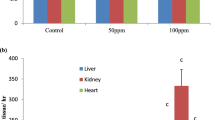
Abstract
Epidemiological evidence demonstrates positive correlation between environmental and occupational arsenic or fluoride exposure and risk to various cardio-respiratory disorders. Arsenic-exposure has been associated with atherosclerosis, hypertension, cerebrovascular diseases, ischemic heart disease, and peripheral vascular disorders, whereas Fluoride-exposure manifests cardiac irregularities and low blood pressure (BP). Present study aims to study the combined effects of these toxicants on various cardio-respiratory variables in male rats. Single intravenous (i.v.) dose of arsenic (1, 5, 10 mg/kg) or fluoride (5, 10, 20, 36.5 mg/kg) either alone or in combination were administered. Individual exposure to arsenic or fluoride led to a significant depletion of mean arterial pressure, heart rate (HR), respiration rate and neuromuscular (NM) transmission in a dose-dependent manner. These changes were accompanied by increased levels of blood reactive oxygen species (ROS) and decreased glutathione (GSH) concentrations. An increase in the blood acetyl cholinesterase (AChE) activity was observed in both arsenic or fluoride exposed rats. These changes were significantly more pronounced in arsenic-exposed animals than in fluoride. During combined exposure to arsenic (5 mg/kg) + fluoride (20 mg/kg) or arsenic (10 mg/kg) + fluoride (36.5 mg/kg) the toxic effects were more pronounced compared to individual toxicities of arsenic or fluoride alone. However, combined exposure to arsenic (5 mg/kg) + fluoride (36.5 mg/kg) resulted in antagonistic effects on variables suggestive of altered cardio-respiratory function and oxidative stress. The results from the present study suggest that arsenic or fluoride individually demonstrate cardio-respiratory failure at all doses whereas during combination exposure these toxins show variable toxicities; both synergistic and antagonistic effects depending upon the dose. Moreover, it may be concluded that arsenic and/or fluoride cardio-respiratory toxicity may be mediated via oxidative stress. However, these results are new in the discipline thus requires further exploration.







Similar content being viewed by others
Abbreviations
- As:
-
Arsenic
- F:
-
Fluoride
- NM:
-
Neuromuscular
- ROS:
-
Reactive oxygen species
- GSH:
-
Reduced glutathione
- AChE:
-
Acetyl cholinesterase
- SBP:
-
Systolic BP
- MAP:
-
Mean arterial pressure
References
Balakumar P, Kaur J (2009) Arsenic exposure and cardiovascular disorders: an overview. Cardiovasc Toxicol 9:169–176
Barbey JT, Pezzullo JC, Soignet SL (2003) Effect of arsenic trioxide on QT interval in patients with advanced malignancies. J Clin Oncol 21:3609–3615
Barbier O, Arreola-Mendoza L, Del Razo LM (2010) Molecular mechanisms of fluoride toxicity. Chem Biol Interact 188:319–333
Chen CJ, Hsueh YM, Lai MS, Shyu MP, Chen SY, Wu MM, Kuo TL, Tai TY (1995) Increased prevalence of hypertension and long-term arsenic exposure. Hypertension 25:53–65
Chevrier PJ, Brownstein S (1980) Complex fluoroanions in solution-X complexes of phosphorus and arsenic fluorides with simple anions. J Inorg Nucl Chem 42:1397–1405
Chiang C, Luk H, Wang T, Ding PY (2002) Prolongation of cardiac repolarization by arsenic trioxide. Blood 100:2249–2252
Chlubek D (2003) Fluoride and oxidative stress. Fluoride 36:217–228
Chouhan S, Flora SJS (2008) Effects of fluoride on the tissue oxidative stress and apoptosis in rats: biochemical assays supported by IR spectroscopy data. Toxicology 254:61–67
Chouhan S, Flora SJS (2010) Arsenic and fluoride: two major ground water pollutants. Indian J Exp Biol 48:666–678
Col M, Col C, Soran A, Sayli BS, Ozturk S (1999) Arsenic-related Bowen’s disease, palmar keratosis, and skin cancer. Environ Health Perspect 107:687–699
Dhar V, Bhatnagar M (2009) Physiology and toxicity of fluoride. Indian J Dent Res 20:350–355
Donmez N, Cinar A (2003) Effects of chronic fluorosis on electrocardiogram in sheep. Biol Trace Elem Res 92:115–122
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem 82:70–77
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetyl cholinesterase activity. Biochem Pharmacol 7:88–95
Farooqi A, Masuda H, Siddiqui R, Naseem M (2009) Sources of arsenic and fluoride in highly contaminated soils causing groundwater contamination in Punjab, Pakistan. Arch Environ Contam Toxicol 56:693–706
Flora SJS, Mittal M, Mishra D (2009) Co-exposure to arsenic and fluoride on oxidative stress, glutathione linked enzymes, biogenic amines and DNA damage in mouse brain. J Neurol Sci 285:198–205
Guha Mazumder DN (2003) Chronic arsenic toxicity: clinical features, epidemiology, and treatment: experience in West Bengal. J Environ Sci Health A Tox Hazard Subst Environ Eng 38:141–163
Imanishi M, Dote T, Tsuji H, Tanida E, Yamadori E, Kono K (2009) Time-dependent changes of blood parameters and fluoride kinetics in rats after acute exposure to subtoxic hydrofluoric acid. J Occup Health 51:287–293
Jollow DJ, Mitchell JR, Zamppaglione Z, Gillette JR (1974) Bromobenzene induced liver necrosis. Protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatotoxic metabolites. Pharmacology 11:151
Kant V, Srivastava AK, Verma PK, Raina R, Pankaj NK (2010) Alterations in electrocardiographic parameters after sub-acute exposure of fluoride and ameliorative action of aluminum sulphate in goats. Biol Trace Elem Res 134:188–194
Kolditz L (1967) Arsenic chemistry with halogens. In: Gutmann V (ed) Halogen chem, vol 2. Academic Press, London, p 142
Kolditz L, Nussbuk BZ (1965) Complexes of arsenic with hydroxides. Anorg Allg Chem 337:191
Lai MW, Boyer EW, Kleinman ME, Rodig NM, Ewald MB (2005) Acute arsenic poisoning in two siblings. Pediatrics 116:249–257
Lee MY, Bae ON, Chung SM, Kang KT, Lee JY, Chung JH (2002) Enhancement of platelet aggregation and thrombus formation by arsenic in drinking water: a contributing factor to cardiovascular disease. Toxicol Appl Pharmacol 179:83–88
Li XS, Zhi JL, Gao RO (1995) Effect of fluoride exposure on intelligence in children. Fluoride 28:189–192
Luoma H, Aromaa A (1983) Risk of myocardial infarction in Finnish men in relation to fluoride, magnesium and calcium concentration in drinking water. Acta Medica Scandinavica 213:171–176
Lynn S, Gurr JR, Lai HT, Jan KY (2000) NADH oxidase activation is involved in arsenite induced oxidative DNA damage in human vascular smooth muscle cells. Circulat Res 86:514–519
Martínez-Barbeito MB, Aceves BN, Teres NL, Martín RG, Verdúguez TQ, Méndez CP (2007) An unusual case of acute arsenic poisoning in which the patient made good progress. Emergencias 19:225–228
Mazej Z, Zemva B (2005) Synthesis of arsenic pentafluoride by static fluorination of As2O3 in a closed system. J Fluorine Chem 126:1432–1434
Mclvor ME (1990) Acute fluoride toxicity. Pathophysiology and management. Drug Safety 5:79–85
Mclvor ME, Cummings CE, Mower MM, Wenk RE, Lustgarten JA, Baltazar RF, Salomon J (1987) Sudden cardiac death from acute fluoride intoxication: the role of potassium. Ann Emerg Med 16:777–781
Mittal M, Flora SJS (2006) Effects of individual and combined exposure to sodium arsenite and sodium fluoride on tissue oxidative stress, arsenic and fluoride levels in male mice. Chem Biol Interact 162:128–139
Mittal M, Flora SJS (2007) Vitamin E protects oxidative stress and essential metal imbalance during concomitant exposure to arsenic and fluoride in male mice. Drug Chem Toxicol 30:263–281
Mordukhovich I, Wright RO, Amarasiriwardena C, Baja E, Baccarelli A, Suh H, Sparrow D, Vokonas P, Schwartz J (2009) Association between low-level environmental arsenic exposure and QT interval duration in a general population study. Am J Epidemiol 170:739–746
Munro NB, Watson AP, Ambrose KR, Griffin GD (1990) Treating exposure to chemical warfare agents: implications for health care providers and community emergency planning. Environ Health Perspect 89:205–215
Navas-Acien A, Sharrett AR, Silbergeld EK, Schwartz BS, Nachman KE, Burke Guallar E (2005) Arsenic exposure and cardiovascular disease: a systematic review of the epidemiologic evidence. Am J Epidemiol 162:1037
Ohnishi K, Yoshida H, Shigeno K (2000) Prolongation of the QT interval and ventricular tachycardia in patients treated with arsenic trioxide for acute promyelocytic leukemia. Ann Intern Med 133:881–885
Ohnishi K, Yoshida H, Shigeno K, Nakamura S, Fujisawa S, Naito K (2002) Arsenic trioxide therapy for relapsed or refractory Japanese patients with acute promyelocytic leukemia: need for careful electrocardiogram monitoring. Leukemia 16:617–622
Okushi I (1954) Experimental studies on the effects of sodium fluoride upon the heart muscle of rabbits. Shikoku Acta Med 5:238–245
Opie LH (2004) Cardiac output and exercise. In: Weinberg RW (ed) Heart physiology: from cell to circulation, 4th edn. Lippincott Williams and Wilkins, Philadelphia, PA, USA, p 461
Pandey A (2010) Prevalence of fluorosis in an endemic village in central India. Trop Doct 40:217–221
Raghu KG, Cherian OL (2009) Characterization of cytotoxicity induced by arsenic trioxide (a potent anti-APL drug) in rat cardiac myocytes. J Trace Elem Med Biol 23:61–68
Rahman M, Tondel M, Ahmad SA, Chowdhary IA, Faruquee MH, Axelson O (1999) Hypertension and arsenic exposure in Bangladesh. Hypertension 33:74–78
Rao MV, Tiwari H (2006) Amelioration by melatonin of chromosomal anomalies induced by arsenic and/or fluoride in human blood lymphocyte cultures. Fluoride 39:255–260
Rao JV, Begum G, Pallela R, Usman PK, Rao RN (2005) Changes in behavior and brain acetyl cholinesterase activity in mosquito fish, Gambusia affinis in response to the sub-lethal exposure to chlorpyrifos. Int J Environ Res Public Health 2:478–483
Rocha-Amador D, Navarro ME, Carrizales L, Morales R, Calderon J (2007) Decreased intelligence in children and exposure to fluoride and arsenic in drinking water. Cad Saude Publica 23:S579–S587
Saralakumari D, Ramakrishna Rao P (1991) Red cell membrane alterations in human chronic fluoride toxicity. Biochem Int 23:639–648
Shan KR, Qi XL, Long YG, Nordberg A, Guan ZZ (2004) Decreased nicotinic receptors in PC12 cells and rat brains influenced by fluoride toxicity—a mechanism relating to a damage at the level in post-transcription of the receptor genes. Toxicology 200:169–177
Shashi A, Thapar SP (2001) Histopathology of myocardial damage in experimental fluorosis in rabbits. Fluoride 34:43–50
Shi Y, Wei Y, Qu S, Wang Y, Li Y, Li R (2010) Arsenic induces apoptosis of human umbilical vein endothelial cells through mitochondrial pathways. Cardiovasc Toxicol 10:153–160
Shivarajashankara YM, Shivashankara AR, Gopalakrishna BP, Hanumanth RS (2001) Effect of fluoride intoxication on lipid peroxidation and antioxidant systems in rats. Fluoride 34:108–113
Smith AH, Steinmaus CM (2000) Arsenic in urine and drinking water. Environ Health Perspect 108:494–505
Socci DJ, Bjugstad KB, Jones HC, Pattisapu JV, Arendash GW (1999) Evidence that oxidative stress is associated with the pathophysiology of inherited hydrocephalus in the H-Tx rat model. Exp Neurol 155:109–117
Straub AC, Stolz DB, Vin H, Ross MA, Soucy NV, Klei LR, Barchowsky A (2007) Low level arsenic promotes progressive inflammatory angiogenesis and liver blood vessel remodeling in mice. Toxicol Appl Pharmacol 222:327–336
Takamori T, Miyanaga S, Kawahara H, Okushi D, Hirao M, Wakatsuki H (1956) Electrocardiographic studies of the inhabitants in high fluoride districts. Tokushima J Exp Med 3:50–53
Tortora GJ, Grabowski SR (1993) The cardiovascular system: blood vessels and hemodynamics. In: Principals of anatomy and physiology, 7th edn. Harper Collins College, pp 366–632
Tseng CH, Chong CK, Tseng CP, Hsueh YM, Chiou H, Tseng CC, Chen CJ (2003) Long-term arsenic exposure and ischemic heart disease in arseniasis-hyperendemic villages in Taiwan. Toxicol Lett 137:15–21
Vani ML, Reddy KP (2000) Effects of fluoride accumulation on some enzymes of brain and gastrocnemius muscle of mice. Fluoride 33:17–26
Wang CH, Jeng JS, Yip PK, Chen CL, Hsu LI, Hsueh YM (2002) Biological gradient between long-term arsenic exposure and carotid atherosclerosis. Circulation 105:1804–1809
Wang CH, Hsiao CK, Chen CL, Hsu LI, Chiou HY, Chen SY, Hsueh YM, Wu MM, Chen CJ (2007) A review of the epidemiologic literature on the role of environmental arsenic exposure and cardiovascular diseases. Toxicol Appl Pharmacol 222:315–326
Westervelt P, Brown RA, Adkins DR, Khoury H, Curtin P, Hurd D, Luger SM, Ma MK, Ley TJ, DiPersio JF (2001) Sudden death among patients with acute promyelocytic leukemia treated with arsenic trioxide. Blood 98:266–271
Whetzel TP, Stevenson TR, Sharman RB, Carlsen RC (1997) The effect of ischemic preconditioning on the recovery of skeletal muscle following tourniquet ischemia. Plast Reconstr Surg 100:1767–1775
Yanez L, Carrizales L, Zanatta MT, Mejia JJ, Batres L, Diaz-Barriga F (1991) Arsenic-cadmium interaction in rats: toxic effects in the heart and tissue metal shifts. Toxicology 67:227–234
Yao H, Wang G (1988a) Acute and chronic experimental studies on joint effect of fluoride and arsenic. Chin J Prevent Med 22:284–286
Yao H, Wang G (1988b) Recent situation of studies on the interactions between fluoride and other chemical substances. Chin J Endemic Dis 7:18
Yolken R, Konecny P, McCarthy P (1976) Acute fluoride poisoning. Pediatrics 58:90–93
Zierold KM, Knobeloch L, Anderson H (2004) Prevalence of chronic diseases in adults exposed to arsenic-contaminated drinking water. Am J Pubic Health 94:1936–1947
Acknowledgment
Authors thank Dr. R. Vijayaraghavan, Director of the establishment for his support and encouragement.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Flora, S.J.S., Pachauri, V., Mittal, M. et al. Interactive effect of arsenic and fluoride on cardio-respiratory disorders in male rats: possible role of reactive oxygen species. Biometals 24, 615–628 (2011). https://doi.org/10.1007/s10534-011-9412-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-011-9412-y