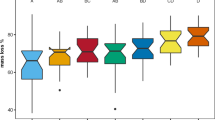
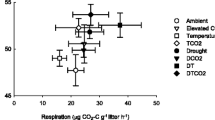
Abstract
Increasing atmospheric CO2 and temperature are predicted to alter litter decomposition via changes in litter chemistry and environmental conditions. The extent to which these predictions are influenced by biotic factors such as litter species composition or decomposer activity, and in particular how these different factors interact, is not well understood. In a 5-week laboratory experiment we compared the decomposition of leaf litter from four temperate tree species (Fagus sylvatica, Quercus petraea, Carpinus betulus and Tilia platyphyllos) in response to four interacting factors: elevated CO2-induced changes in litter quality, a 3°C warmer environment during decomposition, changes in litter species composition, and presence/absence of a litter-feeding millipede (Glomeris marginata). Elevated CO2 and temperature had much weaker effects on decomposition than litter species composition and the presence of Glomeris. Mass loss of elevated CO2-grown leaf litter was reduced in Fagus and increased in Fagus/Tilia mixtures, but was not affected in any other leaf litter treatment. Warming increased litter mass loss in Carpinus and Tilia, but not in the other two litter species and in none of the mixtures. The CO2- and temperature-related differences in decomposition disappeared completely when Glomeris was present. Overall, fauna activity stimulated litter mass loss, but to different degrees depending on litter species composition, with a particularly strong effect on Fagus/Tilia mixtures (+58%). Higher fauna-driven mass loss was not followed by higher C mineralization over the relatively short experimental period. Apart from a strong interaction between litter species composition and fauna, the tested factors had little or no interactive effects on decomposition. We conclude that if global change were to result in substantial shifts in plant community composition and macrofauna abundance in forest ecosystems, these interacting biotic factors could have greater impacts on decomposition and biogeochemical cycles than rising atmospheric CO2 concentration and temperature.


Similar content being viewed by others
References
Aerts R (2006) The freezer defrosting: global warming and litter decomposition rates in cold biomes. J Ecol 94:713–724
Bardgett RD (2005) The biology of soil. A community and ecosystem approach. Oxford University Press, New York
Bradley KL, Pregitzer KS (2007) Ecosystem assembly and terrestrial carbon balance under elevated CO2. Trends Ecol Evol 22:538–547
Chapin FS III, Matson PA, Mooney HA (2002) Principles of terrestrial ecosystem ecology. Springer, New York
Chung H, Zak DR, Lilleskov EA (2006) Fungal community composition and metabolism under elevated CO2 and O3. Oecologia 147:143–154
Chung H, Zak DR, Reich PB, Ellsworth DS (2007) Plant species richness, elevated CO2 and atmospheric nitrogen deposition alter soil microbial community composition and function. Global Change Biol 13:980–989
Cornelissen JHC (1996) An experimental comparison of leaf decomposition rates in a wide range of temperate plant species and types. J Ecol 84:573–582
Cornwell WK, Cornelissen JHC, Amatangelo K, et al. (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071
Cotrufo MF, Ineson P (1996) Elevated CO2 reduces field decomposition rates of Betula pendula Roth. leaf litter. Oecologia 106:525–530
Cotrufo MF, Briones MJI, Ineson P (1998) Elevated CO2 affects field decomposition rate and palatability of tree leaf litter: importance of changes in substrate quality. Soil Biol Biochem 30:1565–1571
Cotrufo MF, De Angelis P, Polle A (2005) Leaf litter production and decomposition in a poplar short-rotation coppice exposed to free air CO2 enrichment (POPFACE). Global Change Biol 11:971–982
Coûteaux MM, Mousseau M, Célérier ML, Bottner P (1991) Increased atmospheric CO2 and litter quality: decomposition of sweet chestnut leaf litter with animal food webs of different complexities. Oikos 61:54–64
Coûteaux MM, Bottner P, Berg B (1995) Litter decomposition, climate and litter quality. Trends Ecol Evol 10:63–66
Coûteaux MM, Aloui A, Kurz-Besson C (2002) Pinus halepensis litter decomposition in laboratory microcosms as influenced by temperature and a millipede, Glomeris marginata. Appl Soil Ecol 20:85–96
David JF (1999) Abundance, biomass and functional structure of the saprophagous macrofauna in the litter and soil of Mediterrenean oak forests. Pedobiologia 43:319–327
David JF, Gillon D (2002) Annual feeding rate of the millipede Glomeris marginata on holm oak (Quercus ilex) leaf litter under Mediterranean conditions. Pedobiologia 46:42–52
David JF, Gillon D (2009) Combined effects of elevated temperatures and reduced leaf litter quality on the life-history parameters of a saprophagous macroarthropod. Global Change Biol 15:156–165
David JF, Malet N, Coûteaux MM, Roy J (2001) Feeding rates of the woodlouse Armadillidium vulgare on herb litters produced at two levels of atmospheric CO2. Oecologia 127:343–349
Epps KY, Comerford NB, Reeves JB, Cropper WP, Araujo QR (2007) Chemical diversity—highlighting a species richness and ecosystem function disconnect. Oikos 116:1831–1840
Fierer N, Craine JM, McLauchlan K, Schimel JP (2005) Litter quality and the temperature sensitivity of decomposition. Ecology 86:320–326
Finzi AC, Allen AS, DeLucia EH, Ellsworth DS, Schlesinger WH (2001) Forest litter production, chemistry and decomposition following two years of free air CO2 enrichment. Ecology 82:470–484
Gartner TB, Cardon ZG (2004) Decomposition dynamics in mixed-species leaf litter. Oikos 104:230–246
Hagedorn F, Machwitz M (2007) Controls on dissolved organic matter leaching from forest litter grown under elevated CO2. Soil Biol Biochem 39:1759–1769
Hättenschwiler S, Gasser P (2005) Soil animals alter plant litter diversity effects on decomposition. Proc Natl Acad Sci USA 102:1519–1524
Hättenschwiler S, Bühler S, Körner C (1999) Quality, decomposition and isopod consumption of tree litter produced under elevated CO2. Oikos 85:271–281
Hättenschwiler S, Tiunov AV, Scheu S (2005) Biodiversity and litter decomposition in terrestrial ecosystems. Annu Rev Ecol Evol Syst 36:191–218
Heal OW, Anderson JM, Swift MJ (1997) Plant litter quality and decomposition: an historical overview. In: Cadisch G, Giller KE (eds) Driven by nature. CAB International, Wallingford, pp 3–30
Hector A, Bagchi R (2007) Biodiversity and ecosystem multifunctionality. Nature 448:188–190
Hedde M, Bureau F, Akpa-Vinceslas M, Aubert M, Decaëns T (2007) Beech leaf degradation in laboratory experiments: effects of eight detritivorous invertebrate species. Appl Soil Ecol 35:291–301
Heemsbergen DA, Berg MP, Loreau M, Van Hal JR, Faber JH, Verhoef HA (2004) Biodiversity effects on soil processes explained by interspecific functional dissimilarity. Science 306:1019–1020
Hoorens B, Aerts R, Stroetenga M (2003) Does initial litter chemistry explain litter mixture effects on decomposition? Oecologia 137:578–586
Hyvönen R, Agren GI, Linder S, et al. (2007) The likely impact of elevated [CO2], nitrogen deposition, increased temperature and management on carbon sequestration in temperate and boreal forest ecosystems: a literature review. New Phytol 173:463–480
IPCC (2007) Climate change 2007: the physical science basis. In: Solomon S, Qin D, Manning M, et al. (eds) Contribution of Working Group I to the fourth assessment report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge
Körner C (2000) Biosphere responses to CO2 enrichment. Ecol Appl 10:1590–1619
Körner C, Asshoff R, Bignucolo O, et al. (2005) Carbon flux and growth in mature deciduous forest trees exposed to elevated CO2. Science 309:1360–1362
Lavelle P, Spain AV (2001) Soil ecology. Kluwer, London
Loranger G, Ponge JF, Imbert D, Lavelle P (2002) Leaf decomposition in two semi-evergreen tropical forests: influence of litter quality. Biol Fertil Soils 35:247–252
Loreau M, Naeem S, Inchausti P, et al. (2001) Biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808
Maraun M, Scheu S (1996) Changes in microbial biomass, respiration and nutrient status of beech (Fagus sylvatica) leaf litter processed by millipedes (Glomeris marginata). Oecologia 107:131–140
Midgley GF, Thuiller W, Higgins SI (2007) Plant species migration as a key uncertainty in predicting future impacts of climate change on ecosystems: progress and challenges. In: Canadell JG, Pataki DE, Pitelka LF (eds) Terrestrial ecosystems in a changing world. Springer, Berlin, pp 129–137
Norby RJ, Cotrufo MF, Ineson P, O’Neill EG, Canadell JG (2001) Elevated CO2, litter chemistry, and decomposition: a synthesis. Oecologia 127:153–165
Parsons WJF, Bockheim JG, Lindroth RL (2008) Independent, interactive and species-specific responses of leaf litter decomposition to elevated CO2 and O3 in a northern hardwood forest. Ecosystems 11:505–519
Pendall E, Bridgham S, Hanson PJ, et al. (2004) Below-ground process responses to elevated CO2 and temperature: a discussion of observations, measurement methods, and models. New Phytol 162:311–322
Pepin S, Körner C (2002) Web-FACE: a new canopy free-air CO2 enrichment system for tall trees in mature forests. Oecologia 133:1–9
Pérez-Harguindeguy N, Díaz S, Cornelissen JHC, Vendramini F, Cabido M, Castellanos A (2000) Chemistry and toughness predict leaf litter decomposition rates over a wide spectrum of functional types and taxa in central Argentina. Plant Soil 218:21–30
Pérez-Harguindeguy N, Blundo CM, Gurvich DE, Díaz S, Cuevas E (2008) More than the sum of its parts? Assessing litter heterogeneity effects on the decomposition of litter mixtures through leaf chemistry. Plant Soil 303:151–159
Robinson CH (2002) Controls on decomposition and soil nitrogen availability at high latitudes. Plant Soil 242:65–81
Sala OE, Chapin FS, Armesto JJ, et al. (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–17774
Saugier B, Roy J, Mooney HA (2001) Estimation of global terrestrial productivity: converging toward a single number? In: Roy J, Saugier B, Mooney HA (eds) Terrestrial global productivity. Academic Press, London, pp 543–557
Sayer EJ (2006) Using experimental manipulation to assess the roles of leaf litter in the functioning of forest ecosystems. Biol Rev 81:1–31
Schaefer M (1991) Secondary production and decomposition. In: Röhrig E, Ulrich B (eds) Ecosystems of the world 7. Temperate deciduous forests. Elsevier, Amsterdam, pp 175–218
Schimel JP, Hättenschwiler S (2007) Nitrogen transfer between decomposing leaves of different N status. Soil Biol Biochem 39:1428–1436
R Development Core Team (2005) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. ISBN 3-900051-07-0. http://www.R-project.org
Thormann MN, Bayley SE, Currah RS (2004) Microcosm tests of the effects of temperature and microbial species number on the decomposition of Carex aquatilis and Sphagnum fuscum litter from southern boreal peatlands. Can J Microbiol 50:793–802
Wall DH, Bradford MA, St. John MG, et al. (2008) Global decomposition experiment shows soil animal impacts on decomposition are climate-dependent. Global Change Biol 14:2661–2677
Walther GR (2004) Plants in a warmer world. Perspectives in plant ecology evolution and systematics 6:169–185
Wan S, Norby RJ, Ledford J, Weltzin JF (2007) Responses of soil respiration to elevated CO2, air warming, and changing soil water availability in a model old-field grassland. Global Change Biol 13:2411–2424
Wardle D (2002) Communities and ecosystems. Linking the aboveground and belowground components. Princeton University Press, Princeton
Wolters V (2000) Invertebrate control of soil organic matter stability. Biol Fertil Soils 31:1–19
Zimmer M, Kautz G, Topp W (2005) Do woodlice and earthworms interact synergistically in leaf litter decomposition? Funct Ecol 19:7–16
Acknowledgements
We thank Benjamin Jackson for his contributions to the set-up of the experiment, animal collections in the field, laboratory support and discussions, Bruno Buatois, Laurette Sonié, Raphaëlle Leclerc, Sandrine Bioulac, Sylvain Coq and Jérémy Devaux for laboratory assistance, Christian Collin and David Delguedre for technical support, Erwin Amstutz, Markus Schindler and Mark Gessner for their help in litter collections, Christian Körner for his support of work at the Swiss Canopy Crane (SCC) research site in Basel, and anonymous reviewers for thoughtful comments that improved previous versions of our manuscript. This research was possible thanks to the support of the SCC research site and the CO2 enrichment facility by the Swiss National Science Foundation (grants 3100-0597769.99 and 3100-067775.02), the Swiss Federal Office for the Environment (BUWAL) and the University of Basel. Funding was provided by the ESF EUROCORES programme EuroDIVERSITY to the collaborative research project BioCycle. BioCycle is endorsed by DIVERSITAS as contributing towards their current scientific research priorities in biodiversity science.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Amy Austin.
Rights and permissions
About this article
Cite this article
Rouifed, S., Handa, I.T., David, JF. et al. The importance of biotic factors in predicting global change effects on decomposition of temperate forest leaf litter. Oecologia 163, 247–256 (2010). https://doi.org/10.1007/s00442-009-1528-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-009-1528-1