Abstract
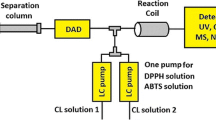
In this study, an end-point-based fluorescence assay for soluble epoxide hydrolase (sEH) was transformed into an on-line continuous-flow format. The on-line biochemical detection system (BCD) was coupled on-line to liquid chromatography (LC) to allow mixture analysis. The on-line BCD was based on a flow system wherein sEH activity was detected by competition of analytes with the substrate hydrolysis. The reaction product was measured by fluorescence detection. In parallel to the BCD data, UV and MS data were obtained through post-column splitting of the LC effluent. The buffer system and reagent concentrations were optimized resulting in a stable on-line BCD with a good assay window and good sensitivity (S/N > 60). The potency of known sEH inhibitors (sEHis) obtained by LC–BCD correlates well with published values. The LC–BCD system was applied to test how oxidative microsomal metabolism affects the potency of three sEHis. After incubation with pig liver microsomes, several metabolites of sEHis were characterized by MS, while their individual potencies were measured by BCD. For all compounds tested, active metabolites were observed. The developed method allows for the first time the detection of sEHis in mixtures providing new opportunities in the development of drug candidates.





Similar content being viewed by others
References
Newman JW, Morisseau C, Hammock BD (2005) Epoxide hydrolases: their roles and interactions with lipid metabolism. Prog Lipid Res 44:1–51
Certikova Chabova V, Walkowska A, Kompanowska-Jezierska E, Sadowski J, Kujal P, Vernerova Z, Vanourkova Z, Kopkan L, Kramer HJ, Falck JR, Imig JD, Hammock BD, Vaneckova I, Cervenka L (2011) Combined inhibition of 20-hydroxyeicosatetraenoic acid formation and of epoxyeicosatrienoic acids degradation attenuates hypertension and hypertension-induced end-organ damage in Ren-2 transgenic rats. Clin Sci (Lond) 118:617–632
Schmelzer KR, Kubala L, Newman JW, Kim IH, Eiserich JP, Hammock BD (2005) Soluble epoxide hydrolase is a therapeutic target for acute inflammation. Proc Natl Acad Sci USA 102:9772–9777
Inceoglu B, Wagner K, Schebb NH, Morisseau C, Jinks SL, Ulu A, Hegedus C, Rose T, Brosnan R, Hammock BD (2011) Analgesia mediated by soluble epoxide hydrolase inhibitors is dependent on cAMP. Proc Natl Acad Sci USA 108:5093–5097
Imig JD, Zhao X, Zaharis CZ, Olearczyk JJ, Pollock DM, Newman JW, Kim IH, Watanabe T, Hammock BD (2005) An orally active epoxide hydrolase inhibitor lowers blood pressure and provides renal protection in salt-sensitive hypertension. Hypertension 46:975–981
Imig JD, Hammock BD (2009) Soluble epoxide hydrolase as a therapeutic target for cardiovascular diseases. Nat Rev Drug Discovery 8:794–805
Wolf NM, Morisseau C, Jones PD, Hock B, Hammock BD (2006) Development of a high-throughput screen for soluble epoxide hydrolase inhibition. Anal Biochem 355:71–80
Jones PD, Wolf NM, Morisseau C, Whetstone P, Hock B, Hammock BD (2005) Fluorescent substrates for soluble epoxide hydrolase and application to inhibition studies. Anal Biochem 343:66–75
Schebb NH, Huby M, Morisseau C, Hwang SH, Hammock BD (2011) Development of an online SPE-LC-MS-based assay using endogenous substrate for investigation of soluble epoxide hydrolase (sEH) inhibitors. Anal Bioanal Chem 400:1359–1366
Ulu A, Davis BB, Tsai HJ, Kim IH, Morisseau C, Inceoglu B, Fiehn O, Hammock BD, Weiss RH (2008) Soluble epoxide hydrolase inhibitors reduce the development of atherosclerosis in apolipoprotein e-knockout mouse model. J Cardiovasc Pharmacol 52:314–323
Ebada SS, Edrada RA, Lin W, Proksch P (2008) Methods for isolation, purification and structural elucidation of bioactive secondary metabolites from marine invertebrates. Nat Protoc 3:1820–1831
Kool J, Giera M, Irth H, Niessen WMA (2011) Advances in mass spectrometry-based post-column bioaffinity profiling of mixtures. Anal Bioanal Chem 399:2655–2668
Schebb NH, Faber H, Maul R, Heus F, Kool J, Irth H, Karst U (2009) Analysis of glutathione adducts of patulin by means of liquid chromatography (HPLC) with biochemical detection (BCD) and electrospray ionization tandem mass spectrometry (ESI-MS/MS). Anal Bioanal Chem 394:1361–1373
Marques LA, Kool J, de Kanter FJJ, Lingeman H, Niessen WMA, Irth H (2010) Production and on-line acetylcholinesterase bioactivity profiling of chemical and biological degradation products of tacrine. J Pharm Biomed Anal 53:609–616
Kool J, van Liempd SM, Harmsen S, Beckman J, van Elswijk D, Commandeur JNM, Irth H, Vermeulen NPE (2007) Cytochrome P450 bio-affinity detection coupled to gradient HPLC: on-line screening of affinities to cytochrome P4501A2 and 2D6. J Chromatogr B 858:49–58
Van Liempd SM, Kool J, Meerman JH, Irth H, Vermeulen NPE (2007) Metabolic profiling of endocrine-disrupting compounds by on-line cytochrome p450 bioreaction coupled to on-line receptor affinity screening. Chem Res Toxicol 20:1825–1832
de Vlieger JSB, Kolkman AJ, Ampt KA, Commandeur JNM, Vermeulen NPE, Kool J, Wijmenga SS, Niessen WMA, Irth H, Honing M (2010) Determination and identification of estrogenic compounds generated with biosynthetic enzymes using hyphenated screening assays, high resolution mass spectrometry and off-line NMR. J Chromatogr B 878:667–674
Falck D, de Vlieger JSB, Niessen WMA, Kool J, Honing M, Giera M, Irth H (2010) Development of an online p38alpha mitogen-activated protein kinase binding assay and integration of LC-HR-MS. Anal Bioanal Chem 398:1771–1780
Beetham JK, Tian T, Hammock BD (1993) cDNA cloning and expression of a soluble epoxide hydrolase from human liver. Arch Biochem Biophys 305:197–201
Kim IH, Morisseau C, Watanabe T, Hammock BD (2004) Design, synthesis, and biological activity of 1,3-disubstituted ureas as potent inhibitors of the soluble epoxide hydrolase of increased water solubility. J Med Chem 47:2110–2122
Jones PD, Tsai HJ, Do ZN, Morisseau C, Hammock BD (2006) Synthesis and SAR of conformationally restricted inhibitors of soluble epoxide hydrolase. Bioorg Med Chem Lett 16:5212–5216
Olearczyk JJ, Field MB, Kim IH, Morisseau C, Hammock BD, Imig JD (2006) Substituted adamantyl-urea inhibitors of the soluble epoxide hydrolase dilate mesenteric resistance vessels. J Pharmacol Exp Ther 318:1307–1314
Hwang SH, Tsai HJ, Liu JY, Morisseau C, Hammock BD (2007) Orally bioavailable potent soluble epoxide hydrolase inhibitors. J Med Chem 50:3825–3840
Morisseau C, Goodrow MH, Newman JW, Wheelock CE, Dowdy DL, Hammock BD (2002) Structural refinement of inhibitors of urea-based soluble epoxide hydrolases. Biochem Pharmacol 63:1599–1608
Falck D, de Vlieger JSB, Giera M, Honing M, Irth H, Niessen WMA, Kool J (2012) On-line electrochemistry-bioaffinity screening with parallel HR-LC-MS for the generation and characterization of modified p38alpha kinase inhibitors. Anal Bioanal Chem 403:367–375
Kool J, Ramautar R, van Liempd SM, Beckman J, de Kanter FJJ, Meerman JH, Schenk T, Irth H, Commandeur JNM, Vermeulen NPE (2006) Rapid on-line profiling of estrogen receptor binding metabolites of tamoxifen. J Med Chem 49:3287–3292
Kool J, Eggink M, van Rossum H, van Liempd SM, van Elswijk DA, Irth H, Commandeur JNM, Meerman JH, Vermeulen NPE (2007) Online biochemical detection of glutathione-S-transferase P1-specific inhibitors in complex mixtures. J Biomol Screen 12:396–405
Schebb NH, Heus F, Saenger T, Karst U, Irth H, Kool J (2008) Development of a countergradient parking system for gradient liquid chromatography with online biochemical detection of serine protease inhibitors. Anal Chem 80:6764–6772
Acknowledgments
This study was supported by NIEHS (R01 ES002710, P42 ES004699), NIOSH (PHS OH07550) and the German Academic Exchange Service. B.D.H is as senior fellow of the American Asthma Society. We acknowledge Lionel Blanchet for his help with data management.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Falck, D., Schebb, N.H., Prihatiningtyas, S. et al. Development of On-line Liquid Chromatography-Biochemical Detection for Soluble Epoxide Hydrolase Inhibitors in Mixtures. Chromatographia 76, 13–21 (2013). https://doi.org/10.1007/s10337-012-2343-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-012-2343-0