Abstract
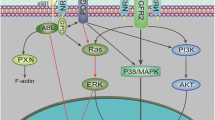
Semaphorin 4D (SEMA4D) is a member of a family of transmembrane and secreted proteins that have been shown to act through its receptor Plexin-B1 to regulate axon growth cone guidance, lymphocyte activation, and bone density. SEMA4D is also overexpressed by some malignancies and plays a role in tumor-induced angiogenesis similar to vascular endothelial growth factor (VEGF), a protein that has been targeted as part of some cancer therapies. In an attempt to examine the different effects on tumor growth and vascularity for these two pro-angiogenic factors, we previously noted that while inhibition of both VEGF and SEMA4D restricted tumor vascularity and size, vessels forming under conditions of VEGF blockade retained their association with pericytes while those arising in a background of SEMA4D/Plexin-B1 deficiency did not, an intriguing finding considering that alteration in pericyte association with endothelial cells is an emerging aspect of anti-angiogenic intervention in the treatment of cancer. Here we show through array analysis, immunoblots, migration and co-culture assays and VE-cadherin immunohistochemistry that SEMA4D production by head and neck carcinoma tumor cells induces expression of platelet-derived growth factor-B and angiopoietin-like protein 4 from endothelial cells in a Plexin-B1/Rho-dependent manner, thereby influencing proliferation and differentiation of pericytes and vascular permeability, whereas VEGF lacks these effects. These results partly explain the differences observed between SEMA4D and VEGF in pathological angiogenesis and suggest that targeting SEMA4D function along with VEGF could represent a novel anti-angiogenic therapeutic strategy for the treatment of solid tumors.





Similar content being viewed by others
Change history
26 February 2020
The Editors-in-Chief have retracted this article [1] following an investigation by the University of Maryland. The institution found that in Figure 1C, the graph showing PDGF-B does not match the original data for the 24-hour time point. The graph shows the value to be over 1000 pg/ml, but the original data have a value of 106.626. In Figure 1F, the data were entered manually to create the standard deviation bars. The data manually entered do not match the original data. When the standard deviations for the original data were calculated, the p values were no longer significant using a paired student t test. In Figure 2C, the original data do not match the published data. In Figure 4B, the images in the first lane and the fifth lane are from the same micrograph (i.e., the same set of conditions). However, the published figure claims that they are different conditions. The metadata in this figure also shows different cell lines than those noted in the article. The first and last images are labelled as Du145 shAR3 anti AR3.jpg. The second image is labelled as Du145 shAR8 anti AR8.jpg. The third image is labelled as Cos1 mARs3 mS3-2 antibody-2.jpg. The fourth image is labelled as R1 3634 bleed.jpg. Due to these errors, the Editors-in-Chief have found that the results are no longer reliable.
26 February 2020
The Editors-in-Chief have retracted this article [1] following an investigation by the University of Maryland. The institution found that in Figure 1C, the graph showing PDGF-B does not match the original data for the 24-hour time point. The graph shows the value to be over 1000 pg/ml, but the original data have a value of 106.626. In Figure 1F, the data were entered manually to create the standard deviation bars. The data manually entered do not match the original data. When the standard deviations for the original data were calculated, the p values were no longer significant using a paired student t test. In Figure 2C, the original data do not match the published data. In Figure 4B, the images in the first lane and the fifth lane are from the same micrograph (i.e., the same set of conditions). However, the published figure claims that they are different conditions. The metadata in this figure also shows different cell lines than those noted in the article. The first and last images are labelled as ���Du145 shAR3 anti AR3.jpg���. The second image is labelled as ���Du145 shAR8 anti AR8.jpg���. The third image is labelled as ���Cos1 mARs3 mS3-2 antibody-2.jpg.��� The fourth image is labelled as ���R1 3634 bleed.jpg���. Due to these errors, the Editors-in-Chief have found that the results are no longer reliable.
References
Tamagnone L, Artigiani S, Chen H, He Z, Ming GI, Song H, Chedotal A, Winberg ML, Goodman CS, Poo M, Tessier-Lavigne M, Comoglio PM (1999) Plexins are a large family of receptors for transmembrane, secreted, and GPI-anchored semaphorins in vertebrates. Cell 99:71–80
Soker S, Takashima S, Miao HQ, Neufeld G, Klagsbrun M (1998) Neuropilin-1 is expressed by endothelial and tumor cells as an isoform-specific receptor for vascular endothelial growth factor. Cell 92:735–745
Hota PK, Buck M (2012) Plexin structures are coming: opportunities for multilevel investigations of semaphorin guidance receptors, their cell signaling mechanisms, and functions. Cell Mol Life Sci 69:3765–3805
Basile JR, Barac A, Zhu T, Guan KL, Gutkind JS (2004) Class IV semaphorins promote angiogenesis by stimulating Rho-initiated pathways through plexin-B. Cancer Res 64:5212–5224
Conrotto P, Valdembri D, Corso S, Serini G, Tamagnone L, Comoglio PM, Bussolino F, Giordano S (2005) Sema4D induces angiogenesis through Met recruitment by Plexin B1. Blood 105:4321–4329
Basile JR, Castilho RM, Williams VP, Gutkind JS (2006) Semaphorin 4D provides a link between axon guidance processes and tumor-induced angiogenesis. PNAS 103:9017–9022
Sun Q, Zhou H, Binmadi NO, Basile JR (2009) Hypoxia-inducible factor-1-mediated regulation of semaphorin 4D affects tumor growth and vascularity. J Biol Chem 284:32066–32074
Zhou H, Binmadi NO, Yang YH, Proia P, Basile JR (2012) Semaphorin 4D cooperates with VEGF to promote angiogenesis and tumor progression. Angiogenesis 15:391–407
Zhou H, Yang YH, Binmadi NO, Proia P, Basile JR (2012) The hypoxia-inducible factor-responsive proteins semaphorin 4D and vascular endothelial growth factor promote tumor growth and angiogenesis in oral squamous cell carcinoma. Exp Cell Res 18:1685–1698
Pan Q, Chanthery Y, Liang WC, Stawicki S, Mak J, Rathore N, Tong RK, Kowalski J, Yee SF, Pacheco G, Ross S, Cheng ZY, Le Couter J, Plowman G, Peale F, Koch AW, Wu Y, Bagri A, Tessier-Lavigne M, Watts RJ (2007) Blocking neuropilin-1 function has an additive effect with anti-VEGF to inhibit tumor growth. Cancer Cell 11:53–67
Betsholtz C (2004) Insight into the physiological functions of PDGF through genetic studies in mice. Cytokine Growth Factor Rev 15:215–228
Bergers G, Song S (2005) The role of pericytes in blood-vessel formation and maintenance. Neuro Oncol 7:452–464
Bergers G, Song S, Meyer-Morse N, Bergsland E, Hanahan D (2003) Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J Clin Invest 111:1287–1295
Ge H, Yang G, Huang L, Motola DL, Pourbahrami T, Li C (2004) Oligomerization and regulated proteolytic processing of angiopoietin-like protein 4. J Biol Chem 279:2038–2045
Mandard S, Zandbergen F, Tan NS, Escher P, Patsouris D, Koenig W, Kleemann R, Bakker A, Veenman F, Wahli W, Muller M, Kersten S (2004) The direct peroxisome proliferator-activated receptor target fasting-induced adipose factor (FIAF/PGAR/ANGPTL4) is present in blood plasma as a truncated protein that is increased by fenofibrate treatment. J Biol Chem 279:34411–34420
Hu Z, Fan C, Livasy C, He X, Oh DS, Ewend MG, Carey LA, Subramanian S, West R, Ikpatt F, Olopade OI, van de Rijn M, Perou CM (2009) A compact VEGF signature associated with distant metastases and poor outcomes. BMC Med 7:9
Nakayama T, Hirakawa H, Shibata K, Nazneen A, Abe K, Nagayasu T, Taguchi T (2011) Expression of angiopoietin-like 4 (ANGPTL4) in human colorectal cancer: ANGPTL4 promotes venous invasion and distant metastasis. Oncol Rep 25:929–935
Le Jan S, Amy C, Cazes A, Monnot C, Lamande N, Favier J, Philippe J, Sibony M, Gasc JM, Corvol P, Germain S (2003) Angiopoietin-like 4 is a proangiogenic factor produced during ischemia and in conventional renal cell carcinoma. Am J Pathol 162:1521–1528
Ma T, Jham BC, Hu J, Friedman ER, Basile JR, Molinolo A, Sodhi A, Montaner S (2010) Viral G protein-coupled receptor up-regulates Angiopoietin-like 4 promoting angiogenesis and vascular permeability in Kaposi’s sarcoma. Proc Natl Acad Sci U S A 107:14363–14368
Huang RL, Teo Z, Chong HC, Zhu P, Tan MJ, Tan CK, Lam CR, Sng MK, Leong DT, Tan SM, Kersten S, Ding JL, Li HY, Tan NS (2011) ANGPTL4 modulates vascular junction integrity by integrin signaling and disruption of intercellular VE-cadherin and claudin-5 clusters. Blood 118:3990–4002
Minn AJ, Gupta GP, Siegel PM, Bos PD, Shu W, Giri DD, Viale A, Olshen AB, Gerald WL, Massague J (2005) Genes that mediate breast cancer metastasis to lung. Nature 436:518–524
Cardinali M, Pietraszkiewicz H, Ensley JF, Robbins KC (1995) Tyrosine phosphorylation as a marker for aberrantly regulated growth-promoting pathways in cell lines derived from head and neck malignancies. Int J Cancer 61:98–103
Dhar K, Dhar G, Majumder M, Haque I, Mehta S, Van Veldhuizen PJ, Banerjee SK, Banerjee S (2010) Tumor cell-derived PDGF-B potentiates mouse mesenchymal stem cells-pericytes transition and recruitment through an interaction with NRP-1. Mol Cancer 9:209
Darland DC, D’Amore PA (2001) TGF beta is required for the formation of capillary-like structures in three-dimensional cocultures of 10T1/2 and endothelial cells. Angiogenesis 4:11–20
Hannon GJ, Conklin DS (2004) RNA interference by short hairpin RNAs expressed in vertebrate cells. Methods Mol Biol 257:255–266
Siolas D, Lerner C, Burchard J, Ge W, Linsley PS, Paddison PJ, Hannon GJ, Cleary MA (2005) Synthetic shRNAs as potent RNAi triggers. Nat Biotechnol 23:227–231
Yang YH, Zhou H, Binmadi NO, Proia P, Basile JR (2011) Plexin-B1 activates NF-kappaB and IL-8 to promote a pro-angiogenic response in endothelial cells. PLoS ONE 6:e25826
Paddison PJ, Caudy AA, Sachidanandam R, Hannon GJ (2004) Short hairpin activated gene silencing in mammalian cells. Methods Mol Biol 265:85–100
Paddison PJ, Silva JM, Conklin DS, Schlabach M, Li M, Aruleba S, Balija V, O’Shaughnessy A, Gnoj L, Scobie K, Chang K, Westbrook T, Cleary M, Sachidanandam R, McCombie WR, Elledge SJ, Hannon GJ (2004) A resource for large-scale RNA-interference-based screens in mammals. Nature 428:427–431
Franco M, Roswall P, Cortez E, Hanahan D, Pietras K (2011) Pericytes promote endothelial cell survival through induction of autocrine VEGF-A signaling and Bcl-w expression. Blood 118:2906–2917
Amornphimoltham P, Sriuranpong V, Patel V, Benavides F, Conti CJ, Sauk J, Sausville EA, Molinolo AA, Gutkind JS (2004) Persistent activation of the Akt pathway in head and neck squamous cell carcinoma: a potential target for UCN-01. Clin Cancer Res 10:4029–4037
Perrot V, Vazquez-Prado J, Gutkind JS (2002) Plexin B regulates Rho through the guanine nucleotide exchange factors leukemia-associated Rho GEF (LARG) and PDZ-RhoGEF. J Biol Chem 277:43115–43120
Aurandt J, Vikis HG, Gutkind JS, Ahn N, Guan KL (2002) The semaphorin receptor plexin-B1 signals through a direct interaction with the Rho-specific nucleotide exchange factor, LARG. Proc Natl Acad Sci U S A 99:12085–12090
Lindblom P, Gerhardt H, Liebner S, Abramsson A, Enge M, Hellstrom M, Backstrom G, Fredriksson S, Landegren U, Nystrom HC, Bergstrom G, Dejana E, Ostman A, Lindahl P, Betsholtz C (2003) Endothelial PDGF-B retention is required for proper investment of pericytes in the microvessel wall. Genes Dev 17:1835–1840
Berger M, Bergers G, Arnold B, Hammerling GJ, Ganss R (2005) Regulator of G-protein signaling-5 induction in pericytes coincides with active vessel remodeling during neovascularization. Blood 105:1094–1101
Elhabazi A, Delaire S, Bensussan A, Boumsell L, Bismuth G (2001) Biological activity of soluble CD100. I. The extracellular region of CD100 is released from the surface of T lymphocytes by regulated proteolysis. J Immunol 166:4341–4347
Basile JR, Holmbeck K, Bugge TH, Gutkind JS (2007) MT1-MMP controls tumor-induced angiogenesis through the release of semaphorin 4D. J Biol Chem 282:6899–6905
Binmadi NO, Yang YH, Zhou H, Proia P, Lin YL, Batista De Paula AM, Sena Guimaraes AL, Poswar FO, Sundararajan D, Basile JR (2012) Plexin-B1 and Semaphorin 4D cooperate to promote perineural invasion in a RhoA/ROK-dependent manner. Am J Pathol 180:1232–1242
Zhu P, Goh YY, Chin HF, Kersten S, Tan NS (2012) Angiopoietin-like 4: a decade of research. Biosci Rep 32:211–219
Bergers G, Benjamin LE (2003) Tumorigenesis and the angiogenic switch. Nat Rev Cancer 3:401–410
Gavard J, Gutkind JS (2006) VEGF controls endothelial-cell permeability by promoting the beta-arrestin-dependent endocytosis of VE-cadherin. Nat Cell Biol 8:1223–1234
Abramsson A, Lindblom P, Betsholtz C (2003) Endothelial and nonendothelial sources of PDGF-B regulate pericyte recruitment and influence vascular pattern formation in tumors. J Clin Invest 112:1142–1151
Baluk P, Hashizume H, McDonald DM (2005) Cellular abnormalities of blood vessels as targets in cancer. Curr Opin Genet Dev 15:102–111
Erber R, Thurnher A, Katsen AD, Groth G, Kerger H, Hammes HP, Menger MD, Ullrich A, Vajkoczy P (2004) Combined inhibition of VEGF and PDGF signaling enforces tumor vessel regression by interfering with pericyte-mediated endothelial cell survival mechanisms. FASEB J 18:338–340
Benjamin LE, Golijanin D, Itin A, Pode D, Keshet E (1999) Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J Clin Invest 103:159–165
Basile JR, Gavard J, Gutkind JS (2007) Plexin-B1 utilizes RhoA and Rho kinase to promote the integrin-dependent activation of Akt and ERK and endothelial cell motility. J Biol Chem 282:34888–34895
Marinissen MJ, Gutkind JS (2001) G-protein-coupled receptors and signaling networks: emerging paradigms. Trends Pharmacol Sci 22:368–376
Armulik A, Abramsson A, Betsholtz C (2005) Endothelial/pericyte interactions. Circ Res 97:512–523
Hellstrom M, Gerhardt H, Kalen M, Li X, Eriksson U, Wolburg H, Betsholtz C (2001) Lack of pericytes leads to endothelial hyperplasia and abnormal vascular morphogenesis. J Cell Biol 153:543–553
Guo P, Hu B, Gu W, Xu L, Wang D, Huang HJ, Cavenee WK, Cheng SY (2003) Platelet-derived growth factor-B enhances glioma angiogenesis by stimulating vascular endothelial growth factor expression in tumor endothelia and by promoting pericyte recruitment. Am J Pathol 162:1083–1093
Yamashita J, Itoh H, Hirashima M, Ogawa M, Nishikawa S, Yurugi T, Naito M, Nakao K (2000) Flk1-positive cells derived from embryonic stem cells serve as vascular progenitors. Nature 408:92–96
Fazzari P, Penachioni J, Gianola S, Rossi F, Eickholt BJ, Maina F, Alexopoulou L, Sottile A, Comoglio PM, Flavell RA, Tamagnone L (2007) Plexin-B1 plays a redundant role during mouse development and in tumour angiogenesis. BMC Dev Biol 7:55
Padua D, Zhang XH, Wang Q, Nadal C, Gerald WL, Gomis RR, Massague J (2008) TGFbeta primes breast tumors for lung metastasis seeding through angiopoietin-like 4. Cell 133:66–77
Acknowledgments
We would like to acknowledge Dr. Snigdha Banerjee for the use of C3H/10T1/2 embryonic mesenchymal stem cells. This work was supported by the National Cancer Institute grant R01-CA133162 to J.R.B.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
The Editors-in-Chief have retracted this article following an investigation by the University of Maryland. The institution found that problems in some figures. Due to these errors, the Editors-in-Chief have found that the results are no longer reliable.
About this article
Cite this article
Zhou, H., Yang, YH. & Basile, J.R. RETRACTED ARTICLE: The Semaphorin 4D-Plexin-B1-RhoA signaling axis recruits pericytes and regulates vascular permeability through endothelial production of PDGF-B and ANGPTL4. Angiogenesis 17, 261–274 (2014). https://doi.org/10.1007/s10456-013-9395-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-013-9395-0