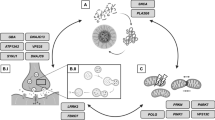
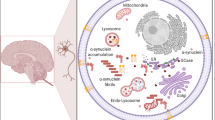
Abstract
From the first descriptions of Parkinson’s disease (PD) and Gaucher’s disease (GD) in the nineteenth century, it took more than 100 years to discover the link between the GBA gene and Parkinsonism. The observation that mutations in the GBA gene represent the most common genetic risk factor for PD so far only came into focus because of astute clinical observation of Gaucher patients and their families. In this review, we (i) outline how GBA was identified as a genetic risk factor for Parkinsonism, (ii) present clinical characteristics of GBA-associated Parkinsonism, (iii) discuss possible mechanisms of the underlying pathogenesis in GBA-associated Parkinsonism, and (iv) provide an outlook on potentially new areas of research and treatment that arise from this important discovery.


Similar content being viewed by others
References
Aarsland D, Perry R, Brown A, Larsen JP, Ballard C (2005) Neuropathology of dementia in Parkinson's disease: a prospective, community-based study. Ann Neurol 58(5):773–776
Aharon-Peretz J, Rosenbaum H, Gershoni-Baruch R (2004) Mutations in the glucocerebrosidase gene and Parkinson's disease in Ashkenazi Jews. N Engl J Med 351(19):1972–1977
Alcalay RN, Caccappolo E, Mejia-Santana H et al (2012) Cognitive performance of GBA mutation carriers with early-onset PD: the CORE-PD study. Neurology 78(18):1434–1440
Bembi B, Zambito Marsala S, Sidransky E et al (2003) Gaucher's disease with Parkinson's disease: clinical and pathological aspects. Neurology 61(1):99–101
Berg D, Lang AE, Postuma RB et al (2013) Changing the research criteria for the diagnosis of Parkinson's disease: obstacles and opportunities. Lancet Neurol 12(5):514–524
Beutler E, Gelbart T, Kuhl W, Zimran A, West C (1992) Mutations in Jewish patients with Gaucher disease. Blood 79(7):1662–1666
Bodennec J, Pelled D, Riebeling C, Trajkovic S, Futerman AH (2002) Phosphatidylcholine synthesis is elevated in neuronal models of Gaucher disease due to direct activation of CTP:phosphocholine cytidylyltransferase by glucosylceramide. FASEB J Off Publ Fed Am Soc Exp Biol 16(13):1814–1816
Brockmann K, Srulijes K, Hauser AK et al (2011) GBA-associated PD presents with nonmotor characteristics. Neurology 77(3):276–280
Brockmann K, Hilker R, Pilatus U et al (2012) GBA-associated PD. Neurodegeneration, altered membrane metabolism, and lack of energy failure. Neurology 79(3):213–220
Brockmann K, Schulte C, Hauser AK, et al (2013) SNCA: Major genetic modifier of age at onset of Parkinson's disease. Mov Disord 28(9):1217-21. doi: 10.1002/mds.25469
Chahine LM, Qiang J, Ashbridge E et al (2013) Clinical and biochemical differences in patients having Parkinson disease with vs without GBA mutations. JAMA Neurol 70(7):852–858
Chaudhuri KR, Schapira AH (2009) Non-motor symptoms of Parkinson's disease: dopaminergic pathophysiology and treatment. Lancet Neurol 8(5):464–474
Gan-Or Z, Giladi N, Rozovski U et al (2008) Genotype-phenotype correlations between GBA mutations and Parkinson disease risk and onset. Neurology 70(24):2277–2283
Gan-Or Z, Giladi N, Orr-Urtreger A (2009) Differential phenotype in Parkinson's disease patients with severe versus mild GBA mutations. Brain J Neurol 132(Pt 10):e125
Gegg ME, Burke D, Heales SJ et al (2012) Glucocerebrosidase deficiency in substantia nigra of parkinson disease brains. Ann Neurol 72(3):455–463
Goker-Alpan O, Schiffmann R, LaMarca ME, Nussbaum RL, McInerney-Leo A, Sidransky E (2004) Parkinsonism among Gaucher disease carriers. J Med Genet 41(12):937–940
Goker-Alpan O, Stubblefield BK, Giasson BI, Sidransky E (2010) Glucocerebrosidase is present in alpha-synuclein inclusions in Lewy body disorders. Acta Neuropathol 120(5):641–649
Halperin A, Elstein D, Zimran A (2006) Increased incidence of Parkinson disease among relatives of patients with Gaucher disease. Blood Cells Mol Dis 36(3):426–428
Hruska KS, LaMarca ME, Scott CR, Sidransky E (2008) Gaucher disease: mutation and polymorphism spectrum in the glucocerebrosidase gene (GBA). Hum Mutat 29(5):567–583
Lwin A, Orvisky E, Goker-Alpan O, LaMarca ME, Sidransky E (2004) Glucocerebrosidase mutations in subjects with parkinsonism. Mol Genet Metab 81(1):70–73
Machaczka M, Rucinska M, Skotnicki AB, Jurczak W (1999) Parkinson's syndrome preceding clinical manifestation of Gaucher's disease. Am J Hematol 61(3):216–217
Mazzulli JR, Xu YH, Sun Y et al (2011) Gaucher disease glucocerebrosidase and alpha-synuclein form a bidirectional pathogenic loop in synucleinopathies. Cell 146(1):37–52
McNeill A, Duran R, Hughes DA, Mehta A, Schapira AH (2012) A clinical and family history study of Parkinson's disease in heterozygous glucocerebrosidase mutation carriers. J Neurol Neurosurg Psychiatry 83(8):853–854
Nalls MA, Duran R, Lopez G et al (2013) A multicenter study of glucocerebrosidase mutations in dementia with Lewy bodies. JAMA Neurol 70(6):727–735
Neudorfer O, Giladi N, Elstein D et al (1996) Occurrence of Parkinson's syndrome in type I Gaucher disease. QJM Mon J Assoc Phys 89(9):691–694
Neumann J, Bras J, Deas E et al (2009) Glucocerebrosidase mutations in clinical and pathologically proven Parkinson's disease. Brain J Neurol 132(Pt 7):1783–1794
Nichols WC, Pankratz N, Marek DK et al (2009) Mutations in GBA are associated with familial Parkinson disease susceptibility and age at onset. Neurology 72(4):310–316
Piccinini M, Scandroglio F, Prioni S et al (2010) Deregulated sphingolipid metabolism and membrane organization in neurodegenerative disorders. Mol Neurobiol 41(2–3):314–340
Ritz B, Rhodes SL, Bordelon Y, Bronstein J (2012) alpha-Synuclein genetic variants predict faster motor symptom progression in idiopathic Parkinson disease. PLoS One 7(5):e36199
Schmid SP, Schleicher ED, Cegan A et al (2012) Cerebrospinal fluid fatty acids in glucocerebrosidase-associated Parkinson's disease. Mov Dis Off J Mov Dis Soc 27(2):288–292
Sidransky E (2004) Gaucher disease: complexity in a “simple” disorder. Mol Genet Metab 83(1–2):6–15
Sidransky E, Lopez G (2012) The link between the GBA gene and parkinsonism. Lancet Neurol 11(11):986–998
Sidransky E, Nalls MA, Aasly JO et al (2009) Multicenter analysis of glucocerebrosidase mutations in Parkinson's disease. N Engl J Med 361(17):1651–1661
Tan EK, Tong J, Fook-Chong S et al (2007) Glucocerebrosidase mutations and risk of Parkinson disease in Chinese patients. Arch Neurol 64(7):1056–1058
Tayebi N, Callahan M, Madike V et al (2001) Gaucher disease and parkinsonism: a phenotypic and genotypic characterization. Mol Genet Metab 73(4):313–321
Tayebi N, Walker J, Stubblefield B et al (2003) Gaucher disease with parkinsonian manifestations: does glucocerebrosidase deficiency contribute to a vulnerability to parkinsonism? Mol Genet Metab 79(2):104–109
Winder-Rhodes SE, Evans JR, Ban M et al (2013) Glucocerebrosidase mutations influence the natural history of Parkinson's disease in a community-based incident cohort. Brain J Neurol 136(Pt 2):392–399
Wirdefeldt K, Adami HO, Cole P, Trichopoulos D, Mandel J (2011) Epidemiology and etiology of Parkinson's disease: a review of the evidence. Eur J Epidemiol 26(Suppl 1):S1–S58
Wong K, Sidransky E, Verma A et al (2004) Neuropathology provides clues to the pathophysiology of Gaucher disease. Mol Genet Metab 82(3):192–207
Wu YR, Chen CM, Chao CY et al (2007) Glucocerebrosidase gene mutation is a risk factor for early onset of Parkinson disease among Taiwanese. J Neurol Neurosurg Psychiatry 78(9):977–979
Yap TL, Velayati A, Sidransky E, Lee JC (2013) Membrane-bound alpha-synuclein interacts with glucocerebrosidase and inhibits enzyme activity. Mol Genet Metab 108(1):56–64
Zimran A, Gelbart T, Westwood B, Grabowski GA, Beutler E (1991) High frequency of the Gaucher disease mutation at nucleotide 1226 among Ashkenazi Jews. Am J Hum Genet 49(4):855–859
Compliance with Ethics Guidelines
Dr. Brockmann has received a grant from the University of Tuebingen (TUEFF) and the Michael J. Fox Foundation as well as funding for travel of the Movement Disorders Society.
Dr. Berg has served on scientific advisory boards for Novartis, UCB/ SCHWARZ PHARMA, GlaxoSmithKline, and Teva Pharmaceutical Industries Ltd.; has received funding for travel or speaker honoraria from Boehringer Ingelheim, Lundbeck Inc., Novartis, GlaxoSmithKline, UCB/ SCHWARZ PHARMA, Merck Serono, Johnson & Johnson, and Teva Pharmaceutical Industries Ltd.; and has received research support from Janssen, Teva Pharmaceutical Industries Ltd., Solvay Pharmaceuticals, Inc./Abbott, Boehringer, UCB, Michael J Fox Foundation, BMBF, dPV (German Parkinson’s disease association), and Center of Integrative Neurosciences.
Conflict of interest
None.
Human and Animal Rights and Informed Consent
This review article provides information on published as well as unpublished data from human subjects obtained by the authors. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutiona and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study.
This review article does not contain any studies with animal subjects performed by the any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Alberto B. Burlina
Rights and permissions
About this article
Cite this article
Brockmann, K., Berg, D. The significance of GBA for Parkinson’s disease. J Inherit Metab Dis 37, 643–648 (2014). https://doi.org/10.1007/s10545-014-9714-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-014-9714-7