Abstract
Aims
Previous studies on the risk of lymphoma in inflammatory bowel disease (IBD) have yielded conflicting results. We aim to determine the incidence and risk factors for lymphoma in a large IBD population.
Methods
Patients with lymphoma were identified from a single-center IBD database. The standardized incidence ratio (SIR) of lymphoma was estimated using data from the Surveillance, Epidemiology and End Results (SEER) registry. Risk factors for lymphoma were determined by comparing cases with a matched IBD control group.
Results
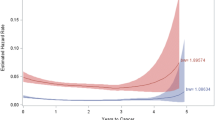
Eleven lymphomas were identified among 3,585 IBD patients during an average of 8.4 years of observation. Three patients were excluded. In the remaining eight, median age at diagnosis was 47 years and mean IBD duration was 20 years (range 7.5–45 years). The SIR for lymphoma was 1.6 [95% confidence interval (CI) 0.6-3.0], and for non-Hodgkin lymphoma (NHL), 1.5 (0.3-2.8). Three lymphoma patients (38%) received prior immunomodulators and two (25%) received biologics, versus 57% and 39% in the control group, respectively (P = 0.4). No correlation was seen with tobacco exposure, disease duration, use, or dose or duration of immunosuppressive therapy.
Conclusions
In this IBD cohort, risk of lymphoma was not increased compared with the general population. Risk of lymphoma was not associated with any demographic or therapy-related factors.
Similar content being viewed by others
Abbreviations
- IBD:
-
Inflammatory bowel disease
- NHL:
-
Non-Hodgkin lymphoma
- HL:
-
Hodgkin lymphoma
- IM:
-
Immunomodulators
- TNF-α:
-
Tumor necrosis factor-α
References
Sartor RB. Probiotic therapy of intestinal inflammation and infections. Curr Opin Gastroenterol. 2005;21:44–50.
Summers RW, Switz DM, Sessions JT Jr, et al. National Cooperative Crohn’s Disease Study: results of drug treatment. Gastroenterology. 1979;77:847–869.
Malchow H, Ewe K, Brandes JW, et al. European Cooperative Crohn’s Disease Study (ECCDS): results of drug treatment. Gastroenterology. 1984;86:249–266.
Candy S, Wright J, Gerber M, Adams G, Gerig M, Goodman R. A controlled double blind study of azathioprine in the management of Crohn’s disease. Gut. 1995;37:674–678.
Ardizzone S, Maconi G, Russo A, Imbesi V, Colombo E, Bianchi Porro G. Randomised controlled trial of azathioprine and 5-aminosalicylic acid for treatment of steroid dependent ulcerative colitis. Gut. 2006;55:47–53.
Hanauer SB, Sandborn WJ, Kornbluth A, et al. Delayed-release oral mesalamine at 4.8 g/day (800 mg tablet) for the treatment of moderately active ulcerative colitis: the ASCEND II trial. Am J Gastroenterol. 2005;100:2478–2485.
Korelitz BI, Sommers SC. Responses to drug therapy in ulcerative colitis. Evaluation by rectal biopsy and histopathological changes. Am J Gastroenterol. 1975;64:365–370.
Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347:417–429.
Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn’s disease: the ACCENT I randomised trial. Lancet. 2002;359:1541–1549.
Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn’s disease: the CHARM trial. Gastroenterology. 2007;132:52–65.
Schreiber S, Khaliq-Kareemi M, Lawrance IC, et al. Maintenance therapy with certolizumab pegol for Crohn’s disease. N Engl J Med. 2007;357:239–250.
Jarnerot G, Hertervig E, Friis-Liby I, et al. Infliximab as rescue therapy in severe to moderately severe ulcerative colitis: a randomized, placebo-controlled study. Gastroenterology. 2005;128:1805–1811.
Rutgeerts P, Sandborn WJ, Feagan BG, et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2005;353:2462–2476.
D’Haens G, Baert F, van Assche G, et al. Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn’s disease: an open randomised trial. Lancet. 2008;371:660–667.
Palli D, Trallori G, Bagnoli S, et al. Hodgkin’s disease risk is increased in patients with ulcerative colitis. Gastroenterology. 2000;119:647–653.
Arseneau KO, Stukenborg GJ, Connors AF Jr, Cominelli F. The incidence of lymphoid and myeloid malignancies among hospitalized Crohn’s disease patients. Inflamm Bowel Dis. 2001;7:106–112.
Bernstein CN, Blanchard JF, Kliewer E, Wajda A. Cancer risk in patients with inflammatory bowel disease: a population-based study. Cancer. 2001;91:854–862.
Lewis JD, Bilker WB, Brensinger C, Deren JJ, Vaughn DJ, Strom BL. Inflammatory bowel disease is not associated with an increased risk of lymphoma. Gastroenterology. 2001;121:1080–1087.
Askling J, Brandt L, Lapidus A, et al. Risk of haematopoietic cancer in patients with inflammatory bowel disease. Gut. 2005;54:617–622.
Armstrong RG, West J, Card TR. Risk of cancer in inflammatory bowel disease treated with azathioprine: a UK population-based case-control Study. Am J Gastroenterol 2010.
Colombel JF, Loftus EV Jr, Tremaine WJ, et al. The safety profile of infliximab in patients with Crohn’s disease: the Mayo clinic experience in 500 patients. Gastroenterology. 2004;126:19–31.
Ljung T, Karlen P, Schmidt D, et al. Infliximab in inflammatory bowel disease: clinical outcome in a population based cohort from Stockholm County. Gut. 2004;53:849–853.
Bongartz T, Sutton AJ, Sweeting MJ, Buchan I, Matteson EL, Montori V. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA. 2006;295:2275–2285.
Lichtenstein GR, Feagan BG, Cohen RD, et al. Serious infections and mortality in association with therapies for Crohn’s disease: TREAT registry. Clin Gastroenterol Hepatol. 2006;4:621–630.
McDonald CJ, Overhage JM, Barnes M, et al. The Indiana network for patient care: a working local health information infrastructure. An example of a working infrastructure collaboration that links data from five health systems and hundreds of millions of entries. Health Aff (Millwood). 2005;24:1214–1220.
McDonald CJ, Overhage JM, Tierney WM, et al. The Regenstrief Medical Record System: a quarter century experience. Int J Med Inform. 1999;54:225–253.
Overhage J, McDonald CJ, Suico JG. The Regenstrief medical record system 2000:Expanding the breadth and depth of a community wide EMR. Proc AMIA Symp 2000:1173.
Greenstein AJ, Gennuso R, Sachar DB, et al. Extraintestinal cancers in inflammatory bowel disease. Cancer. 1985;56:2914–2921.
Farrell RJ, Ang Y, Kileen P, et al. Increased incidence of non-Hodgkin’s lymphoma in inflammatory bowel disease patients on immunosuppressive therapy but overall risk is low. Gut. 2000;47:514–519.
Loftus EV Jr, Tremaine WJ, Habermann TM, Harmsen WS, Zinsmeister AR, Sandborn WJ. Risk of lymphoma in inflammatory bowel disease. Am J Gastroenterol. 2000;95:2308–2312.
Feinstein AR, Walter SD, Horwitz RI. An analysis of Berkson’s bias in case-control studies. J Chronic Dis. 1986;39:495–504.
Kandiel A, Fraser AG, Korelitz BI, Brensinger C, Lewis JD. Increased risk of lymphoma among inflammatory bowel disease patients treated with azathioprine and 6-mercaptopurine. Gut. 2005;54:1121–1125.
Beaugerie L, Brousse N, Bouvier AM, et al. Lymphoproliferative disorders in patients receiving thiopurines for inflammatory bowel disease: a prospective observational cohort study. Lancet. 2009;374:1617–1625.
Rosh JR, Gross T, Mamula P, Griffiths A, Hyams J. Hepatosplenic T-cell lymphoma in adolescents and young adults with Crohn’s disease: a cautionary tale? Inflamm Bowel Dis. 2007;13:1024–1030.
Peyrin-Biroulet L, Deltenre P, de Suray N, Branche J, Sandborn WJ, Colombel JF. Efficacy and safety of tumor necrosis factor antagonists in Crohn’s disease: meta-analysis of placebo-controlled trials. Clin Gastroenterol Hepatol. 2008;6:644–653.
Acknowledgments
This paper was presented in part at the Digestive Disease Week, May 2007 in Washington, DC. We would like to thank Bridget Galetti for assistance with collecting the data.
Statement of interests
Dr. Michael Chiorean has received consulting/speaker fees from Centocor, Abbott, and UCB, who had no intellectual or financial input in this study. The other authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chiorean, M.V., Pokhrel, B., Adabala, J. et al. Incidence and Risk Factors for Lymphoma in a Single-Center Inflammatory Bowel Disease Population. Dig Dis Sci 56, 1489–1495 (2011). https://doi.org/10.1007/s10620-010-1430-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-010-1430-z