Abstract
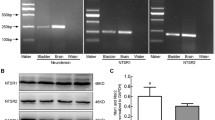
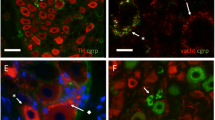
β3-Adrenergic receptor agonists are currently under clinical development for the treatment of overactive bladder, a condition that is prevalent in postmenopausal women. These agents purportedly relax bladder smooth muscle through a direct action at the myocyte β3-receptor. The aim of this study was to examine the expression of the individual beta-adrenergic receptors in full thickness sections from ageing human female bladder. We obtained a series of rabbit polyclonal antibodies generated against each of the three β-adrenergic receptors, and validated their receptor specificity in CHOK1 cells expressing each of the individual receptors. Immunostaining for β1, β2, and β3 were each more prominent in the urothelium than in the detrusor, with all receptors expressed in the same cell types, indicating co-expression of all three receptors throughout the urothelium in addition to the detrusor. Staining of all receptors was also observed in suburothelial myofibroblast-like cells, intramural ganglion cells, and in Schwann cells of intramural nerves. The β3-receptor in the human urothelium appears to be functional, as two different selective β3-receptor agonists, TAK677 and BRL37344, stimulate cAMP formation in UROtsa cells. Densitometry analysis indicates a persistent expression of all receptors throughout the bladder with increasing age, with the exception of the β2-receptor in the urothelium of the trigone, which appears to decrease slightly in older women. These data indicate that β3-receptor expression is maintained with age, but may function in concert with other β-receptors. Activation of the myocyte receptor may be influenced by action on non-myocyte structures including the intramural ganglion cells and myofibroblasts.








Similar content being viewed by others
References
Aizawa N, Igawa Y, Nishizawa O, Wyndaele JJ (2009) Effects of CL316, 243 a β3-adrenoceptor agonist, and intravesical prostaglandin E2 on the primary bladder afferent activity of the rat. Neurourol Urodynam 29:771–776
Badawi JK, Seja T, Uecelehan H, Honeck P, Kwon ST, Bross S, Langbein S (2007) Relaxation of human detrusor muscle by selective beta-2 and beta-3 agonists and endogenous catecholamines. Urology 69:785–790
Birder LA, Nealen ML, Kiss S, de Groat WC, Caterina MJ, Wang E, Apodaca G, Kanai AJ (2002) beta -adrenoceptor agonists stimulate endothelial nitric oxide synthase in rat urinary bladder urothelial cells. J Neurosci 22:8063–8070
Breit A, Lagace M, Bouvier M (2004) Hetero-oligomerization between {beta}2- and {beta}3-adrenergic receptors generates a {beta}-adrenergic signaling unit with distinct functional properties. J Biol Chem 279:28756–28765
Cellek S, Thangiah R, Bassil AK, Campbell CA, Gray KM, Stretton JL, Lalude O, Vivekanandan S, Wheeldon A, Winchester WJ, Sanger GJ, Schemann M, Lee K (2007) Demonstration of functional neuronal beta3-adrenoceptors within the enteric nervous system. Gastroenterology 133:175–183
de Groat WC (2004) The urothelium in overactive bladder: passive bystander or active participant? Urology 64:7–11
de Groat WC, Saum WR (1972) Sympathetic inhibition of the urinary bladder and of pelvic ganglionic transmission in the cat. J Physiol 220:297–314
Derweesh IH, Wheeler MA, Weiss RM (2000) Alterations in G-proteins and beta -adrenergic responsive adenylyl cyclase in rat urinary bladder during aging. J Pharmacol Exp Ther 294:969–974
Fowler CJ, Griffiths D, de Groat WC (2008) The neural control of micturition. Nat Rev Neurosci 9:453–466
Frazier EP, Schneider T, Michel MC (2006) Effects of gender, age and hypertension on beta-adrenergic receptor function in rat urinary bladder. Naunyn Schmiedebergs Arch Pharmacol 373:300–309
Fry CH, Ikeda Y, Harvey R, Wu C, Sui GP (2004) Control of bladder function by peripheral nerves: avenues for novel drug targets. Urology 63:24–31
Fujimura T, Tamura K, Tsutsumi T, Yamamoto T, Nakamura K, Koibuchi Y, Kobayashi M, Yamaguchi O (1999) Expression and possible functional role of the β3-adrenoceptor in human and rat detrusor muscle. J Urol 161:680–685
Goepel M, Wittman A, Rubben H, Michel MC (1997) Comparison of adrenoceptor subtype expression in porcine and human bladder and prostate. Urol Res 25:199–206
Hamdani N, van der Velden J (2009) Lack of specificity of antibodies directed against human beta-adrenergic receptors. Naunyn Schmiedebergs Arch Pharmacol 379:403–407
Harmon E, Porter J, Porter J (2005) beta-adrenergic receptor activation in immortalized human urothelial cells stimulates inflammatory responses by PKA-independent mechanisms. Cell Communication and Signaling 3:10
Igawa Y, Yamazaki Y, Takeda H, Hayakawa K, Akahane M, Ajisawa Y, Yoneyama T, Nishizawa O, Andersson KE (1999) Functional and molecular biological evidence for a possible β3-adrenoceptor in the human detrusor muscle. Br J Pharmacol 126:819–825
Igawa Y, Yamazaki Y, Takeda H, Kaidoh K, Akahane M, Ajisawa Y, Yoneyama T, Nishizawa O, Andersson KE (2001) Relaxant effects of isoproterenol and selective β3-adrenoceptor agonists on normal, low compliant and hyperreflexic human bladders. J Urol 165:240–244
Keast JR, Kawatani M, de Groat WC (1990) Sympathetic modulation of cholinergic transmission in cat vesical ganglia is mediated by alpha 1- and alpha 2-adrenoceptors. Am J Physiol 258:R44–R50
Klausner AP, Rourke KF, Miner AS, Ratz PH (2009) Potentiation of carbachol-induced detrusor smooth muscle contractions by beta-adrenoceptor activation. Eur J Pharmacol 606:191–198
Kullmann FA, Limberg BJ, Artim DE, Shah M, Downs TR, Contract D, Wos J, Rosenbaum JS, de Groat WC (2009) Effects of β3-adrenergic receptor activation on rat urinary bladder hyperactivity induced by ovariectomy. J Pharmacol Exp Ther 330:704–717
Kullmann FA, Downs TR, Artim D, Limberg BJ, Shah M, Contract D, de Groat WC, Rosenbaum JS (2010) Urothelial beta3 adrenergic receptors in the rat bladder. Neurourol Urodynam (In press)
Li G, Li K, Li Z, Wang P (2003) Age-dependent changes in beta-adrenoceptor function in human detrusors and possible mechanisms. Chin Med J (Engl ) 116:1511–1514
Lindholm P, Lose G (1986) Terbutaline (Bricanyl) in the treatment of female urge incontinence. Urol Int 41:158–160
Longhurst PA, Levendusky M (1999) Pharmacological characterization of [beta]-adrenoceptors mediating relaxation of the rat urinary bladder in vitro. Br J Pharmacol 127:1744–1750
Masunaga K, Chappl CR, McKay NG, Yoshida M, Sellers DJ (2010) The β3-adrenoceptor mediates the inhibitory effects of β-adrenoceptor agonists via the urothelium in pig bladder dome. Neurourol Urodynam 29:1320–1325
Michel MC, Vrydag W (2006) alpha(1)-, alpha(2)- and beta-adrenoceptors in the urinary bladder, urethra and prostate. Br J Pharmacol 147(Suppl 2):S88–S119
Michel MC, Wieland T, Tsujimoto G (2009) How reliable are G-protein-coupled receptor antibodies? Naunyn Schmiedebergs Arch Pharmacol 379:385–388
Morita T, Iizuka H, Iwata T, Kondo S (2000) Function and distribution of β3-adrenoceptors in rat, rabbit and human urinary bladder and external urethral sphincter. J Smooth Muscle Res 36:21–32
Murakami S, Chapple CR, Akino H, Sellers DJ, Chess-Williams R (2007) The role of the urothelium in mediating bladder responses to isoprenaline. BJU Int 99:669–673
Niclauss N, Michel-Reher MB, Alewijnse AE, Michel MC (2006) Comparison of three radioligands for the labelling of human beta-adrenoceptor subtypes. Naunyn Schmiedebergs Arch Pharmacol 374:99–105
Nishimoto T, Latifpour J, Wheeler MA, Yoshida M, Weiss RM (1995) Age-dependent alterations in beta-adrenergic responsiveness of rat detrusor smooth muscle. J Urol 153:1701–1705
Nomiya M, Yamaguchi O (2003) A quantitative analysis of mRNA expression of alpha 1 and beta-adrenoceptor subtypes and their functional roles in human normal and obstructed bladders. J Urol 170:649–653
Otsuka A, Shinbo H, Matsumoto R, Kurita Y, Ozono S (2008) Expression and functional role of beta-adrenoceptors in the human urinary bladder urothelium. Naunyn Schmiedebergs Arch Pharmacol 377:473–481
Pradidarcheep W, Stallen J, Labruyere WT, Dabhoiwala NF, Michel MC, Lamers WH (2009) Lack of specificity of commercially available antisera against muscarinergic and adrenergic receptors. Naunyn Schmiedebergs Arch Pharmacol 379:397–402
Rossi MR, Masters JR, Park S, Todd JH, Garrett SH, Sens MA, Somji S, Nath J, Sens DA (2001) The immortalized UROtsa cell line as a potential cell culture model of human urothelium. Environ Health Perspect 109:801–808
Skliris GP, Parkes AT, Limer JL, Burdall SE, Carder PJ, Speirs V (2002) Evaluation of seven oestrogen receptor β antibodies for immunohistochemistry, western blotting, and flow cytometry in human breast tissue. J Pathol 197:155–162
Takeda M, Obara K, Mizusawa T, Tomita Y, Arai K, Tsutsui T, Hatano A, Takahashi K, Nomura S (1999) Evidence for beta 3-adrenoceptor subtypes in relaxation of the human urinary bladder detrusor: analysis by molecular biological and pharmacological methods. J Pharmacol Exp Ther 288:1367–1373
Ursino MG, Vasina V, Raschi E, Crema F, De PF (2009) The β3-adrenoceptor as a therapeutic target: current perspectives. Pharmacol Res 59:221–234
Ushiroyama T, Ikeda A, Ueki M (2000) Clinical efficacy of clenbuterol and propiverine in menopausal women with urinary incontinence: improvement in quality of life. J Med 31:311–319
Vrydag W, Michel MC (2007) Tools to study β3-adrenoceptors. Naunyn Schmiedebergs Arch Pharmacol 374:385–398
Wheeler MA, Ayyagari RR, Wheeler GL, Weiss RM (2005) Regulation of cyclic nucleotides in the urinary tract. J Smooth Muscle Res 41:1–21
Wuest M, Eichhorn B, Grimm MO, Wirth MP, Ravens U, Kaumann AJ (2009) Catecholamines relax detrusor through beta 2-adrenoceptors in mouse and beta 3-adrenoceptors in man. J Pharmacol Exp Ther 328:213–222
Yamanishi T, Chapple CR, Yasuda K, Yoshida K, Chess-Williams R (2002) The role of β3-adrenoceptors in mediating relaxation of porcine detrusor muscle. Br J Pharmacol 135:129–134
Yamazaki Y, Takeda H, Akahane M, Igawa Y, Nishizawa O, Ajisawa Y (1998) Species differences in the distribution of [beta]-adrenoceptor subtypes in bladder smooth muscle. Br J Pharmacol 124:593–599
Acknowledgements
We thank the staff of Asterand Ltd for excellent technical assistance with the expanded human IHC samples, and Dr. Michael P. Meredith (The Procter & Gamble Company) for help with statistical analysis during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
Validation of specific staining for the human β3-AR with the KG115 antibody. The antibody was generated against the human β3-AR -COOH terminus. The antibody was used at a concentration of 10 μg/ml. Staining (red) were observed in transiently transfected CHOK1 cells with the human β2-AR (b) and β3-AR (c) but not β1-AR (a) and empty vector (d). This suggests that the KG115 does not specifically recognize β3-AR by immunostaining. (GIF 179 kb)
Supplemental Figure 2
Specific Binding of β3-AR selective ligands in Mock CHOK1 cells. Whole cell binding was performed of CHOK1 cells transiently transfected with pcDNA3.1 (Mock), cDNA to illustrate endogenous expression of hβ3-AR in CHOK1 cells. The binding study was performed using 46 pM [125I] I-CYP tracer and Isoprotenerol (■) a non-selective β-AR ligand or the two β3-AR selective ligands BRL37344(∇) and TAK677(●). Relative affinity (Ki) values were 1.35 × 10-3 M, 9.35 × 10-5 M, and 1.31 × 10-5 M, respectively, for the experiment shown. The error bars represent SEM of three determinations. (GIF 3 kb)
Rights and permissions
About this article
Cite this article
Limberg, B.J., Andersson, KE., Aura Kullmann, F. et al. β−Adrenergic receptor subtype expression in myocyte and non-myocyte cells in human female bladder. Cell Tissue Res 342, 295–306 (2010). https://doi.org/10.1007/s00441-010-1053-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-010-1053-x