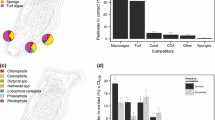
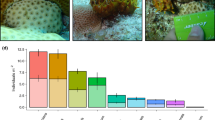
Abstract
The community structure of sedentary organisms is largely controlled by the outcome of direct competition for space. Understanding factors defining competitive outcomes among neighbors is thus critical for predicting large-scale changes, such as transitions to alternate states within coral reefs. Using a spatially explicit model, we explored the importance of variation in two spatial properties in benthic dynamics on coral reefs: (1) patterns of herbivory are spatially distinct between fishes and sea urchins and (2) there is wide variation in the areal extent into which different coral species can expand. We reveal that the size-specific, competitive asymmetry of corals versus fleshy algae highlights the significance of spatial patterning of herbivory and of coral growth. Spatial dynamics that alter the demographic importance of coral recruitment and maturation have profound effects on the emergent structure of the reef benthic community. Spatially constrained herbivory (as by sea urchins) is more effective than spatially unconstrained herbivory (as by many fish) at opening space for the time needed for corals to settle and to recruit to the adult population. Further, spatially unconstrained coral growth (as by many branching coral species) reduces the number of recruitment events needed to fill a habitat with coral relative to more spatially constrained growth (as by many massive species). Our model predicts that widespread mortality of branching corals (e.g., Acropora spp) and herbivorous sea urchins (particularly Diadema antillarum) in the Caribbean has greatly reduced the potential for restoration across the region.




Similar content being viewed by others
References
Adler PB, Raff DA, Lauenroth WK (2001) The effect of grazing on the spatial heterogeneity of vegetation. Oecologia 128:465–479
Aronson RB, Macintyre IG, Precht WF, Murdoch TJT, Wapnick CM (2002) The expanding scale of species turnover events on coral reefs in Belize. Ecol Monogr 72:233–249
Bak RPM, Meesters EH (1999) Population structure as a response of coral communities to global change. Am Zool 39:56–65
Bak RPM, Nieuwland G (1995) Long-term change in coral communities along depth gradients over leeward reefs in the Netherlands Antilles. Bull Mar Sci 56:609–619
Baums IB, Miller MW, Hellberg ME (2006) Geographic variation in clonal structure in a reef-building Caribbean coral, Acropora palmata. Ecol Appl 76:503–519
Bellwood DR, Hughes TP, Folke C, Nystrom M (2004) Confronting the coral reef crisis. Nature 429:827–833
Birkeland C (2004) Ratcheting down the coral reefs. Bioscience 54:1021–1027
Boyer KE, Fong P, Armitage AR, Cohen RA (2004) Elevated nutrient content of tropical macroalgae increases rates of herbivory in coral, seagrass, and mangrove habitats. Coral Reefs 23:530–538
Burkepile DE, Hay ME (2008) Herbivore species richness and feeding complementarity affect community structure and function on a coral reef. Proc Natl Acad Sci USA 105:16201–16206
Burkepile DE, Hay ME (2010) Impact of herbivore identity on algal succession and coral growth on a Caribbean reef. PLoS ONE 5:e8963
Buss LW, Jackson JBC (1979) Competitive networks: nontransitive competitive relationships in cryptic coral reef environments. Am Nat 113:223–234
Carpenter RC (1984) Predator and population density control of homing behavior in the Caribbean echinoid Diadema antillarum. Mar Biol 82:101–108
Carpenter RC (1986) Partitioning herbivory and its effects on coral reef algal communities. Ecol Monogr 56:346–363
Carpenter RC (1988) Mass mortality of a Caribbean sea urchin: immediate effects on community metabolism and other herbivores. Proc Natl Acad Sci USA 85:511–514
Carpenter RC (1990) Mass mortality of Diadema antillarum. Mar Biol 104:67–77
Carpenter RC, Edmunds PJ (2006) Local and regional scale recovery of Diadema promotes recruitment of scleractinian corals. Ecol Lett 9:268–277
Connell JH (1971) On the role of natural enemies in preventing competitive exclusion in some marine animals and in rain forest trees. In: Den Boer PJ, Gradwell GR (eds) Dynamics of populations. Center for Agricultural Publication and Documentation, Wageningen, pp 298–312
Connell JH (1997) Disturbance and recovery of coral assemblages. Coral Reefs 16:S101–S113
Diaz-Pulido G, McCook LJ (2002) The fate of bleached corals: patterns and dynamics of algal recruitment. Mar Ecol Prog Ser 232:115–128
Done TJ (1992) Phase shifts in coral reef communities and their ecological significance. Hydrobiologia 247:121–132
Dulvy NK, Freckleton RP, Polunin NVC (2004) Coral reef cascades and the indirect effects of predator removal by exploitation. Ecol Lett 7:410–416
Edmunds PJ, Carpenter RC (2001) Recovery of Diadema antillarum reduces macroalgal cover and increases abundance of juvenile corals on a Caribbean reef. Proc Natl Acad Sci USA 98:5067–5071
Edmunds PJ, Bruno JF, Carlon DB (2004) Effects of depth and microhabitat on growth and survivorship of juvenile corals in the Florida keys. Mar Ecol Prog Ser 278:115–124
Graham NAJ, Nash KL, Kool JT (2011) Coral reef recovery dynamics in a changing world. Coral Reefs 30:283–294
Gross EM (2003) Allelopathy of aquatic autotrophs. Crit Rev Plant Sci 22:313–339
Grubb PJ (1977) The maintenance of species-richness in plant communities: the importance of the regeneration niche. Biol Rev 52:107–145
Harper JL (1977) Population biology of plants. Academic, New York
Harrington L, Fabricius K, De’ath G, Negri A (2004) Recognition and selection of settlement substrata determine post-settlement survival in corals. Ecology 85:3428–3437
Hoey AS, Bellwood DR (2008) Cross-shelf variation in the role of parrotfishes on the Great Barrier Reef. Coral Reefs 27:37–47
Horn HS (1975) Markovian properties of forest succession. In: Cody ML, Diamond JM (eds) Ecology and evolution of communities. Belknap, Cambridge, pp 196–211
Hughes TP (1984) Population dynamics based on individual size rather than age: a general model with a reef coral example. Am Nat 123:778–795
Hughes TP, Reed DC, Boyle M-J (1987) Herbivory on coral reefs: community structure following mass mortalities of sea urchins. J Exp Mar Biol Ecol 113:39–59
Hughes TP et al (2007) Phase shifts, herbivory, and the resilience of coral reefs to climate change. Curr Biol 17:360–365
Hughes TP, Graham NAJ, Jackson JBC, Mumby PJ, Steneck RS (2010) Rising to the challenge of sustaining coral reef resilience. Trends Ecol Evol 25:633–642
Huston M (1985) Variation in coral growth rates with depth at Discovery Bay, Jamaica. Coral Reefs 4:19–25
Idjadi JA, Haring RN, Precht WF (2010) Recovery of the sea urchin Diadema antillarum promotes scleractinian coral growth and survivorship on shallow Jamaican reefs. Mar Ecol Prog Ser 403:91–100
Jackson JBC (1977) Competition on marine hard substrata: the adaptive significance of solitary and colonial strategies. Am Nat 111:743–767
Jackson JBC (1979) Morphological strategies of sessile animals. In: Larwood G, Rosen BR (eds) Biology and systematics of colonial organisms. Academic, New York, pp 499–555
Janzen DH (1970) Herbivores and the number of tree species in tropical forests. Am Nat 104:501–528
Knowlton N (1992) Thresholds and multiple stable states in coral reef community dynamics. Am Zool 32:674–682
Kuffner IB, Walters LJ, Becerro MA, Paul VJ, Ritson-Williams R, Beach KS (2006) Inhibition of coral recruitment by macroalgae and cyanobacteria. Mar Ecol Prog Ser 323:107–117
Levins R, Culver D (1971) Regional coexistence of species and competition between rare species. Proc Natl Acad Sci USA 68:1246–1248
Lewis SM (1986) The role of herbivorous fishes in the organization of a Caribbean reef community. Ecol Monogr 56:183–200
Lirman D (2001) Competition between macroalgae and corals: effects of herbivore exclusion and increased algal biomass on coral survivorship and growth. Coral Reefs 19:392–399
May RM (1977) Thresholds and breakpoints in ecosystems with a multiplicity of stable states. Nature 269:471–477
McClanahan TR, Shafir SH (1990) Causes and consequences of sea urchin abundance and diversity in Kenyan coral reef lagoons. Oecologia 83:362–370
McClanahan TR, Cokos BA, Sala E (2002) Algal growth and species composition under experimental control of herbivory, phosphorus and coral abundance in Glovers Reef, Belize. Mar Pollut Bull 44:441–451
McClanahan TR et al (2003) Interaction between nutrients and herbivory in controlling algal communities and coral condition on Glover’s Reef, Belize. Mar Ecol Prog Ser 261:135–147
McCook LJ, Jompa J, Diaz-Pulido G (2001) Competition between corals and algae on coral reefs: a review of evidence and mechanisms. Coral Reefs 19:400–417
Meesters EH, Wesseling I, Bak RPM (1996) Partial mortality in three species of reef-building corals and the relation with colony morphology. Bull Mar Sci 58:838–852
Mumby PJ (2009) Phase shifts and the stability of macroalgal communities on Caribbean coral reefs. Coral Reefs 28:761–773
Mumby PJ, Harborne AR (2010) Marine reserves enhance the recovery of corals on Caribbean reefs. PLoS ONE 5:e8657
Mumby PJ, Wabnitz CCC (2002) Spatial patterns of aggression, territory size, and harem size in five sympatric Caribbean parrotfish species. Environ Biol Fish 63:265–279
Mumby PJ, Hastings A, Edwards HJ (2007) Thresholds and the resilience of Caribbean coral reefs. Nature 450:98–101
Myhre S, Acevedo-Gutiérrez A (2007) Recovery of sea urchin Diadema antillarum populations is correlated to increased coral and reduced macroalgal cover. Mar Ecol Prog Ser 329:205–210
Newman MJH, Paredes GA, Sala E, Jackson JBC (2006) Structure of Caribbean coral reef communities across a large gradient of fish biomass. Ecol Lett 9:1216–1227
Nugues MM, Smith GW, Hooidonk RJ, Seabra MI, Bak RPM (2004) Algal contact as a trigger for coral disease. Ecol Lett 7:919–923
Ogden JC (1976) Some aspects of herbivore-plant relationships on Caribbean reefs and seagrass beds. Aquat Bot 2:103–116
Ogden JC, Brown RA, Salesky N (1973) Grazing by the echinoid Diadema antillarum Philippi: formation of halos around West Indian patch reefs. Science 182:715–717
Prins HHT, van der Jeugd HP (1993) Herbivore population crashes and woodland structure in East Africa. J Ecol 81:305–314
Randall JE (1961) Overgrazing of algae by herbivorous marine fishes. Ecology 42:812
Rasher DB, Hay ME (2010) Chemically rich seaweeds poison corals when not controlled by herbivores. Proc Natl Acad Sci USA 107:9683–9688
Rinkevich B, Loya Y (1986) Senescence and dying signals in a reef building coral. Experientia 42:320–322
Scheffer M, Hosper SH, Meijer M-L, Moss B, Jeppesen E (1993) Alternative equilibria in shallow lakes. Trends Ecol Evol 8:275–279
Selig ER, Bruno JF (2010) A global analysis of the effectiveness of marine protected areas in preventing coral loss. PLoS ONE 5:e9278
Semmens BX, Brumbaugh DR, Drew JA (2005) Interpreting space use and behavior of blue tang, Acanthurus coeruleus, in the context of habitat, density, and intra-specific interactions. Environ Biol Fish 74:99–107
Simenstad CA, Estes JA, Kenyon KW (1978) Aleuts, sea otters, and alternate stable-state communities. Science 200:403–411
Smith JE et al (2006) Indirect effects of algae on coral: algae-mediated, microbe-induced coral mortality. Ecol Lett 9:835–845
Soong K (1993) Colony size as a species character in massive reef corals. Coral Reefs 12:77–83
Sousa WP, Schroeter SC, Gaines SD (1981) Latitudinal variation in intertidal algal community structure: the influence of grazing and vegetative propagation. Oecologia 48:297–307
Tanner JE (1995) Competition between scleractinian corals and macroalgae: An experimental investigation of coral growth, survival and reproduction. J Exp Mar Biol Ecol 190:151–168
van Rooij JM, Kok JP, Videler JJ (1996) Local variability in population structure and density of the protogynous reef herbivore Sparisoma viride. Environ Biol Fish 47:65–80
Vermeij MJA (2006) Early life-history dynamics of Caribbean coral species on artificial substratum: the importance of competition, growth and variation in life-history strategy. Coral Reefs 25:59–71
Vermeij MJA, Sandin SA (2008) Density-dependent settlement and mortality structure the earliest life phases of a coral population. Ecology 89:1994–2004
Wolfram S (1984) Universality and complexity in cellular automata. Phys D: Nonlinear Phenom 10:1–35
Acknowledgments
We extend thanks to F. Rohwer for motivation, M.J.A. Vermeij for ideas and insights, and J.E. Smith, G.J. Williams, and C.S. Dugas for candid advice. E. Scripps provided invaluable support enabling this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jeff Shima.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sandin, S.A., McNamara, D.E. Spatial dynamics of benthic competition on coral reefs. Oecologia 168, 1079–1090 (2012). https://doi.org/10.1007/s00442-011-2156-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-011-2156-0