Abstract
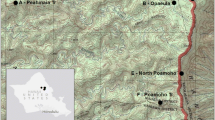
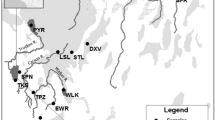
Habitat loss and isolation is pervasive in the Midwest U.S. Wetlands are experiencing particularly dramatic declines, yet there is a paucity of information on the genetic impacts of these losses to obligate wetland vertebrates. We quantified the genetic variation of extant populations of a shallow wetland specialist and evaluated potential reductions in population size (i.e. bottlenecks) using seven polymorphic microsatellite DNA markers. We analyzed 228 copperbelly water snakes (Nerodia erythrogaster neglecta), representing populations from three states. Moderate genetic differentiation exists among all three regions (F ST = 0.12, P < 0.001), with evidence for low levels of differentiation within the federally protected Ohio region (F ST = 0.025, P = 0.007), and moderate to strong differentiation within the Indiana region (F ST = 0.23, P < 0.001). Furthermore, Bayesian clustering (i.e. STRUCTURE) supports the separation of the Indiana sites, both from each other and from all other sampling sites. However, it does not support the separation of the Ohio sites from the Kentucky sites. Differentiation among sampling sites did not appear to be related to geographic distance, but rather depended on the quality of terrestrial corridors used for dispersal. Mode shifts in allele frequencies and excess heterozygosity tests were negative, while M-ratio tests were nearly all positive, indicating the likelihood of historical rather than contemporary population bottlenecks. However, potential subspecific intergradation in the Kentucky region may have artificially lowered the M-ratio, and we suggest caution when using the M-ratio approach if intergradation is suspected. Our results have conservation implications for wetland management and management of the copperbelly populations, and emphasizes the importance of protecting wetland complexes.


Similar content being viewed by others
References
Benjamini Y, Yekutieli D (2001) The control of false discovery rate under dependency. Ann Stat 29:1165–1188. doi:10.1214/aos/1013699998
Brandon RA, Blanford MJ (1995) Research concerning the current distribution, habitat requirements, and hibernation sites of the copperbelly water snake (Nerodia erythrogaster neglecta) and intergradation with the yellowbelly water snake (Nerodia erythrogaster flavigaster). Final report to the United States Fish and Wildlife Service
Buhlmann KA, Gibbons JW (1997) Imperiled aquatic reptiles of the Southeastern United States: historical review and current conservation status. In: Benz G, Collins D (eds) Aquatic fauna in peril: the southeastern perspective. Special publication 1, Southeast Aquatic Research Institute, Lenz Design & Communications, Decatur, GA, pp 201–231
Busch JD, Waser PM, DeWoody JA (2007) Recent demographic bottlenecks are not accompanied by a genetic signature in banner-tailed kangaroo rats (Dipodomys spectabilis). Mol Ecol 16:2450–2462. doi:10.1111/j.1365-294X.2007.03283.x
Bushar LM, Reinert HK, Gelbert L (1998) Genetic variation and gene flow within and between local populations of the timber rattlesnake, Crotalus horridus. Copeia 1998:411–422. doi:10.2307/1447435
Conant JR (1949) Two new races of Natrix erythrogaster. Copeia 1949:1–15. doi:10.2307/1437656
Conant JR, Collins JT (1991) A field guide to reptiles and amphibians: Eastern and Central North America. Houghton Mifflin Company, Boston
Coppola CJ (1999) Spatial ecology of southern populations of the copperbelly water snake, Nerodia erythrogaster neglecta. MS thesis, Indiana-Purdue University, Fort Wayne, IN
Cornuet JM, Luikart G (1996) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144:2001–2014
Da Silva A, Luikart G, Yoccoz NG, Cohas A, Alkine D (2006) Genetic diversity-fitness correlation revealed by microsatellite analyses in European alpine marmots (Marmota marmota). Conserv Genet 7:371–382. doi:10.1007/s10592-005-9048-y
Debinski DM, Holt RD (2000) A survey and overview of habitat fragmentation experiments. Conserv Biol 14:342–355. doi:10.1046/j.1523-1739.2000.98081.x
DeWoody YD, DeWoody JA (2005) On the estimation of genome-wide hertozygosity using molecular markers. J Hered 96:85–88. doi:10.1093/jhered/esi017
DeWoody J, Nason JD, Hipkins VD (2006) Mitigating scoring errors in microsatellite data from wild populations. Mol Ecol Notes 6:951–957. doi:10.1111/j.1471-8286.2006.01449.x
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. doi:10.1111/j.1365-294X.2005.02553.x
Ficetola GF, De Bernardi F (2004) Amphibians in a human-dominated landscape: the community structure is related to habitat features and isolation. Biol Conserv 119:219–230. doi:10.1016/j.biocon.2003.11.004
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Bio 16:265–280. doi:10.1111/j.1466-8238.2007.00287.x
Frankham R (1995) Conservation genetics. Annu Rev Genet 29:305–327. doi:10.1146/annurev.ge.29.120195.001513
Garner TWJ, Pearman PB, Gregory PT, Wischniowski TG, Hosken DJ (2004) Microsatellite markers developed from Thamnophis elegans and Thamnophis sirtalis and their utility in three species of garter snakes. Mol Ecol 4:369–371. doi:10.1111/j.1471-8286.2004.00656.x
Garza JC, Williamson EG (2001) Detection of reduction in population size using data from microsatellite loci. Mol Ecol 10:305–318. doi:10.1046/j.1365-294x.2001.01190.x
Gautschi B, Widmer A, Joshi J, Koella JC (2002) Increased frequency of scale anomalies and loss of genetic variation in serially bottlenecked populations of the dice snake (Natrix tessellata). Conserv Genet 3:235–245. doi:10.1023/A:1019924514465
Gibbons JW, Scott DE, Ryan TJ, Buhlmann KA, Tuberville TD, Metts BS et al (2000) The global decline of reptiles, Déjà vu amphibians. BioSci 50:653–666. doi:10.1641/0006-3568(2000)050[0653:TGDORD]2.0.CO;2
Gibbs HL, Prior KA, Weatherhead PJ, Johnson G (1997) Genetic structure of populations of the threatened eastern massasauga rattlesnake, Sistrurus c catenatus: evidence from microsatellite DNA markers. Mol Ecol 6:1123–1132. doi:10.1046/j.1365-294X.1997.00284.x
Glaubitz JC (2004) convert: a user-friendly program to reformat diploid genotypic data for commonly used population genetic software packages. Mol Ecol Notes 4:309–310. doi:10.1111/j.1471-8286.2004.00597.x
Goudet J (1995) FSTAT (version 1.2): a computer program to calculate F-statistics. J Hered 86:485–486
Hardy OJ, Vekemans X (2002) SPAGeDi: a versatile computer program to analyse spatial genetic structure at the individual or population levels. Mol Ecol Notes 2:618–620. doi:10.1046/j.1471-8286.2002.00305.x
Hartl DL, Clark AG (1989) Principles of population genetics, 2nd edn. Sinauer Associates, Sunderland, MA
Hyslop NL (2001) Spatial ecology and habitat use of the copperbelly water snake (Nerodia erythrogaster neglecta) in a fragmented landscape. MS thesis, Indiana-Purdue University, Fort Wayne, IN
King RB, Lawson R (2001) Patterns of population subdivision and gene flow in three sympatric natricine snakes. Copeia 2001:602–614. doi:10.1643/0045-8511(2001)001[0602:POPSAG]2.0.CO;2
Kingsbury BA, Laurent EJ (2000) Copperbelly water snake conservation agreement research: 2000 annual report. Report to the Kentucky Department of Fish and Wildlife Resources and for the Copperbelly Water Snake Conservation Agreement Technical Advisory Group
Lacy RC (1987) Loss of genetic diversity from managed populations: interacting effects of drift, mutation, immigration, selection, and population subdivision. Conserv Biol 1:143–158. doi:10.1111/j.1523-1739.1987.tb00023.x
Laurent EJ (2000) A hierarchal population study of the endangered copperbelly water snake. MS thesis. Indiana-Purdue University, Fort Wayne, IN
Laurent EJ, Kingsbury BA (2003) Habitat separation among three species of water snakes in northwestern Kentucky. J Herp 37:229–235. doi:10.1670/0022-1511(2003)037[0229:HSATSO]2.0.CO;2
Lawson R, Meier AJ, Frank PG, Moler PE (1991) Allozyme variation and systematics of the Nerodia fasciata-Nerodia clakii complex of water snakes (Serpentes: Colubridae). Copeia 1991:638–659. doi:10.2307/1446391
Lesbarreres D, Primmer CR, Lode T, Merila J (2006) The effects of 20 years of highway presence on the genetic structure of Rana dalmatina populations. Ecoscience 13:531–538. doi:10.2980/1195-6860(2006)13[531:TEOYOH]2.0.CO;2
Lewis PO, Zaykin D (2001) Genetic data analysis: computer program for the analysis of allelic data. Version 1.0 (d16c). Free program distributed by the authors over the internet from http://lewis.eeb.uconn.edu/lewishome/software.html
Lougheed SC, Gibbs HL, Prior KA, Weatherhead PJ (2000) A comparison of RAPD versus Microsatellite DNA markers in population studies of the massasauga rattlesnake. J Hered 91:458–463. doi:10.1093/jhered/91.6.458
Luikart G, Cornuet JM (1998) Empirical evaluation of a test for identifying recently bottlenecked populations from allele frequency data. Conserv Biol 12:228–237. doi:10.1046/j.1523-1739.1998.96388.x
Madsen T, Stille B, Shine R (1995) Inbreeding depression in an isolated population of adders, Vipera berus. Biol Conserv 75:113–118. doi:10.1016/0006-3207(95)00067-4
Madsen T, Olsson M, Wittzell H et al (2000) Population size and genetic diversity in sand lizards (Lacerta agilis) and adders (Vipera berus). Biol Conserv 94:257–262. doi:10.1016/S0006-3207(99)00127-5
Manier MK, Arnold SJ (2006) Ecological correlates of population genetic structure: a comparative approach using a vetebrate metacommunity. Proc R Soc Lond B Biol Sci 273:3001–3009. doi:10.1098/rspb.2006.3678
McCracken GF, Burghardt GM, Houts SE (1999) Microsatellite markers and multiple paternity in the garter snake Thamnophis sirtalis. Mol Ecol 8:1475–1479. doi:10.1046/j.1365-294x.1999.00720.x
Naeem S, Thompson LJ, Lawler SP, Lawton JH, Woodfin RM (1994) Declining biodiversity can alter performance of ecosystems. Nature 368:734–737. doi:10.1038/368734a0
Narum SR (2006) Beyond Bonferroni: less conservative analyses for conservation genetics. Conserv Genet 7:783–787. doi:10.1007/s10592-005-9056-y
Nei M (1978) Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 89:583–590
Ohta T, Kimura M (1973) The model of mutation appropriate to estimate the number of electrophoretically detectable alleles in a genetic population. Genet Resear Camb 22:201–204
Oosterhout V, Hutchinson WF, Wills DPM, Shipley P (2004) MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538. doi:10.1111/j.1471-8286.2004.00684.x
Palsboll PJ, Berube M, Allendorf FW (2007) Identification of management units using population genetic data. Trends Ecol Evol 22:11–16. doi:10.1016/j.tree.2006.09.003
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295. doi:10.1111/j.1471-8286.2005.01155.x
Piry S, Luikart G, Cornuet J-M (1999) BOTTLENECK: a computer program for detecting recent reductions in the effective population size using allele frequency data. J Hered 90:502–503. doi:10.1093/jhered/90.4.502
Prior KA, Gibbs HL, Weatherhead PJ (1997) Population genetic structure in the black rate snake: implications for management. Conserv Biol 11:1147–1158. doi:10.1046/j.1523-1739.1997.96098.x
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus geneotype data. Genetics 155:945–959
Prosser MR, Gibbs HL, Weatherhead PJ (1999) Microgeographic population genetic structure in the northern water snake, Nerodia sipedon sipedon detected using microsatellite DNA loci. Mol Ecol 8:329–333. doi:10.1046/j.1365-294X.1999.00530.x
Pruitt S, Szymanski J (1997) Endangered and threatened wildlife and plants; determination of threatened status for the northern population of the copperbelly water snake. Fed Regist 62:4183–4193
Raymond M, Rousset F (1995) GENEPOP (Version 1.2): a population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Roe JH, Georges A (2007) Heterogeneous wetland complexes, buffer zones, and travel corridors: landscape management for freshwater reptiles. Biol Conserv 135:67–76. doi:10.1016/j.biocon.2006.09.019
Roe JH, Kingsbury BA, Herbert NR (2003) Wetland and upland use patterns in semi-aquatic snakes: implications for wetland conservation. Wetlands 23:1003–1014. doi:10.1672/0277-5212(2003)023[1003:WAUUPI]2.0.CO;2
Semlitsch RD, Bodie JR (2003) Biological criteria for buffer zones around wetlands and riparian habitats for amphibians and reptiles. Conserv Biol 17:1219–1228. doi:10.1046/j.1523-1739.2003.02177.x
Spear SF, Peterson CR, Matocq MD, Storfer A (2006) Molecular evidence for historical and recent population size reductions of tiger salamanders (Ambystoma tigrinum) in Yellowstone National Park. Conserv Genet 7:605–611. doi:10.1007/s10592-005-9095-4
Ujvari B, Madsen T, Kotenko T, Olsson M, Shine R, Wittzell H (2002) Low genetic diversity threatens imminent extinction for the Hungarian meadow viper (Vipera ursinii rakasiensis). Biol Conserv 105:127–130. doi:10.1016/S0006-3207(01)00176-8
Waples RS, Gaggiotti O (2006) What is a population? An empirical evaluation of some genetic methods for identifying the number of gene pools abd their degree of connectivity. Mol Ecol 15:1419–1439. doi:10.1111/j.1365-294X.2006.02890.x
Weber JL, Wong C (1993) Mutation of human short tandem repeats. Hum Mol Genet 2:1123–1128. doi:10.1093/hmg/2.8.1123
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evol Int J Org Evol 38:1358–1370. doi:10.2307/2408641
Zar JH (1996) Biostatistical analysis, 3rd edn. Prentice-Hall, Upper Saddle River, New Jersey, USA
Acknowledgements
Financial support for this project was provided by the Indiana Academy of Science, the Center for Reptile and Amphibian Conservation and Management (Indiana-Purdue University Fort Wayne), and Purdue University. The study was conducted under Purdue University Animal Care and Use Committee approval (04–023), Kentucky Department of Fish and Wildlife Resources scientific collection permit (SC0611073), Indiana Department of Natural Resources collection permit (06-0029), Muscatatuck National Wildlife Refuge Special Use permit (31530–06005), and Federal permit (TE839779-6). We thank Mike Morton and his staff at Sloughs State Fish and Wildlife Management Area for their field assistance and tissue collection at our two Kentucky sites. Marc Webber, Susan Knowles, Theresa Daily, Cameron Young, Zack Walker, and many field volunteers and refuge interns aided in tissue collection at MNWR and Austin. John Roe, Nathan Herbert, and Yu Man Lee assisted in tissue collection from our Ohio sites. Additionally, Robert Makowsky collected the yellowbelly tissue from Alabama. Monika Zavodna, Emily Latch, and many others were very helpful in the laboratory. Joe Busch provided insightful suggestions for the bottleneck analyses. Finally, we are grateful for helpful comments on earlier drafts of this manuscript by Mark Jordan, Joe Busch, and especially Rick Howard.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marshall, J.C., Kingsbury, B.A. & Minchella, D.J. Microsatellite variation, population structure, and bottlenecks in the threatened copperbelly water snake. Conserv Genet 10, 465–476 (2009). https://doi.org/10.1007/s10592-008-9624-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-008-9624-z