Abstract
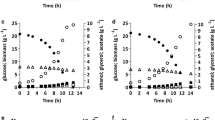
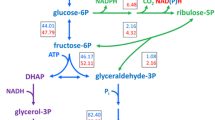
Glycerol is a major by-product of ethanol fermentation by Saccharomyces cerevisiae and typically 2–3% of the sugar fermented is converted to glycerol. Replacing the NAD+-regenerating glycerol pathway in S. cerevisiae with alternative NADH reoxidation pathways may be useful to produce metabolites of biotechnological relevance. Under fermentative conditions yeast reoxidizes excess NADH through glycerol production which involves NADH-dependent glycerol-3-phosphate dehydrogenases (Gpd1p and Gpd2p). Deletion of these two genes limits fermentative activity under anaerobic conditions due to accumulation of NADH. We investigated the possibility of converting this excess NADH to NAD+ by transforming a double mutant (gpd1∆gpd2∆) with alternative oxidoreductase genes that might restore the redox balance and produce either sorbitol or propane-1,2-diol. All of the modifications improved fermentative ability and/or growth of the double mutant strain in a self-generated anaerobic high sugar medium. However, these strain properties were not restored to the level of the parental wild-type strain. The results indicate an apparent partial NAD+ regeneration ability and formation of significant amounts of the commodity chemicals like sorbitol or propane-1,2-diol. The ethanol yields were maintained between 46 and 48% of the sugar mixture. Other factors apart from the maintenance of the redox balance appeared to influence the growth and production of the alternative products by the genetically manipulated strains.



Similar content being viewed by others
References
Akinterinwa O, Khankal R, Cirino PC (2008) Metabolic engineering for bioproduction of sugar alcohols. Curr Opin Biotechnol 19:461–467
Albertyn J, Hohmann S, Thevelein JM, Prior BA (1994) GPD1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic stress in Saccharomyces cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol Cell Biol 14:4135–4144
Ansell R, Granath K, Hohmann S, Thevelein JM, Adler L (1997) The two isoenzymes for yeast NAD+-dependent glycerol 3-phosphate dehydrogenase encoded by GPD1 and GPD2 have distinct roles in osmoadaptation and redox regulation. EMBO J 16:2179–2187
Bakker BM, Overkamp KM, van Maris AJ, Kötter P, Luttik MA, van Dijken JP, Pronk JT (2001) Stoichiometry and compartmentation of NADH metabolism in Saccharomyces cerevisiae. FEMS Microbiol Rev 25:15–37
Bennett GN, San KY (2001) Microbial formation, biotechnological production and applications of 1,2-propanediol. Appl Microbiol Biotechnol 55:1–9
Berben G, Dumont J, Gilliquet V, Bolle PA, Hilger F (1991) The YDp plasmids: a uniform set of vectors bearing versatile gene disruption cassettes for Saccharomyces cerevisiae. Yeast 7:475–477
Bisson LF, Fraenkel DG (1983) Involvement of kinases in glucose and fructose uptake by Saccharomyces cerevisiae. Proc Natl Acad Sci U S A 80:1730–1734
Bjorkqvist S, Ansell R, Adler L, Liden G (1997) Physiological response to anaerobicity of glycerol-3-phosphate dehydrogenase mutants of Saccharomyces cerevisiae. Appl Environ Microbiol 63:128–132
Brachmann CB, Davies A, Cost GJ, Caputo E, Li J, Hieter P, Boeke JD (1998) Designer deletion strains derived from Saccharomyces cerevisiae S288C: a useful set of strains and plasmids for PCR-mediated gene disruption and other applications. Yeast 14:115–132
Clifton D, Walsh RB, Fraenkel DG (1993) Functional studies of yeast glucokinase. J Bacteriol 175:3289–3294
da Silva GP, Mack M, Contiero J (2009) Glycerol: a promising and abundant carbon source for industrial microbiology. Biotechnol Adv 27:30–39
de Smidt O, du Preez JC, Albertyn J (2008) The alcohol dehydrogenases of Saccharomyces cerevisiae: a comprehensive review. FEMS Yeast Res 8:967–978
Gancedo C, Gancedo JM, Sols A (1968) Glycerol metabolism in yeasts. Eur J Biochem 5:165–172
Gellissen G, Hollenberg CP (1997) Application of yeasts in gene expression studies: a comparison of Saccharomyces cerevisiae, Hansenula polymorpha and Kluyveromyces lactis—a review. Gene 190:87–97
Gietz RD, Schiestl RH (2007) High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nat Protoc 2:31–34
Gordon JH, Dubos R (1970) The anaerobic bacterial flora of the mouse cecum. J Exp Med 132:251–260
Gururajan VT, Gorwa-Grauslund MF, Hahn-Hagerdal BH (2007) A constitutive catabolite repression mutant of a recombinant Saccharomyces cerevisiae strain improves xylose consumption during fermentation. Ann Microbiol 57:85–92
Harju S, Fedosyuk H, Peterson KR (2004) Rapid isolation of yeast genomic DNA: Bust n’ Grab. BMC Biotechnol 4:8–12
Hirai M (1981) Purification and characteristics of sorbitol-6-phosphate dehydrogenase from loquat leaves. Plant Physiol 67:221–224
Hohmann S (2002) Osmotic stress signaling and osmoadaptation in yeasts. Microbiol Mol Biol Rev 66:300–372
Jung JY, Choi ES, Oh MK (2008) Enhanced production of 1,2-propanediol by tpi1 deletion in Saccharomyces cerevisiae. J Microbiol Biotechnol 18:1797–1802
Kuhn A, van Zyl C, van Tonder A, Prior BA (1995) Purification and partial characterization of an aldo-keto reductase from Saccharomyces cerevisiae. Appl Environ Microbiol 61:1580–1585
Ladero V, Ramos A, Wiersma A, Goffin P, Schanck A, Kleerebezem M, Hugenholtz J, Smid EJ, Hols P (2007) High-level production of the low calorie sugar sorbitol by Lactobacillus plantarum through metabolic engineering. Appl Environ Microbiol 73:1864–1872
Louw L, Roux K, Tredoux A, Tomic O, Naes T, Nieuwoudt HH, van Rensburg P (2009) Characterization of selected South African young cultivar wines using FTMIR spectroscopy, gas chromatography, and multivariate data analysis. J Agric Food Chem 57:2623–2632
Lunzer R, Mamnun Y, Haltrich D, Kulbe KD, Nidetzky B, Lunzer R (1998) Structural and functional properties of a yeast xylitol dehydrogenase, a Zn2+-containing metalloenzyme similar to medium-chain sorbitol dehydrogenases. Biochem J 336:91–99
Luyten K, Albertyn J, Skibbe WF, Prior BA, Ramos J, Thevelein JM, Hohmann S (1995) Fps1, a yeast member of the MIP family of channel proteins, is a facilitator for glycerol uptake and efflux and it is inactive under osmotic stress. EMBO J 14:1360–1371
Malherbe DF (2010) Characterization and evaluation of glucose oxidase activity in recombinant Saccharomyces cerevisiae strains. PhD Thesis, Stellenbosch University
Martins AM, Cordeiro CA, Ponces Freire AM, Martins AM (2001) In situ analysis of methylglyoxal metabolism in Saccharomyces cerevisiae. FEBS Lett 499:41–44
Mulligan KJ (1996) A procedure to determine diethylene glycol (2,2′-oxybisethanol) and ethylene glycol (1,2-ethanediol) in glycerin and selected products. Labo Inform Bull 12:1–7
Nissen TL, Hamann CW, Kielland-Brandt MC, Nielsen J, Villadsen J (2000) Anaerobic and aerobic batch cultivations of Saccaharomyces cerevisiae mutants impaired in glycerol synthesis. Yeast 16:463–474
Novotny MJ, Reizer J, Esch F, Saier MH (1984) Purification and properties of D-mannitol-1-phosphate dehydrogenase and D-glucitol-6-phosphate dehydrogenase from Escherichia coli. J Bacteriol 159:986–990
Pahlman AK, Granath K, Ansell R, Hohmann S, Adler L (2001) The yeast glycerol 3-phosphatases Gpp1p and Gpp2p are required for glycerol biosynthesis and differentially involved in the cellular responses to osmotic, anaerobic, and oxidative stress. J Biol Chem 276:3555–3563
Parent SA, Fenimore CM, Bostian KA (1985) Vector systems for the expression, analysis and cloning of DNA sequences in S. cerevisiae. Yeast 1:83–138
Prior BA, Hohmann S (1997) Glycerol production and osmoregulation. In: Zimmermann FK, Entian KD (eds) Yeast sugar metabolism. Lancaster, USA, pp 313–337
Saadat D, Harrison DH (1998) Identification of catalytic bases in the active site of Escherichia coli methylglyoxal synthase: cloning, expression, and functional characterization of conserved aspartic acid residues. Biochem 37:10074–10086
Sarthy AV, Schopp C, Idler KB (1994) Cloning and sequence determination of the gene encoding sorbitol dehydrogenase from Saccharomyces cerevisiae. Gene 140:121–126
Shen B, Hohmann S, Jensen RG, Bohnert HJ (1999) Roles of sugar alcohols in osmotic stress adaption. Replacement of glycerol by mannitol and sorbitol in yeast. Plant Physiol 123:45–52
Tao R, Uratsu SL, Dandekar AM (1995) Sorbitol synthesis in transgenic tobacco with apple cDNA encoding NADP+-dependent sorbitol-6-phosphate dehydrogenase. Plant Cell Physiol 36:525–532
Truniger V, Boos W (1994) Mapping and cloning of gldA, the structural gene of the Escherichia coli glycerol dehydrogenase. J Bacteriol 176:1796–1800
van Dijken JP, Scheffers WA (1986) Redox balances in the metabolism of sugars by yeasts. FEMS Microbiol Rev 32:199–224
van Eck JH, Prior BA, Brandt EV (1993) The water relations of growth and polyhydroxy alcohol production by ascomycetous yeasts. J Gen Microbiol 139:1047–1054
Verduyn C, Van Kleef R, Frank J, Schreuder H, Van Dijken JP, Scheffers WA (1985) Properties of the NAD(P)H-dependent xylose reductase from the xylose-fermenting yeast Pichia stipitis. Biochem J 226:669–677
Volschenk H, Viljoen M, Grobler J, Petzold B, Bauer F, Subden RE, Young RA, Lonvaud A, Denayrolles M, van Vuuren HJ (1997) Engineering pathways for malate degradation in Saccharomyces cerevisiae. Nat Biotechnol 15:253–257
Acknowledgments
This work was supported by the National Research Foundation (NRF) of South Africa and by Winetech, the research funding body of the South African wine industry. We thank D. Malherbe and V. T. Gururajan for kindly providing the pDMPM and pSTAH vectors respectively for cloning purposes. The technical assistance of A. Tredoux and Marieke Stander in analysis is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jain, V.K., Divol, B., Prior, B.A. et al. Elimination of glycerol and replacement with alternative products in ethanol fermentation by Saccharomyces cerevisiae . J Ind Microbiol Biotechnol 38, 1427–1435 (2011). https://doi.org/10.1007/s10295-010-0928-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-010-0928-x