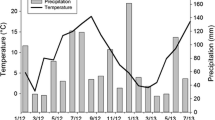
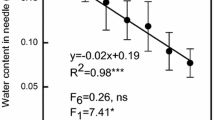
Abstract
Conifer needles are extraordinarily variable and much of this diversity is linked to the water transport capacity of the xylem and to xylem conduit properties. However, we still know little about how anatomical characteristics influence the hydraulic efficiency of needle xylem in different parts of the crown. In this study we evaluated needle function and anatomy in Norway spruce families exposed to different light conditions. We measured tracheid and needle characteristics of sun-exposed and shaded current-year needles in two experimental plots: a control plot and a thinned plot with 50% reduction in stand density. Sun-exposed needles had a larger tracheid lumen area than shaded needles, and this was caused by a larger maximum tracheid lumen diameter, while the minimum lumen diameter was less plastic. Sun-exposed needles had also higher theoretical hydraulic conductivity than shaded needles. Thinning leads to increased radiation to the lower branches, and presumably exposes the upper branches to stronger water stress than before thinning. Thinning affected several needle parameters both in sun-exposed and shaded needles; tracheid lumens were more circular and minimum tracheid lumen diameter was larger in the thinned plot, whereas maximum tracheid lumen diameter was less plastic on both plots. This study demonstrates that needle xylem structure in Norway spruce is clearly influenced by the light gradient within the tree crown.




Similar content being viewed by others
References
Alavi G, Jansson PE, Haellgren JE, Bergholm J (2001) Interception of a dense spruce forest—performance of a simplified canopy water balance model. Nord Hydrol 32:265–284
Aloni R (1987) Differentiation of vascular tissues. Annu Rev Plant Physiol 38:179–204
Boyer JS (1968) Relationship of water potential to growth of leaves. Plant Physiol 43:1056–1062
Brodribb TJ, Holbrook NM, Edwards EJ, Gutiérrez MV (2003) Relations between stomatal closure, leaf turgor and xylem vulnerability in eight tropical dry forest trees. Plant Cell Environ 26:443–450
Čermák J, Deml M, Penka M (1973) A new method of sap flow rate determination in trees. Biol Plant 15:171–178
Cochard H, Froux F, May S, Coutand C (2004) Xylem collapse in water-stressed pine needles. Plant Physiol 134:401–408
Cruiziat P, Cochard H, Améglio T (2002) Hydraulic architecture of trees: main concepts and results. Ann For Sci 59:723–752
Domec JC, Lachenbruch B, Meinzer FC (2006) Bordered pit structure and function determine spatial patterns of air-seeding thresholds in xylem of Douglas-fir (Pseudotsuga menziesii; Pinaceae) trees. Am J Bot 93:1588–1600
Domec JC, Palmroth S, Ward E, Maier CA, Thérézien M, Oren R (2009) Acclimation of leaf hydraulic conductance and stomatal conductance of Pinus taeda (loblolly pine) to long term growth in elevated CO2 (FACE) and N-fertilization. Plant Cell Environ 32:1500–1512
Froux F, Huc R, Ducrey M, Dreyer E (2002) Xylem hydraulic efficiency versus vulnerability in seedlings of four contrasting Mediterranean tree species (Cedrus atlantica, Cupressus sempervirens, Pinus halepensis and Pinus nigra). Ann For Sci 59:409–418
Gebauer R, Volařík D, Urban J, Børja I, Nagy NE, Eldhuset TD, Krokene P (2011) Effect of thinning on the anatomical adaptation of Norway spruce needles. Tree Physiol. doi:10.1093/treephys/tpr081
Groninger JW, Seiler JR, Peterson JA, Kreh RE (1996) Growth and photosynthetic responses of four Virginia Piedmont tree species to shade. Tree Physiol 16:773–778
Hacke UG, Sperry JS (2001) Functional and ecological xylem anatomy. Plant Ecol Evol Syst 4:97–115
Hacke UG, Sperry JS, Pittermann J (2005) Efficiency versus safety tradeoffs for water conduction in angiosperm vessels versus gymnosperm tracheids. In: Holbrook NM, Zwieniecki MA (eds) Vascular transport in plants. Elsevier Academic Press, USA, pp 333–354
Hansen VT, Grimenes AA (2003) Meteorologiske data for Ås 2002. Institutt for tekniske fag, Norges landbrukshogskole
Hinckley TM, Lassoie JP, Running SW (1978) Temporal and spatial variations in the water status of forest trees. For Sci Monogr 20:1–72
Johnson DM, Meinzer FC, Woodruff DR, McCulloh KA (2009) Leaf xylem embolism, detected acoustically and by cryo-SEM, corresponds to decreases in leaf hydraulic conductance in four evergreen species. Plant Cell Environ 32:828–836
Kirkham MB, Gardner WR, Gerloff GC (1972) Regulation of cell division and cell enlargement by turgor pressure. Plant Physiol 49:961–962
Koch GW, Sillett SC, Jennings GM, Davis SD (2004) The limits to tree height. Nature 428:851–854
Kučera J, Čermák J, Penka M (1977) Improved thermal method of continual recording the transpiration flow rate dynamics. Biol Plant 19:413–420
Lagergren F, Lindroth A (2004) Variation in sapflow and stem growth in relation to tree size, competition and thinning in a mixed forest of pine and spruce in Sweden. Forest Ecol Manag 188:51–63
Lhotáková Z, Albrechtová J, Malenovský Z, Rock BN, Polák T, Cudlín P (2007) Does the azimuth orientation of Norway spruce (Picea abies/L./Karst.) branches within sunlit crown part influence the heterogeneity of biochemical, structural and spectral characteristics of needles? Environ Exp Bot 59:283–292
Martre P, Durand JL, Cochard H (2000) Changes in axial hydraulic conductivity along elongating leaf blades in relation to xylem maturation in tall feste. New Phytol 146:235–247
Messier C, Doucet R, Ruel JC, Claveau Y, Kelly C, Lechowicz MJ (1999) Functional ecology of advance regeneration in relation to light in boreal forests. Can J For Res 29:812–823
Mitchell AK (1998) Acclimation of Pacific yew (Taxus brevifolia) foliage to sun and shade. Tree Physiol 18:749–757
Morén AS, Lindroth A, Flower-Ellis J, Cienciala E, Mölder M (2000) Branch transpiration of pine and spruce scaled to tree and canopy using needle biomass distributions. Trees 14:384–397
Murtuagh PA (2007) Simplicity and complexity in ecological data analysis. Ecology 88:56–62
Nardini A, Salleo S (2000) Limitation of stomatal conductance by hydraulic traits: sensing or preventing xylem cavitation? Trees 15:14–24
Nardini A, Gortan E, Salleo S (2005) Hydraulic efficiency of the leaf venation system in sun- and shade-adapted species. Funct Plant Biol 32:953–961
Němec B, Bartoš J, Hršel I, Chaloupka J, Lhotský O, Luxová M, Milovidov P, Nečásek J, Pazourková Z, Pazourek J, Sosnová V (1962) Botanical microtechnic (in Czech). Československá Akademie Věd, Prague, pp 60–414
Niinemets Ü (1997) Distribution patterns of foliar carbon and nitrogen as affected by tree dimensions and relative light conditions in the canopy of Picea abies. Trees 11:144–154
Niinemets Ü, Kull O (1995) Effects of light availability and tree size on the architecture of assimilative surface in the canopy of Picea abies: variation in needle morphology. Tree Physiol 15:307–315
Niinemets Ü, Ellsworth DS, Lukjanova A, Tobias M (2001) Site fertility and the morphological and photosynthetic acclimation of Pinus sylvestris needles to light. Tree Physiol 21:1231–1244
Niinemets Ü, Lukjanova A, Turnbull MH, Sparrow AD (2007) Plasticity in mesophyll volume fraction modulates light-acclimation in needle photosynthesis in two pines. Tree Physiol 27:1137–1151
Nobel PS (2005) Physicochemical and environmental plant physiology. Elsevier Academic Press, USA, pp 446–454
Pothier D, Margolis HA, Waring RH (1989) Patterns of change in saturated sapwood permeability and sapwood conductance with stand development. Can J For Res 19:432–439
Protz CG, Silins U, Lieffers VJ (2000) Reduction in branch sapwood hydraulic permeability as a factor limiting survival of lower branches of lodgepole pine. Can J For Res 30:1088–1095
R Development Core Team (2010) R: a language and environment for statistical computing
Reich PB, Tjoelker MG, Walters MB, Vanderklein DW, Bushena C (1998) Close association of RGR, leaf and root morphology, seed mass and shade tolerance in seedlings of nine boreal tree species grown in high and low light. Funct Ecol 12:327–338
Sack L, Holbrook NM (2006) Leaf hydraulics. Annu Rev Plant Biol 57:361–381
Sack L, Melcher PJ, Zwieniecki MA, Holbrook NM (2002) The hydraulic conductance of the angiosperm leaf lamina: a comparison of three measurement methods. J Exp Bot 53:2177–2184
Sack L, Cowan PD, Jaikumar N, Holbrook NM (2003) The ‘hydrology’ of leaves: co-ordination of structure and function in temperate woody species. Plant Cell Environ 26:1343–1356
Sack L, Tyree MT, Holbrook NM (2005) Leaf hydraulic architecture correlates with regeneration irradiance in tropical rainforest trees. New Phytol 167:403–413
Schultz HR, Matthews MA (1993) Xylem development and hydraulic conductance in sun and shade shoots of grapevine (Vitis vinifera L.): evidence that low light uncouples water transport capacity from leaf area. Planta 190:393–406
Sellin A (1993) Resistance to water flow in xylem of Picea abies (L.) Karst. trees grown under contrasting light conditions. Trees 7:220–226
Sellin A (2001) Morphological and stomatal responses of Norway spruce foliage to irradiance within a canopy depending on shoot age. Environ Exp Bot 45:115–131
Sellin A, Kupper P (2004) Within-crown variation in leaf conductance of Norway spruce: effects of irradiance, vapour pressure deficit, leaf water status and plant hydraulic constraints. Ann For Sci 61:419–429
Sperry J, Hacke U, Pitterman J (2006) Size and function in conifer tracheids and angiosperm vessels. Am J Bot 93:1490–1500
Stenberg P, Smolander H, Sprugel DG, Smolander S (1998) Shoot structure, light interception, and distribution of nitrogen in an Abies amabilis canopy. Tree Physiol 18:759–767
Tyree MT, Zimmermann MH (2002) Xylem structure and the ascent of sap. Springer, Berlin
Tyree MT, Nardini A, Salleo S, Sack L, El Omari B (2005) The dependence of leaf hydraulic conductance on irradiance during HPFM measurements: any role for stomatal response? J Exp Bot 56:737–744
Woodruff DR, McCulloh KA, Warren JM, Meinzer FC, Lachenbruch B (2007) Impacts of tree height on leaf hydraulic architecture and stomatal control in Douglas-fir. Plant Cell Environ 30:559–569
Woodruff DR, Meizer FC, Lachenbruch B (2008) Height-related trends in leaf xylem anatomy and shoot hydraulic characteristics in a tall conifer: safety versus efficiency in water transport. New Phytol 180:90–99
Yang SD, Tyree MT (1994) Hydraulic architecture of Acer saccharum and A. rubrum: comparison of branches to whole trees and the contribution of leaves to hydraulic resistance. J Exp Bot 45:179–186
Youngblood A, Ferguson DE (2003) Changes in needle morphology of shade-tolerant seedlings after partial overstory canopy removal. Can J Forest Res 33:1315–1322
Zwieniecki MA, Hutyra L, Thompson MV, Holbrook NM (2000) Dynamic changes in petiole specific conductivity in red maple (Acer rubrum L.), tulip tree (Liriodendron tulipifera L.) and northern fox grape (Vitis labrusca L.). Plant Cell Environ 23:407–414
Zwieniecki MA, Stone HA, Leigh A, Boyce CK, Holbrook NM (2006) Hydraulic design of pine needles: one-dimensional optimization of single-vein leaves. Plant Cell Environ 29:803–809
Acknowledgments
This work was funded from the Czech Ministry of Education (grant no. 6215648902), from Iceland, Liechtenstein and Norway through the EEA Financial Mechanism together with the Norwegian Financial Mechanism (grant no. A/CZ0046/2/0009) and from Mendel University in Brno (grant IGA 12/2010). We are also grateful to Jaromíra Dreslerová, Darina Smerekovská and Alžběta Malotínová for laboratory work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Zwieniecki.
Rights and permissions
About this article
Cite this article
Gebauer, R., Volařík, D., Urban, J. et al. Effects of different light conditions on the xylem structure of Norway spruce needles. Trees 26, 1079–1089 (2012). https://doi.org/10.1007/s00468-012-0684-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-012-0684-9