Abstract
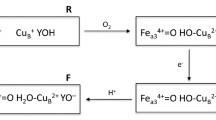
The reaction of the catalase-peroxidase of Burkholderia pseudomallei with peroxyacetic acid has been analyzed using stopped-flow spectrophotometry. Two well-defined species were observed, the first defined by an increase in intensity and narrowing of the Soret band at 407 nm and a 10-nm shift of the charge transfer band from 635 to 625 nm. These features are consistent with a ferric spectrum with a greater proportion of sixth-coordination character and are assigned to an FeIII–peroxyacetic acid complex. Complementary 9-GHz EPR characterization of the changes in the ferric signal of the resting enzyme induced by the binding of acetate in the heme pocket substantiates the proposal. Kinetic analysis of the spectral changes as a function of peroxyacetic acid concentration revealed two independent peroxyacetic acid binding events, one coincident with formation of the FeIII–peroxyacetic acid complex and the other coincident with the heme oxidation to the subsequent ferryl intermediate. A model to explain the need for two peroxyacetic acid binding events is proposed. The reaction of the W330F variant followed similar kinetics, although the characteristic spectral features of the FeIV=O Por•+ species were detected. The variant D141A lacking an aspartate at the entrance to the heme cavity as well as the R108A and D141A/R108A variants showed no evidence for the FeIII–peroxyacetic acid complex, only the formation of ferryl species with absorbance maxima at 414, 545, and 585 nm.






Similar content being viewed by others
References
Singh R, Wiseman B, Deemagarn T, Donald LJ, Duckworth HW, Carpena X, Fita I, Loewen PC (2004) J Biol Chem 279:43098–43106
Singh R, Wiseman B, Deemagarn T, Jha V, Switala J, Loewen PC (2008) Arch Biochem Biophys 417:207–214
Claiborne A, Fridovich I (1979) J Biol Chem 254:4245–4252
Loewen PC, Triggs BL, George CS, Hrabarchuk BE (1985) J Bacteriol 162:661–667
Triggs-Raine BL, Doble BL, Mulvey MR, Sorby PA, Loewen PC (1988) J Bacteriol 170:4415–4419
Zhang Y, Heym B, Allen B, Young D, Cole S (1992) Nature 358:591–593
Sivaraja M, Goodin DB, Smith M, Hoffman B (1989) Science 245:738–740
Blodig W, Smith AT, Winterhalter K, Piontek K (1999) Arch Biochem Biophys 370:86–92
Singh R, Switala J, Loewen PC, Ivancich A (2007) J Am Chem Soc 129:15954–15963
Chouchane S, Lippai I, Magliozzo RS (2000) Biochemistry 39:9975–9983
Jakopitsch C, Auer M, Regelsberger G, Jantschko W, Furtmuller PG, Ruker F, Obinger C (2003) Biochemistry 42:5292–5300
Jakopitsch C, Ivancich A, Schmuckenschlager F, Wanasinghe A, Potl G, Furtmuller PG, Ruker F, Obinger C (2004) J Biol Chem 279:46082–46095
Jakopitsch C, Auer M, Regelsberger G, Jantschko W, Furtmuller PG, Ruker F, Obinger C (2003) Eur J Biochem 270:1006–1013
Jakopitsch C, Auer M, Ivancich A, Ruker F, Furtmuller PG, Obinger C (2003) J Biol Chem 278:20185–20191
Regelsberger G, Jakopitsch D, Ruker F, Krois D, Peschek GA, Obinger C (2000) J Biol Chem 275:22854–22861
Jakopitsch C, Vlasits J, Wiseman B, Loewen PC, Obinger C (2007) Biochemistry 46:1183–1193
Ghiladi RA, Knudsen GM, Medzihradszky KF, Ortiz de Montellano PR (2005) J Biol Chem 280:22651–22663
Spotilak T, Dawson JH, Ballou DP (2005) J Biol Chem 280:20300–20309
Carpena X, Switala J, Loprasert S, Mongkolsuk S, Fita I, Loewen PC (2002) Acta Crystallogr D 58:2184–2186
Rørth M, Jensen PK (1967) Biochim Biophys Acta 139:171–173
Childs RE, Bardsley WG (1975) Biochem J 145:93–103
Layne E (1957) Methods Enzymol 3:447–454
Carpena X, Wiseman B, Deemagarn T, Herguedas B, Ivancich A, Singh R, Loewen PC, Fita I (2006) Biochemistry 45:5171–5179
Emsley P, Cowtan K (2004) Acta Crystallogr D 60:2126–2132
Deemagarn T, Wiseman B, Carpena X, Ivancich A, Fita I, Loewen PC (2007) Proteins 66:219–228
Schonbaum GR (1973) J Biol Chem 248:502–511
Henriksen A, Schuller DJ, Meno K, Welinder KG, Smith AT, Gajhede M (1998) Biochemisty 37:8054–8060
Loewen PC, Carpena X, Rovira C, Haas R, Odenbreit S, Nicholls P, Fita I (2004) Biochemistry 43:3089–3103
Dunford HB (1999) Heme peroxidases. Wiley, New York
Fielding AJ, Singh R, Boscolo B, Loewen PC, Ghibaudi EI, Ivancich A (2008) Biochemistry 47:9781–9792
Ivancich A, Jakopitsch C, Auer M, Un S, Obinger C (2003) J Am Chem Soc 125:14093–14102
Erman JE, Vitello LB, Mauro JM, Kraut J (1989) Biochemistry 28:7992–7995
Rodriguez-Lopez JN, Smith AT, Thorneley RNF (1996) J Biol Chem 271:4023–4030
Loew G, Dupuis M (1996) J Am Chem Soc 118:10584–10587
Derat E, Shaik S, Rovira C, Vidossich P, Alfonso-Prieto M (2007) J Am Chem Soc 129:6346–6347
Acknowledgments
This work was supported by the French CNRS and CEA Saclay (to A.I.), a Ph.D. Fellowship (CFR contract from CEA Saclay to J.C.), and grants from the Natural Sciences and Engineering Research Council of Canada (to P.C.L.) and the Canadian Research Chair Program (to P.C.L.).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wiseman, B., Colin, J., Smith, A.T. et al. Mechanistic insight into the initiation step of the reaction of Burkholderia pseudomallei catalase-peroxidase with peroxyacetic acid. J Biol Inorg Chem 14, 801–811 (2009). https://doi.org/10.1007/s00775-009-0493-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-009-0493-9