Abstract
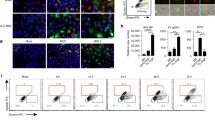
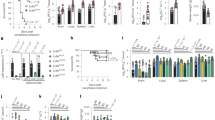
Recently, it has been demonstrated that the MHV-68 ORF20-encoded gene product induces cell-cycle arrest at the G2/M phase, followed by apoptosis. To study the role of this conserved gene in vivo, two independent ORF20-deficient MHV-68 viruses and their revertants were constructed. As the replication in vitro of both mutants followed similar kinetics to that of the wild-type and revertant viruses, ORF20 is therefore a nonessential virus gene. No cell cycle arrest could be observed upon infection of cells with wild type MHV-68 or mutant viruses. In addition, no major differences were detected between mock- and virus-infected cells when protein and inactivation levels of the mitotic promoter factor cdc2/cyclinB were analyzed. Following intranasal infection, the recovery of mutant, revertant and wild-type viruses in the lungs was similar. With the ORF20-deficient viruses, however, there was a significant delay of four days in clearance of virus from the lungs. Surprisingly, the magnitude and cell population distribution in the exudates of the lung was essentially similar to mice infected with wild-type, revertant or ORF20-deleted viruses. Subsequent establishment of latency was normal for both mutants, demonstrating that ORF20 does not play a critical role in establishment of a persistent infection. These results indicate that while expression of ORF20 may impact on the pathogenicity of the infection, the observed induction of G2/M arrest in ORF20-expressing cells may not be the primary function of ORF20 in the context of viral infection.







Similar content being viewed by others
References
Adler H, Messerle M, Wagner M, Koszinowski UH (2000) Cloning and mutagenesis of the murine gammaherpesvirus 68 genome as an infectious bacterial artificial chromosome. J Virol 74:6964-6974
Adler H, Messerle M, Koszinowski UH (2003) Cloning of herpesviral genomes as bacterial artificial chromosomes. Rev Med Virol 13:111-121
Belz GT, Doherty PC (2001) Virus-specific and bystander CD8+ T-cell proliferation in the acute and persistent phases of a gammaherpesvirus infection. J Virol 75:4435-4438
Bertrand L, Pearson A (2008) The conserved N-terminal domain of herpes simplex virus 1 UL24 protein is sufficient to induce the spatial redistribution of nucleolin. J Gen Virol 89:1142-1151
Blakeney S, Kowalski J, Tummolo D, DeStefano J, Cooper D, Guo M, Gangolli S, Long D, Zamb T, Natuk RJ, Visalli RJ (2005) Herpes simplex virus type 2 UL24 gene is a virulence determinant in murine and guinea pig disease models. J Virol 79:10498-10506
Bortz E, Whitelegge JP, Jia Q, Zhou ZH, Stewart JP, Wu TT, Sun R (2003) Identification of proteins associated with murine gammaherpesvirus 68 virions. J Virol 77:13425-13432
Castillo JP, Kowalik TF (2002) Human cytomegalovirus immediate early proteins and cell growth control. Gene 290:19-34
Cotter MA, 2nd, Robertson ES (2002) Molecular biology of Kaposi’s sarcoma-associated herpesvirus. Front Biosci 7:d358-375
Direkze S, Laman H (2004) Regulation of growth signalling and cell cycle by Kaposi’s sarcoma-associated herpesvirus genes. Int J Exp Pathol 85:305-319
Dittmer D, Mocarski ES (1997) Human cytomegalovirus infection inhibits G1/S transition. J Virol 71:1629-1634
Dolcetti R, Masucci MG (2003) Epstein-Barr virus: induction and control of cell transformation. J Cell Physiol 196:207-218
Ehtisham S, Sunil-Chandra NP, Nash AA (1993) Pathogenesis of murine gammaherpesvirus infection in mice deficient in CD4 and CD8 T cells. J Virol 67:5247-5252
Flemington EK (2001) Herpesvirus lytic replication and the cell cycle: arresting new developments. J Virol 75:4475-4481
Hobbs WE, 2nd, DeLuca NA (1999) Perturbation of cell cycle progression and cellular gene expression as a function of herpes simplex virus ICP0. J Virol 73:8245-8255
Jacobson JG, Martin SL, Coen DM (1989) A conserved open reading frame that overlaps the herpes simplex virus thymidine kinase gene is important for viral growth in cell culture. J Virol 63:1839-1843
Jacobson JG, Chen SH, Cook WJ, Kramer MF, Coen DM (1998) Importance of the herpes simplex virus UL24 gene for productive ganglionic infection in mice. Virology 242:161-169
Lymberopoulos MH, Pearson A (2007) Involvement of UL24 in herpes-simplex-virus-1-induced dispersal of nucleolin. Virology 363:397-409
Nascimento R, Parkhouse RM (2007) Murine gammaherpesvirus 68 ORF20 induces cell-cycle arrest in G2 by inhibiting the Cdc2-cyclin B complex. J Gen Virol 88:1446-1453
Nascimento R, Dias JD, Parkhouse RM (2009) The conserved UL24 family of human alpha, beta and gamma herpesviruses induces cell cycle arrest and inactivation of the cyclinB/cdc2 complex. Arch Virol 154:1143-1149
Pearson A, Coen DM (2002) Identification, localization, and regulation of expression of the UL24 protein of herpes simplex virus type 1. J Virol 76:10821-10828
Sanchez V, McElroy AK, Yen J, Tamrakar S, Clark CL, Schwartz RA, Spector DH (2004) Cyclin-dependent kinase activity is required at early times for accurate processing and accumulation of the human cytomegalovirus UL122-123 and UL37 immediate-early transcripts and at later times for virus production. J Virol 78:11219-11232
Sanchez V, Spector DH (2006) Cyclin-dependent kinase activity is required for efficient expression and posttranslational modification of human cytomegalovirus proteins and for production of extracellular particles. J Virol 80:5886-5896
Sanders PG, Wilkie NM, Davison AJ (1982) Thymidine kinase deletion mutants of herpes simplex virus type 1. J Gen Virol 63:277-295
Schang LM, Phillips J, Schaffer PA (1998) Requirement for cellular cyclin-dependent kinases in herpes simplex virus replication and transcription. J Virol 72:5626-5637
Schang LM, Rosenberg A, Schaffer PA (1999) Transcription of herpes simplex virus immediate-early and early genes is inhibited by roscovitine, an inhibitor specific for cellular cyclin-dependent kinases. J Virol 73:2161-2172
Schang LM, Rosenberg A, Schaffer PA (2000) Roscovitine, a specific inhibitor of cellular cyclin-dependent kinases, inhibits herpes simplex virus DNA synthesis in the presence of viral early proteins. J Virol 74:2107-2120
Simas JP, Efstathiou S (1998) Murine gammaherpesvirus 68: a model for the study of gammaherpesvirus pathogenesis. Trends Microbiol 6:276-282
Simas JP, Swann D, Bowden R, Efstathiou S (1999) Analysis of murine gammaherpesvirus-68 transcription during lytic and latent infection. J Gen Virol 80 (Pt 1):75-82
Song MJ, Hwang S, Wong WH, Wu TT, Lee S, Liao HI, Sun R (2005) Identification of viral genes essential for replication of murine gamma-herpesvirus 68 using signature-tagged mutagenesis. Proc Natl Acad Sci U S A 102:3805-3810
Stewart JP, Usherwood EJ, Ross A, Dyson H, Nash T (1998) Lung epithelial cells are a major site of murine gammaherpesvirus persistence. J Exp Med 187:1941-1951
Sunil-Chandra NP, Efstathiou S, Arno J, Nash AA (1992) Virological and pathological features of mice infected with murine gamma-herpesvirus 68. J Gen Virol 73 (Pt 9):2347-2356
Sunil-Chandra NP, Efstathiou S, Nash AA (1992) Murine gammaherpesvirus 68 establishes a latent infection in mouse B lymphocytes in vivo. J Gen Virol 73 (Pt 12):3275-3279
Sunil-Chandra NP, Arno J, Fazakerley J, Nash AA (1994) Lymphoproliferative disease in mice infected with murine gammaherpesvirus 68. Am J Pathol 145:818-826
Taylor SL, Kinchington PR, Brooks A, Moffat JF (2004) Roscovitine, a cyclin-dependent kinase inhibitor, prevents replication of varicella-zoster virus. J Virol 78:2853-2862
Townsley AC, Dutia BM, Nash AA (2004) The m4 gene of murine gammaherpesvirus modulates productive and latent infection in vivo. J Virol 78:758-767
Wang SK, Duh CY, Chang TT (2000) Cloning and identification of regulatory gene UL76 of human cytomegalovirus. J Gen Virol 81:2407-2416
Weck KE, Kim SS, Virgin HI, Speck SH (1999) Macrophages are the major reservoir of latent murine gammaherpesvirus 68 in peritoneal cells. J Virol 73:3273-3283
Weck KE, Kim SS, Virgin HI, Speck SH (1999) B cells regulate murine gammaherpesvirus 68 latency. J Virol 73:4651-4661
Acknowledgments
This work was supported by Fundação Portuguesa para a Ciência e Tecnologia (POCI/SAU-MMO/59444/2004). Nascimento R. was the receipient of a fellowship from Fundação Portuguesa para a Ciência e Tecnologia (SFRH/BD/4853/2001)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nascimento, R., Costa, H., Dias, J.D. et al. MHV-68 Open Reading Frame 20 is a nonessential gene delaying lung viral clearance. Arch Virol 156, 375–386 (2011). https://doi.org/10.1007/s00705-010-0862-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-010-0862-2