Abstract
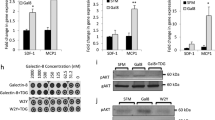
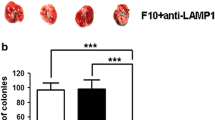
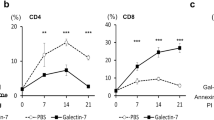
Galectin-1 is a β-galactoside-binding protein overexpressed by cancer cells. The primary roles of galectin-1 in cancer progression and metastasis are attributed to suppression of T cell immune responses, promotion of tumor angiogenesis and increased tumor cell adhesion and invasion. Using pulmonary metastasis models of murine breast (4T1) and colon (CT26) cancer, we demonstrate that targeting galectin-1 with thiodigalactoside (TDG) or shRNA galectin-1 knockdown (G1KD) results in a significant reduction in lung metastasis. Increased numbers of CD4+ helper T cells and CD8+ cytotoxic T lymphocytes were found in the peripheral blood of both TDG-treated and G1KD cell challenged mice. The levels of TUNEL+ apoptotic cancer cells and the presence of CD3+ T cells were also increased in lung metastases. Furthermore, galectin-1 was found to bind to the adhesion molecules, CD44 and CD326, which are also known as markers of breast and colon cancer stem cells, and TDG likely blocks galectin-1 binding to these molecules. The TDG-mediated inhibition of galectin-1 binding reduced 4T1 cell adhesion to the basement membrane protein laminin, Matrigel and EAhy926 endothelial cell surfaces. These findings establish possible mechanisms for the anti-metastatic effect of galectin-1 inhibition and suggest that targeting galectin-1 may represent a promising and effective anti-metastatic therapy.






Similar content being viewed by others
References
Nguyen DX, Bos PD, Massague J (2009) Metastasis: from dissemination to organ-specific colonization. Nat Rev Cancer 9(4):274–284
Chiang AC, Massague J (2008) Molecular basis of metastasis. N Engl J Med 359(26):2814–2823
Pages F, Galon J, Dieu-Nosjean MC, Tartour E, Sautes-Fridman C, Fridman WH (2009) Immune infiltration in human tumors: a prognostic factor that should not be ignored. Oncogene 29(8):1093–1102
Nicol AJ, Tokuyama H, Mattarollo SR, Hagi T, Suzuki K, Yokokawa K, Nieda M (2011) Clinical evaluation of autologous gamma delta T cell-based immunotherapy for metastatic solid tumours. Br J Cancer 105(6):778–786
Talmadge JE (2010) Immune cell infiltration of primary and metastatic lesions: mechanisms and clinical impact. Semin Cancer Biol 21(2):131–138
Yang L (2010) TGFbeta, a potent regulator of tumor microenvironment and host immune response, implication for therapy. Curr Mol Med 10(4):374–380
Prendergast GC (2008) Immune escape as a fundamental trait of cancer: focus on IDO. Oncogene 27(28):3889–3900
Rabinovich GA (2005) Galectin-1 as a potential cancer target. Br J Cancer 92(7):1188–1192
Kortylewski M, Yu H (2008) Role of Stat3 in suppressing anti-tumor immunity. Curr Opin Immunol 20(2):228–233
Watanabe MA, Oda JM, Amarante MK, Cesar Voltarelli J (2010) Regulatory T cells and breast cancer: implications for immunopathogenesis. Cancer Metastasis Rev 29(4):569–579
Iiizumi M, Mohinta S, Bandyopadhyay S, Watabe K (2007) Tumor-endothelial cell interactions: therapeutic potential. Microvasc Res 74(2–3):114–120
Miles FL, Pruitt FL, van Golen KL, Cooper CR (2008) Stepping out of the flow: capillary extravasation in cancer metastasis. Clin Exp Metastasis 25(4):305–324
Klingbeil P, Marhaba R, Jung T, Kirmse R, Ludwig T, Zoller M (2009) CD44 variant isoforms promote metastasis formation by a tumor cell-matrix cross-talk that supports adhesion and apoptosis resistance. Mol Cancer Res 7(2):168–179
Baeuerle PA, Gires O (2007) EpCAM (CD326) finding its role in cancer. Br J Cancer 96(3):417–423
Frank NY, Schatton T, Frank MH (2010) The therapeutic promise of the cancer stem cell concept. J Clin Invest 120(1):41–50
Marhaba R, Klingbeil P, Nuebel T, Nazarenko I, Buechler MW, Zoeller M (2008) CD44 and EpCAM: cancer-initiating cell markers. Curr Mol Med 8(8):784–804
Li F, Tiede B, Massague J, Kang Y (2007) Beyond tumorigenesis: cancer stem cells in metastasis. Cell Res 17(1):3–14
Barondes SH, Castronovo V, Cooper DN, Cummings RD, Drickamer K, Feizi T, Gitt MA, Hirabayashi J, Hughes C, Kasai K et al (1994) Galectins: a family of animal beta-galactoside-binding lectins. Cell 76(4):597–598
Liu FT, Rabinovich GA (2005) Galectins as modulators of tumour progression. Nat Rev Cancer 5(1):29–41
Yang RY, Rabinovich GA, Liu FT (2008) Galectins: structure, function and therapeutic potential. Expert Rev Mol Med 10:e17
Perillo NL, Pace KE, Seilhamer JJ, Baum LG (1995) Apoptosis of T cells mediated by galectin-1. Nature 378(6558):736–739
Banh A, Zhang J, Cao H, Bouley DM, Kwok S, Kong C, Giaccia AJ, Koong AC, Le QT (2011) Tumor galectin-1 mediates tumor growth and metastasis through regulation of T-cell apoptosis. Cancer Res 71(13):4423–4431
Kovacs-Solyom F, Blasko A, Fajka-Boja R, Katona RL, Vegh L, Novak J, Szebeni GJ, Krenacs L, Uher F, Tubak V, Kiss R, Monostori E (2010) Mechanism of tumor cell-induced T-cell apoptosis mediated by galectin-1. Immunol Lett 127(2):108–118
Pace KE, Hahn HP, Pang M, Nguyen JT, Baum LG (2000) CD7 delivers a pro-apoptotic signal during galectin-1-induced T cell death. J Immunol 165(5):2331–2334
Garin MI, Chu CC, Golshayan D, Cernuda-Morollon E, Wait R, Lechler RI (2007) Galectin-1: a key effector of regulation mediated by CD4+CD25+T cells. Blood 109(5):2058–2065
Tinari N, Kuwabara I, Huflejt ME, Shen PF, Iacobelli S, Liu FT (2001) Glycoprotein 90K/MAC-2BP interacts with galectin-1 and mediates galectin-1-induced cell aggregation. Int J Cancer 91(2):167–172
Hittelet A, Legendre H, Nagy N, Bronckart Y, Pector JC, Salmon I, Yeaton P, Gabius HJ, Kiss R, Camby I (2003) Upregulation of galectins-1 and -3 in human colon cancer and their role in regulating cell migration. Int J Cancer 103(3):370–379
Camby I, Belot N, Lefranc F, Sadeghi N, de Launoit Y, Kaltner H, Musette S, Darro F, Danguy A, Salmon I, Gabius HJ, Kiss R (2002) Galectin-1 modulates human glioblastoma cell migration into the brain through modifications to the actin cytoskeleton and levels of expression of small GTPases. J Neuropathol Exp Neurol 61(7):585–596
Clausse N, van den Brule F, Waltregny D, Garnier F, Castronovo V (1999) Galectin-1 expression in prostate tumor-associated capillary endothelial cells is increased by prostate carcinoma cells and modulates heterotypic cell–cell adhesion. Angiogenesis 3(4):317–325
Thijssen VL, Hulsmans S, Griffioen AW (2008) The galectin profile of the endothelium: altered expression and localization in activated and tumor endothelial cells. Am J Pathol 172(2):545–553
Thijssen VL, Postel R, Brandwijk RJ, Dings RP, Nesmelova I, Satijn S, Verhofstad N, Nakabeppu Y, Baum LG, Bakkers J, Mayo KH, Poirier F, Griffioen AW (2006) Galectin-1 is essential in tumor angiogenesis and is a target for antiangiogenesis therapy. Proc Natl Acad Sci USA 103(43):15975–15980
Ito K, Scott SA, Cutler S, Dong LF, Neuzil J, Blanchard H, Ralph SJ (2011) Thiodigalactoside inhibits murine cancers by concurrently blocking effects of galectin-1 on immune dysregulation, angiogenesis and protection against oxidative stress. Angiogenesis 14(3):293–307
Thijssen VL, Barkan B, Shoji H, Aries IM, Mathieu V, Deltour L, Hackeng TM, Kiss R, Kloog Y, Poirier F, Griffioen AW (2010) Tumor cells secrete galectin-1 to enhance endothelial cell activity. Cancer Res 70(15):6216–6224
Stannard KA, Collins PM, Ito K, Sullivan EM, Scott SA, Gabutero E, Darren Grice I, Low P, Nilsson UJ, Leffler H, Blanchard H, Ralph SJ (2010) Galectin inhibitory disaccharides promote tumour immunity in a breast cancer model. Cancer Lett 299(2):95–110
Camp RL, Scheynius A, Johansson C, Pure E (1993) CD44 is necessary for optimal contact allergic responses but is not required for normal leukocyte extravasation. J Exp Med 178(2):497–507
Barrow H, Rhodes JM, Yu LG (2011) The role of galectins in colorectal cancer progression. Int J Cancer 129(1):1–8
Ohannesian DW, Lotan D, Lotan R (1994) Concomitant increases in galectin-1 and its glycoconjugate ligands (carcinoembryonic antigen, lamp-1, and lamp-2) in cultured human colon carcinoma cells by sodium butyrate. Cancer Res 54(22):5992–6000
Baum LG, Pang M, Perillo NL, Wu T, Delegeane A, Uittenbogaart CH, Fukuda M, Seilhamer JJ (1995) Human thymic epithelial cells express an endogenous lectin, galectin-1, which binds to core 2 O-glycans on thymocytes and T lymphoblastoid cells. J Exp Med 181(3):877–887
Motran CC, Molinder KM, Liu SD, Poirier F, Miceli MC (2008) Galectin-1 functions as a Th2 cytokine that selectively induces Th1 apoptosis and promotes Th2 function. Eur J Immunol 38(11):3015–3027
Chung CD, Patel VP, Moran M, Lewis LA, Miceli MC (2000) Galectin-1 induces partial TCR zeta-chain phosphorylation and antagonizes processive TCR signal transduction. J Immunol 165(7):3722–3729
Delaine T, Cumpstey I, Ingrassia L, Le Mercier M, Okechukwu P, Leffler H, Kiss R, Nilsson UJ (2008) Galectin-inhibitory thiodigalactoside ester derivatives have antimigratory effects in cultured lung and prostate cancer cells. J Med Chem 51(24):8109–8114
de Visser KE, Eichten A, Coussens LM (2006) Paradoxical roles of the immune system during cancer development. Nat Rev Cancer 6(1):24–37
Pages F, Berger A, Camus M, Sanchez-Cabo F, Costes A, Molidor R, Mlecnik B, Kirilovsky A, Nilsson M, Damotte D, Meatchi T, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Galon J (2005) Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med 353(25):2654–2666
Harrington LE, Galvan M, Baum LG, Altman JD, Ahmed R (2000) Differentiating between memory and effector CD8 T cells by altered expression of cell surface O-glycans. J Exp Med 191(7):1241–1246
Galvan M, Tsuboi S, Fukuda M, Baum LG (2000) Expression of a specific glycosyltransferase enzyme regulates T cell death mediated by galectin-1. J Biol Chem 275(22):16730–16737
Rambaruth ND, Dwek MV (2011) Cell surface glycan-lectin interactions in tumor metastasis. Acta Histochem 113(6):591–600
Fidler IJ, Talmadge JE (1986) Evidence that intravenously derived murine pulmonary melanoma metastases can originate from the expansion of a single tumor cell. Cancer Res 46(10):5167–5171
Pace KE, Lee C, Stewart PL, Baum LG (1999) Restricted receptor segregation into membrane microdomains occurs on human T cells during apoptosis induced by galectin-1. J Immunol 163(7):3801–3811
Skelton TP, Zeng C, Nocks A, Stamenkovic I (1998) Glycosylation provides both stimulatory and inhibitory effects on cell surface and soluble CD44 binding to hyaluronan. J Cell Biol 140(2):431–446
Acknowledgments
The authors acknowledge Dr Robin Anderson, Peter MacCallum Cancer Centre, for providing us the 4T1.2 mCherry+ cell line. We thank Dr Helen Blanchard, the Institute for Glycomics Griffith University, for contribution to recombinant galectin-1 purification. We also thank Dr. Elwyn Gabutero, Dr Andy Wu and Miss Amanda Clark for the manuscript editing. Finally, we thank Dr Adrian Meedeniya and Dr Cameron Flegg, Griffith Imaging Facility, for technical supports in microscopy.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ito, K., Ralph, S.J. Inhibiting galectin-1 reduces murine lung metastasis with increased CD4+ and CD8+ T cells and reduced cancer cell adherence. Clin Exp Metastasis 29, 561–572 (2012). https://doi.org/10.1007/s10585-012-9471-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-012-9471-7