Abstract
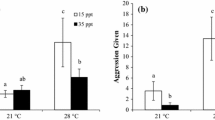
The interplay of abiotic factors and competition has a long history in ecology, although there are very few studies on the interaction of salinity and competition in fish. Mosquitofish (Gambusia holbrooki) are among the most invasive fish worldwide, with well documented ecological impacts on several taxa such as amphibians and small native fish. It has been previously hypothesized, based on field observations, that salinity limits the invasive success of mosquitofish and provides a competitive refuge for Mediterranean cyprinodonts. We experimentally tested this hypothesis by examining the agonistic behaviour and food competition between mosquitofish and an endangered native cyprinodont (Aphanius fasciatus) at three salinities (0, 15, 25‰). Intraspecific aggressive behaviour for both species was not significantly affected by salinity. As salinity increased, mosquitofish decreased their aggressive behaviour towards cyprinodonts and captured less prey. In contrast, the cyprinodonts did not change their behaviour with different salinity treatments, with the possible exception of increased defensive acts in higher salinities, but captured more prey with increasing salinity because of the reduced efficiency of mosquitofish. Our study confirms previous field observations that salinity limits the invasive success of mosquitofish and provides one of the few experimental demonstrations that it may mediate behavioural and competitive interactions between fish species. Condition-specific competition of mosquitofish might be expected with other species and ecosystems worldwide and illustrates the importance of integrating biotic and abiotic factors in the study of interspecific interactions.


Similar content being viewed by others
References
Alcaraz C, García-Berthou E (2007) Life history variation of an invasive fish (Gambusia holbrooki) along a salinity gradient. Biol Conserv 139:83–92. doi:10.1016/j.biocon.2007.06.006
Arthington AH, Lloyd LN (1989) Introduced poeciliids in Australia and New Zealand. In: Meffe GK, Snelson Jr FF (eds) Ecology and evolution of livebearing fishes (Poeciliidae). Prentice Hall, Englewood Cliffs, N.J., pp 333–348
Bagiella E, Sloan RP, Heitjan DF (2000) Mixed-effects models in psychophysiology. Psychophysiology 37:13–20. doi:10.1017/S0048577200980648
Banarescu P (1990). Zoogeography of freshwaters, vol 1. General distribution and dispersal of freshwater animals. AULA, Wiesbaden
Bianco PG (1995) Mediterranean endemic freshwater fishes of Italy. Biol Conserv 72:159–170. doi:10.1016/0006-3207(94)00078-5
Brooker R, Kikvidze Z, Pugnaire FI, Callaway RM, Choler P, Lortie CJ, Michalet R (2005) The importance of importance. Oikos 109:63–70. doi:10.1111/j.0030-1299.2005.13557.x
Caiola N, de Sostoa A (2005) Possible reasons for the decline of two native toothcarps in the Iberian Peninsula: evidence of competition with the introduced Eastern mosquitofish. J Appl Ichthyol 21:358–363. doi:10.1111/j.1439-0426.2005.00684.x
Chervinski J (1983) Salinity tolerance of the mosquito fish, Gambusia affinis (Baird and Girard). J Fish Biol 22:9–11. doi:10.1111/j.1095-8649.1983.tb04720.x
Connell JH (1961) The influence of interspecific competition and other factors on the distribution of the barnacle Chthamalus stellatus. Ecology 42:710–723
Courtenay WR Jr, Meffe GK (1989) Small fishes in strange places: a review of introduced poeciliids. In: Meffe GK, Snelson Jr FF (eds) Ecology and evolution of livebearing fishes (Poeciliidae). Prentice Hall, Englewood Cliffs, N.J., pp 319–331
Cowardin LM (1979) Classification of wetlands and deepwater habitats of the United States. Fish and Wildlife Service, US Department of the Interior
Crain CM, Silliman BR, Bertness S, Bertness MD (2004) Physical and biotic drivers of plant distribution across estuarine salinity gradients. Ecology 85:2539–2549
de Staso III J, Rahel FJ (1994) Influence of water temperature on interactions between juvenile Colorado river cutthroat trout and brook trout in a laboratory stream. Trans Am Fish Soc 123:289–297
Doadrio I (2002) Atlas y libro rojo de los peces continentales de España. Ministerio de Medio Ambiente, Madrid
Dukes JS, Mooney HA (2004) Disruption of ecosystem processes in western North America by invasive species. Rev Chil Hist Nat 77:411–437
Dunson WA, Travis J (1991) The role of abiotic factors in community organization. Am Nat 138:1067-1091. doi:10.1086/285270
Dunson WA, Travis J (1994) Patterns in the evolution of physiological specialization in salt-marsh animals. Estuaries 17:102–110
Fernández-Delgado C, Hernando-Casal JA, Herrera M, Bellido M (1988) Age, growth and reproduction of Aphanius iberus (Cuv. and Val., 1846) in the lower reaches of the Guadalquivir river (south-west Spain). Freshwater Biol 20:227–234. doi:10.1111/j.1365-2427.1988.tb00446.x
García-Berthou E, Moreno-Amich R (1992) Age and growth of an Iberian cyprinodont, Aphanius iberus (Cuv. and Val.), in its most northerly population. J Fish Biol 40:929–937. doi:10.1111/j.1095-8649.1992.tb02638.x
García-Berthou E, Alcaraz C, Pou-Rovira Q, Zamora L, Coenders G, Feo C (2005) Introduction pathways and establishment rates of invasive aquatic species in Europe. Can J Fish Aquat Sci 62:453–463. doi:10.1139/f05-017
Hamer AJ, Lane SJ, Mahony MJ (2002) The role of introduced mosquitofish (Gambusia holbrooki) in excluding the native green and golden bell frog (Litoria aurea) from original habitats in south-eastern Australia. Oecologia 132:445–452. doi:10.1007/s00442-002-0968-7
Haney DC, Nordlie FG, Binell J (1999) Influence of simulated tidal changes in ambient salinity on routine metabolic rate in Cyprinodon variegatus. Copeia 1999:509–514
Holway DA, Suarez AV (1999) Animal behavior: an essential component of invasion biology. Trends Ecol Evol 14:328–330. doi:10.1016/S0169-5347(99)01636-5
Inouye BD (2001) Response surface experimental designs for investigating interspecific competition. Ecology 82:2696–2706
Kats LB, Ferrer RP (2003) Alien predators and amphibian declines: review of two decades of science and the transition to conservation. Divers Distrib 9:99–110. doi:10.1046/j.1472-4642.2003.00013.x
Keivany Y, Soofiani NM (2004) Contribution to the biology of Zagros tooth-carp, Aphanius vladykovi (Cyprinodontidae) in central Iran. Environ Biol Fishes 71:165–169. doi:10.1007/s10641-004-0106-y
Krumholz LA (1948) Reproduction in the western mosquitofish, Gambusia affinis affinis, and its use in mosquito control. Ecol Monogr 18:1–43
La Peyre MKG, Grace JB, Hahn E, Mendelssohn IA (2001) The importance of competition in regulating plant species abundance along a salinity gradient. Ecology 82:62–69
Leonardos I, Sinis A (1998) Reproductive strategy of Aphanius fasciatus Nardo, 1827 (Pisces: Cyprinodontidae) in the Mesolongi and Etolikon lagoons (W. Greece). Fish Res 35:171–181. doi:10.1016/S0165-7836(98)00082-4
Leonardos I, Sinis A (1999) Population age and sex structure of Aphanius fasciatus (Nardo, 1827) (Pisces: Cyprinodontidae) in the Mesolongi and Etolikon lagoons (W. Greece). Fish Res 40:227–235. doi:10.1016/S0165-7836(98)00231-8
Lortie CJ, Callaway RM (2006) Re-analysis of meta-analysis: support for the stress-gradient hypothesis. J Ecol 94:7–16. doi:10.1111/j.1365-2745.2005.01066.x
Maestre FT, Valladares F, Reynolds JF (2006) The stress-gradient hypothesis does not fit all relationships between plant–plant interactions and abiotic stress: further insights from arid environments. J Ecol 94:17–22. doi:10.1111/j.1365-2745.2005.01089.x
Mills MD, Rader RB, Belk MC (2004) Complex interactions between native and invasive fish: the simultaneous effects of multiple negative interactions. Oecologia 141:713–721. doi:10.1007/s00442-004-1695-z
Myers GS (1949). Salt-tolerance of fresh-water fish groups in relation to zoogeographical problems. Bijdr Dierkd 28:315–322
Nordlie FG (1987) Salinity tolerance and osmotic regulation in the diamond killifish, Adinia xenica. Environ Biol Fishes 20:229–232. doi:10.1007/BF00004957
Nordlie FG, Mirandi A (1996) Salinity relationships in a freshwater population of eastern mosquitofish. J Fish Biol 49:1226–1232. doi:10.1006/jfbi.1996.0249
Nordlie FG, Haney DC, Walsh SJ (1992) Comparisons of salinity tolerances and osmotic regulatory capabilities in populations of sailfin molly (Poecilia latipinna) from brackish and fresh waters. Copeia 1992:741–746
Oltra R, Todolí R (2000) Reproduction of the endangered killifish Aphanius iberus at different salinities. Environ Biol Fish 57:113–115. doi:10.1023/A:1007579527064
Pilastro A, Benetton S, Bisazza A (2003) Female aggregation and male competition reduce costs of sexual harassment in the mosquitofish Gambusia holbrooki. Anim Behav 65:1161–1167. doi:10.1006/anbe.2003.2118
Plaut I (2000) Resting metabolic rate, critical swimming speed, and routine activity of the euryhaline cyprinodontid, Aphanius dispar, acclimated to a wide range of salinities. Physiol Biochem Zool 73:590–596. doi:10.1086/317746
Rehage JS, Barnett BK, Sih A (2005) Behavioural responses to a novel predator and competitor of invasive mosquitofish and their non-invasive relatives (Gambusia sp.). Behav Ecol Sociobiol 57:256–266
Rice JA, Sax DF (2005) Testing fundamental evolutionary questions at large spatial and demographic scales: species invasions as an underappreciated tool. In: Sax DF, Stachowicz JJ, Gaines SD (eds) Species Invasions: insights into ecology, evolution and biogeography. Sinauer, Sunderland, Mass., pp 291–308
Rincón PA, Correas AM, Morcillo F, Risueño P, Lobón-Cerviá J (2002) Interaction between the introduced eastern mosquitofish and two autochthonous Spanish toothcarps. J Fish Biol 61:1560–1585. doi:10.1006/jfbi.2002.2175
Snelson FF Jr (1989) Social and environmental control of life history traits in poeciliid fishes. In: Meffe GK, Snelson Jr FF (eds) Ecology and evolution of livebearing fishes (Poeciliidae). Prentice Hall, Englewood Cliffs, N.J., pp 149–161
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research. Freeman, New York
Swanson C (1998) Interactive effects of salinity on metabolic rate, activity, growth and osmoregulation in the euryhaline milkfish (Chanos chanos). J Exp Biol 201:3355–3366
Tabachnick BG, Fidell LS (2001) Computer-assisted research design and analysis. Allyn and Bacon, Boston, Mass.
Taniguchi Y, Nakano S (2000) Condition-specific competition: implications for the altitudinal distribution of stream fishes. Ecology 81:2027–2039
Taniguchi Y, Rahel FJ, Novinger DC, Gerow KG (1998) Temperature mediation of competitive interactions among three fish species that replace each other along longitudinal stream gradients. Can J Fish Aquat Sci 55:1894–1901. doi:10.1139/cjfas-55-8-1894
Tansley AG (1917) On competition between Galium saxatile L. (G. hercynicum Weig.) and Galium sylvestre Poll. (G. asperum Schreb.) on different types of soil. J Ecol 5:173–179
Thomas ML, Holway DA (2005) Condition-specific competition between invasive Argentine ants and Australian Iridomyrmex. J Anim Ecol 74:532-542. doi:10.1111/j.1365-2656.2005.00952.x
von Ende CN (2001) Repeated-measures analysis: growth and other time-dependent measures. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments. Oxford University Press, New York, pp 134–157
Acknowledgements
The research was authorized by the Istituto Superiore di Sanità and the fish were caught under a fishing permit from the Regione Veneto. Financial support was provided by the Spanish Ministry of Education (REN2003-00477) and the Government of Catalonia (Catalan Government Distinction Award for university research 2004). C. A. held a doctoral fellowship (FPU AP 2002–0206) from the Spanish Ministry of Education and additional financial support was provided by a travel grant (FPU program). We thank the Comparative Psychology Research Group for their hospitality and assistance during the research stay and two anonymous reviewers for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Wolf Mooij.
Rights and permissions
About this article
Cite this article
Alcaraz, C., Bisazza, A. & García-Berthou, E. Salinity mediates the competitive interactions between invasive mosquitofish and an endangered fish. Oecologia 155, 205–213 (2008). https://doi.org/10.1007/s00442-007-0899-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-007-0899-4