration of fibronectin in human seminal plasma with abnormal semen parameters. Clin. Chem. Lab. Med. 44, 1119–1125 (2006)
Knibbs, R., Goldstein, I.J., Ratcliff, R.M., Shibuya, N.: Characterization of the carbohydrate binding specificity of the leukoagglutinating lectin from Maackia amurensis. Comparison with other sialic acid-specific lectins. J. Biol. Chem. 266, 83–88 (1991)
Köttgen, E., Hell, B., Müller, C., Kainer, F., Tauber, R.: Developmental changes in the glycosylation and binding properties of human fibronectins, Characterization of glycan structures and ligand binding of human fibronectins from adult plasma, cord blood and amniotic fluid. Biol. Chem. Hoppe-Seyler 370, 1285–1294 (1989)
Kriegsmann, J., Berndt, A., Hansen, T., Borsi, L., Zardi, L., Bräuer, P.K., Otto, M., Kirkpatrick, C.J., Gay, S., Kosmehl, H.: Expression of fibronectin splice variants and oncofetal glycosylated fibronectin in the synovial membranes o patients wit rheumatoid arthritis and osteoarthritis. Rheumatol. Int. 24, 25–33 (2004)
Matsuura, H., Greene, T., Hakomori, S.: An alpha-N-acetylgalactosaminylation at the threonine residue of a defined peptide sequence creates the oncofetal peptide epitope in human fibronectin. J. Biol. Chem. 264, 10472–10476 (1989)
Millard, C.H., Campbell, I.D., Pickford, A.R.: Gelatin binding to the 8F19F1 module pair of human fibronectin requires site-specific N-glycosylation. FEBS Lett. 579, 4529–4534 (2005)
Peters, J.H., Carsons, S., Yoshida, M., Ko, F., McDougall, S., Loredo, G.A., Hahn, T.J.: Electrophoretic characterization of species of fibronectin bearing sequences from the N-terminal heparin binding domain in synovial fluid samples from patients with osteoarthritis and rheumatoid arthritis. Arthritis Res. Ther. 5, R329–R339 (2003)
Przybysz, M., Borysewicz, K., Szechiñski, J., Kątnik-Prastowska, I.: Synovial fibronectin fragmentation and domain expressions in relation to rheumatoid arthritis progression. Rheumatology 46, 1071–1075 (2007)
Raza, K., Falciani, F., Curnow, S.J., Ross, E.J., Lee, C., Akbar, A.N., Lord, J.M., Gordon, C., Buckley, C.D., Salmon, M.: Early rheumatoid arthritis is characterized by a distinct and transient synovial fluid cytokine profile of T cell and stromal cell origin. Arthritis Res. Ther. 7, R784–R795 (2005)
Rees-Milton, K.J., Terry, D., Anastassiades, T.P.: Hyperglycosylation of fibronectin by TGF-β1-stimulated chondrocyte. Biochem. Biophys. Res. Commun. 317, 844–850 (2004)
Renton, P.: Imaging in rheumatoid arthritis. In: Maddison, P.J., Isenberg, D.A., Woo, P., Glass, D.N. (eds.) Oxford Textbook of Rheumatology, pp. 24–51. Oxford University Press, Oxford (1998)
Ryden, I., Påhlsson, P., Lundbland, A., Skogh, T.: Fucosylation of α1-acid glycoprotein (orosomucoid) compared with traditional biochemical markers of inflammation in recent onset rheumatoid arthritis. Clin. Chim. Acta 317, 221–229 (2002)
Sanchez-Pernaute, O., Lopez-Armada, M.J., Calvo, E., Diez-Ortego, I., Largo, R., Egido, J., Herrero-Beaumont, G.: Fibrin generated in the synovial fluid activates intimal cells from their apical surface, a sequential morphological study in antigen-induced arthritis. Rheumatology 42, 19–25 (2003)
Schauer, R.: Sialic acids, fascinating sugars in higher animals and man. Zoology 107, 49–64 (2004)
Shibuya, N., Goldstein, I.J., Broekaert, W.F., Nsimba-Lubaki, M., Peeters, B., Peumans, W.J.: The elderberry (Sambucus nigra L) bark lectin recognizes the Neu5Ac(α2-6)Gal/GalNAc sequence. J. Biol. Chem. 262, 1596–1601 (1987)
Smith, K.D., Pollacchi, A., Field, M., Watson, J.: The heterogeneity of the glycosylation of alpha-1-acid glycoprotein between the sera and synovial fluid in rheumatoid arthritis. Biomed. Chromatogr. 16, 261–266 (2002)
Tajiri, M., Yoshida, S., Wada, Y.: Differential analysis of site-specific glycans on plasma and cellular fibronectins, application of a hydrophilic affinity method for glycopeptide enrichment. Glycobiology 15, 1332–1340 (2005)
Tak, P.P.: Is early rheumatoid arthritis the same disease process as late rheumatoid arthritis. Best Practice & Research in Clinical Rheumatology 15, 17–26 (2001)
Van Dijk, W., Brinkman-van der Linden, E.C., Havenaar, E.C.: Glycosylation of α1-acid glycoprotein (orosomucoid) in health and disease, Occurrence, regulation and possible functional implications. Trends Glycosci. Glycotechnol. 10, 235–245 (1998)
Wang, X., Gu, J., Ihara, H., Miyoshi, E., Honke, K., Taniguchi, N.: Core fucosylation regulates epidermal growth factor receptor-mediated intracellular signaling. J. Biol. Chem. 281, 2572–2577 (2006)
Wierzbicka-Patynowski, I., Schwarzbauer, J.E.: The ins and outs of fibronectin matrix assembly. J. Cell Sci. 116, 3269–3276 (2003)
Zerfaoui, M., Fukuda, M., Sbarra, V., Lombardo, D., El-Battari, A.: Alpha(1,2)-fucosylation prevents sialyl Lewis x expression and E-selectin-mediated adhesion of fucosyltransferase VII-transfected cells. Eur. J. Biochem. 267, 53–61 (2000)
Zheng, M., Hakomori, S.: Soluble fibronectin interaction with cell surface and extracellular matrix is mediated by carbohydrate-to-carbohydrate interaction. Arch. Biochem. Biophys. 374, 93–99 (2000)
Yamashita, K., Kochibe, N., Ohkura, T., Ueda, I., Kobata, A.: Fractionation of l-fucose-containing oligosaccharides on immobilized Aleuria aurantia lectin. J. Biol. Chem. 260, 4688–4693 (1985)
Yan, L., Wilkins, P.P., Alvarez-Manilla, G., Do, S.I., Smith, D.F., Cummings, R.D.: Immobilized Lotus tetragonolobus agglutinin binds oligosaccharides containing the Lex determinant. Glycoconj. J. 14, 45–55 (1997)
Yasuda, T.: Cartilage destruction by matrix degradation products. Mod. Rheumatol. 16, 197–205 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Przybysz, M., Maszczak, D., Borysewicz, K. et al. Relative sialylation and fucosylation of synovial and plasma fibronectins in relation to the progression and activity of rheumatoid arthritis. Glycoconj J 24, 543–550 (2007). https://doi.org/10.1007/s10719-007-9049-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-007-9049-9