Abstract
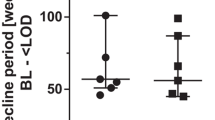
Antiretroviral therapy is limited by the development of human immunodeficiency virus (HIV) resistance mutations. Although resistance testing is recommended during therapy failure, little is known about the optimal time points for testing or its impact on treatment. In this study, we investigated HIV polymorphisms and mutations and assessed their influence on the outcome of highly active antiretroviral therapy (HAART). We focused on viral load and CD4+ cell counts as the most important parameters for therapy response. Resistance mutations were present in 19% of all patients prior to antiretroviral treatment. Mutations causing direct antiretroviral drug resistance were observed in 10%. Analyzing therapy response, we found a significant correlation between resistance mutations and impaired CD4+ cell recovery six months after the initiation of antiretroviral treatment. Lower CD4+ cell counts were also observed in a subgroup of patients infected with a virus presenting mutations that directly lowered drug susceptibility.




Similar content being viewed by others
Abbreviations
- ABC:
-
Abacavir, Ziagen
- AIDS:
-
Acquired immunodeficiency syndrome
- APV:
-
Amprenavir, agenerase
- ART:
-
Antiretroviral therapy
- ATV:
-
Atazanavir, Reyataz
- AZT:
-
Zidovudine, Retrovir
- CDC:
-
Centers for disease control
- CCR:
-
Chemokine co-receptor
- CD:
-
Cluster of differentiation
- d4T:
-
Stavudine, Zerit
- ddC:
-
Zalcitabine, Hivid
- ddl:
-
Didanosine, Videx
- DLV:
-
Delavirdine, rescriptor
- EFV:
-
Efavirenz, Sustiva
- FACS:
-
Fluorescence-activated cell sorter
- FTC:
-
Emtricitabine, Emtriva
- HAART:
-
Highly active antiretroviral therapy
- HIV:
-
Human immunodeficiency virus
- HT:
-
Heterozygote
- IDV:
-
Indinavir, Crixivan
- LPV:
-
Lopinavir+Ritonavir, Kaletra
- NFV:
-
Nelfinavir, Viracept
- NVP:
-
Nevirapine, Viramune
- NRTI:
-
Nucleoside reverse transcriptase inhibitor
- NNRTI:
-
Non-nucleoside reverse transcriptase inhibitor
- PCR:
-
Polymerase chain reaction
- PE:
-
Phycoerytrin
- PI:
-
Protease inhibitor
- SOC:
-
Standard of care
- T20:
-
Enfuvirtide, Fuzeon
- TDF:
-
Tenofovir disoproxil fumarate, Viread
- RT:
-
Reverse transcriptase
- RTV:
-
Ritonavir, Norvir
- SQV:
-
Saquinavir, Fortovase
- WHO:
-
World Health Organisation
- WT:
-
Wild type
- 3TC:
-
Lamivudine; Epivir
References
WHO, UNICEF and UNAIDS (2010) Towards universal access: scaling up priority HIV/AIDS interventions in the health sector: Progress report 2009. Geneva, World Health Organization (http://www.who.int/hiv/2009progressreport/report/en/index.html)
UNAIDS (2010) Global report: UNAIDS report on the global AIDS epidemic 2010. UNAIDS/10.11E / JC1985E Nov 2010
Hecht FM, Grant RM, Petropoulos CJ, Dillon B, Chesney MA, Tian H, Hellmann NS, Bandrapalli NI, Digilio L, Branson B, Kahn JO (1998) Sexual transmission of an HIV-1 variant resistant to multiple reverse-transcriptase and protease inhibitors. N Engl J Med 339:307–311
Imrie A, Beveridge A, Genn W, Vizzard J, Cooper DA (1997) Transmission of human immunodeficiency virus type 1 resistant to nevirapine and zidovudine: Sydney primary HIV infection study group. J Infect Dis 175:1502–1506
Grant RM, Hecht FM, Warmerdam M, Liu L, Liegler T, Petropoulos CJ, Hellmann NS, Chesney M, Busch MP, Kahn JO (2002) Time trends in primary HIV-1 drug resistance among recently infected persons. JAMA 288:181–188
Little SJ, Holte S, Routy JP, Daar ES, Markowitz M, Collier AC, Koup RA, Mellors JW, Connick E, Conway B, Kilby M, Wang L, Whitcomb JM, Hellmann NS, Richman DD (2002) Antiretroviral-drug resistance among patients recently infected with HIV. N Engl J Med 347:385–394
Wensing AM, Vijver A, Angarano G, Asjo B, Balotta C, Zazzi M, Boucher C (2005) Prevalence of drug-resistant HIV-1 variants in untreated individuals in Europe: implications for clinical management. J Infect Dis 192:958–966
Oette M, Kaiser R, Däumer R, Akbari R, Fätkenheuer G, Rockstroh JK, Stechel J, Rieke A, Mauss S, Schmalöer D, Göbels K, Voigt C, Wettstein M, Häussinger D (2004) Primary drug-resistance in HIV-positive patients on initiation of first-line antiretroviral therapy in Germany. Eur J Med Res 9:278
Alexander CS, Dong W, Chan K, Jahnke N, O’Shaughnessy MV, Mo T, Piaseczny MA, Montaner JS, Harrigan PR (2001) HIV protease and reverse transcriptase variation and therapy outcome in antiretroviral-naive individuals from a large North American cohort. AIDS 15:601–607
Perno CF, Cozzi-Lepri A, Balotta C, Bertoli A, Violin M, Monno L, Zauli T, Montroni M, Ippolito G, D’Arminio-Monforte A (2002) Low prevalence of primary mutations associated with drug resistance in antiviral-naive patients at therapy initiation. AIDS 16:619–624
Geretti AM (2007) Epidemiology of antiretroviral drug resistance in drug-naive persons. Curr Opin Infect Dis 20:22–32
EuroGuidelines Group for HIV Resistance (2001) Clinical and laboratory guidelines for the use of HIV-1 drug resistance testing as part of treatment management: Recommendation for the European setting. AIDS 15:309–320
Durant J, Clevenbergh P, Halfon P, Delgiudice P, Porsin S, Simonet P, Montagne N, Boucher CA, Schapiro JM, Dellamonica P (1999) Drug-resistance genotyping in HIV-1 therapy: the VIRADAPT randomised controlled trial. Lancet 353:2195–2199
Tural C, Ruiz L, Holtzer C et al (2002) Clinical utility of HIV-genotyping and expert advice: the Havana trial. AIDS 16:209–218
Meynard JL, Vray M, Morand-Joubert L et al (2002) Impact of treatmentguided by phentotypic or genotypic resistance tests on the response to antiretroviral therapy: a randomized trial (NARVAL, ANRS 088). AIDS 16:727–736
Vray M, Meynard JL, Dalban C, Morand-Joubert L, Clavel F, Brun-Vezinet F, Peytavin G, Costagliola D, Girard PM (2003) Predictors of the virological response to a change in the antiretroviral treatment regimen in HIV-1-infected patients enrolled in a randomized trial comparing genotyping, phenotyping and standard of care (Narval trial, ANRS 088). Antivir.Ther 8:427–434
Mellors JW, Rinaldo CR Jr, Gupta P (1996) Prognosis in HIV-1 infection predicted by the quantity of virus in plasma. Science 272:1167–1170
Hulgan T, Shepherd BE, Raffanti SP, Fusco JS, Beckerman R, Barkanic G, Sterling TR (2007) Absolute count and percentage of CD4+ Lymphocytes are independent predictors of disease progression in HIV-infected persons initiating highly active antiretroviral therapy. J Infect Dis 195:425–431
Perno CF, Lepri AC, Forbici F, Bertoli A, Violin M, Mura MS, Cadeo G, Orani A, Chirianni A, Stefano C, Balotta C, Monforte AD (2004) Minor mutations in HIV protease at baseline and apperance of primary mutation 90M in patients for whom their first protease-inhibitor antiretroviral regimens failed. J Infect Dis 189:1983–1987
Shafer RW (2006). Rationale and uses of a public HIV drug-resistance database. J Infect Dis 194(Suppl 1):S51–S58. http://hivdb.stanford.edu
Ioannidis JP, Rosenberg PS, Goedert JJ, Ashton LJ, Benfield TL, Buchbinder SP, Coutinho RA, Eugen-Olsen J, Gallart T, Katzenstein TL, Kostrikis LG, Kuipers H, Louie LG, Mallal SA, Margolick JB, Martinez OP, Meyer L, Michael NL, Operskalski E, Pantaleo G, Rizzardi GP, Schuitemaker H, Sheppard HW, Stewart GJ, Theodorou ID, Ullum H, Vicenzi E, Vlahov D, Wilkinson D, Workman C, Zagury JF, O’brien TR (2001) Effects of CCR5-Delta32, CCR2-64I, and SDF-1 3′A alleles on HIV-1 disease progression: An international meta-analysis of individual-patient data. Ann Intern Med 135:782–795
Mansky LM (1998) Retrovirus mutation rates and their role in genetic variation. J Gen Virol 79:1337–1345
Shafer RW, Rhee SY, Pillay D, Miller V, Sandstrom P, Schapiro JM, Kuritzkes DR, Bennett D (2007) HIV-1 protease and reverse transcriptase mutations for drug resistance surveillance. AIDS 21:215–223
Vandamme AM, Sonnerborg A, Ait-Khaled M, Albert J, Asjo B, Bacheler L, Banhegyi D, Boucher C, Brun-Vezinet F, Camacho R, Clevenbergh P, Clumeck N, Dedes N, De Luca A, Doerr HW, Faudon JL, Gatti G, Gerstoft J, Hall WW, Hatzakis A, Hellmann N, Horban A, Lundgren JD, Kempf D, Miller M, Miller V, Myers TW, Nielsen C, Opravil M, Palmisano L, Perno CF, Phillips A, Pillay D, Pumarola T, Ruiz L, Salminen M, Schapiro J, Schmidt B, Schmit JC, Schuurman R, Shulse E, Soriano V, Staszewski S, Vella S, Youle M, Ziermann R, Perrin L (2004) Updated European recommendations for the clinical use of HIV drug resistance testing. Antivir Ther 9:829–848
Weinstein MC, Goldie SJ, Losina E, Cohen CJ, Baxter JD, Zhang H, Kimmel AD, Freedberg KA (2001) Use of genotypic resistance testing to guide HIV therapy: clinical impact and cost-effectiveness. Ann Intern Med 134:440–450
Fox JM, Fidler S, Weber J (2006) Resistance to HIV drugs in UK may be lower in some areas. BMJ 332:179–180
Mocroft A, Phillips AN, Gatell J et al (2007) Normalisation of CD4 counts in patients with HIV-1 infection and maximum virological suppression who are taking combination antiretroviral therapy: an observational cohort study. Lancet 370:407–413
Hammer SM, Saag MS, Schechter M, Montaner JS, Schooley RT, Jacobsen DM, Thompson MA, Carpenter CC, Fischl MA, Gazzard BG, Gatell JM, Hirsch MS, Katzenstein DA, Richman DD, Vella S, Yeni PG, Volberding PA (2006) Treatment for adult HIV infection: 2006 recommendations of the International AIDS Society-USA panel. JAMA 296:827–843
Richman DD, Morton SC, Wrin T, Hellmann N, Berry S, Shapiro MF, Bozzette SA (2004) The prevalence of antiretroviral drug resistance in the United States. AIDS 18:1393–1401
Wensing AM, Boucher CA (2003) Worldwide transmission of drug-resistant HIV. AIDS Rev 5:140–155
Deeks SG, Wrin T, Liegler T et al (2001) Virologic and immunologic consequences of discontinuing combination antiretroviral drug therapy in HIV-infected patients with detectable viremia. N Engl J Med 344:472–480
Barbour JD, Wrin T, Grant RM et al (2002) Evolution of phenotypic drug susceptibility and viral replication capacity during longterm virologic failure of protease inhibitor therapy in human immunodeficiency virus-infected adults. J Virol 76:11104–11112
Taiwo B, Gallien S, Aga E, Ribaudo H, Haubrich R, Kuritzkes DR, Eron JJ Jr (2011) Antiretroviral drug resistance in HIV-1-infected patients experiencing persistent low-level viremia during first-line therapy. J Infect Dis 204(4):515–520
Cappuccio A, Castiglione F, Piccoli B, Tozzi V (2008) Evaluation of HIV-1 and CD4+ T cell dynamic parameters in patients treated with genotypic resistance testing-guided HAART. Curr HIV Res 6(4):363–369
Little SJ, Holte S, Routy JP, Daar ES, Markowitz M, Collier AC, Koup RA, Mellors JW, Connick E, Conway B, Kilby M, Wang L, Whitcomb JM, Hellmann NS, Richman DD (2002) Antiretroviral-drug resistance among patients recently infected with HIV. N Engl J Med 347:385–394
Little SJ (2001) Is transmitted drug resistance in HIV on the rise? It seems so. BMJ 322:1074–1075
Blower S, Ma L, Farmer P, Koenig S (2003) Predicting the impact of antiretrovirals in resource-poor settings: preventing HIV infections whilst controlling drug resistance. Curr Drug Targets Infect Disord 3:345–353
Smith D, Moini N, Pesano R, Cachay E, Aiem H, Lie Y, Richman D, Little S (2007) Clinical utility of HIV standard genotyping among antiretroviral-naive individuals with unknown duration of infection. Clin Infect Dis 44:456–458
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sühs, KW., Stoll, M., Diem, R. et al. Impaired CD4+ cell recovery during antiretroviral therapy in patients with HIV resistance mutations. Arch Virol 157, 433–440 (2012). https://doi.org/10.1007/s00705-011-1191-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-011-1191-9