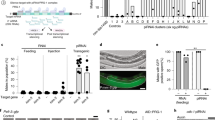
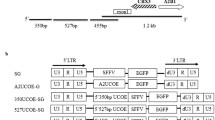
Abstract
Silencing of a target locus by an unlinked silencing locus can result from transcription inhibition (transcriptional gene silencing; TGS) or mRNA degradation (post-transcriptional gene silencing; PTGS), owing to the production of double-stranded RNA (dsRNA) corresponding to promoter or transcribed sequences, respectively. The involvement of distinct cellular components in each process suggests that dsRNA-induced TGS and PTGS likely result from the diversification of an ancient common mechanism. However, a strict comparison of TGS and PTGS has been difficult to achieve because it generally relies on the analysis of distinct silencing loci. We describe a single transgene locus that triggers both TGS and PTGS, owing to the production of dsRNA corresponding to promoter and transcribed sequences of different target genes. We describe mutants and epigenetic variants derived from this locus and propose a model for the production of dsRNA. Also, we show that PTGS, but not TGS, is graft-transmissible, which together with the sensitivity of PTGS, but not TGS, to RNA viruses that replicate in the cytoplasm, suggest that the nuclear compartmentalization of TGS is responsible for cell-autonomy. In contrast, we contribute local and systemic trafficking of silencing signals and sensitivity to viruses to the cytoplasmic steps of PTGS and to amplification steps that require high levels of target mRNAs.







Similar content being viewed by others
Abbreviations
- dsRNA:
-
Double-stranded RNA
- GUS:
-
ß-Glucuronidase
- NIR:
-
Nitrate reductase
- PTGS:
-
Post-transcriptional gene silencing
- siRNA:
-
Short interfering RNA
- ssRNA:
-
Single stranded RNA
- TGS:
-
Transcriptional gene silencing
References
Chan SW, Zilberman D, Xie Z, Johansen LK, Carrington JC, Jacobsen SE (2004) RNA silencing genes control de novo DNA methylation. Science 303:1336
Choi YG, Randles JW (1997) Microgranular cellulose improves dsRNA recovery from plant nucleic acid extracts. Biotechniques 23:610–611
Chuang CF, Meyerowitz EM (2000) Specific and heritable genetic interference by double-stranded RNA in Arabidopsis thaliana. Proc Natl Acad Sci USA 97:4985–4990
Crete P, Leuenberger S, Iglesias VA, Suarez V, Schob H, Holtorf H, van Eeden S, Meins F (2001) Graft transmission of induced and spontaneous post-transcriptional silencing of chitinase genes. Plant J 28:493–501
Dunoyer P, Voinnet O (2005) The complex interplay between plant viruses and host RNA-silencing pathways. Curr Opin Plant Biol 8:415–423
Elmayan T, Balzergue S, Beon F, Bourdon V, Daubremet J, Guenet Y, Mourrain P, Palauqui JC, Vernhettes S, Vialle T, Wostrikoff K, Vaucheret H (1998) Arabidopsis mutants impaired in cosuppression. Plant Cell 10:1747–1758
Elmayan T, Vaucheret H (1996) Expression of single copies of a strongly expressed 35S transgene can be silenced post-transcriptionally. Plant J 9:787–797
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391:806–811
Gasciolli V, Mallory AC, Bartel DP, Vaucheret H (2005) Partially redundant functions of arabidopsis DICER-like enzymes and a role for dcl4 in producing trans-acting siRNAs. Curr Biol 15:1494–1500
Herr AJ, Jensen MB, Dalmay T, Baulcombe DC (2005) RNA polymerase IV directs silencing of endogenous DNA. Science 308:118–120
Jones L, Ratcliff F, Baulcombe DC (2001) RNA-directed transcriptional gene silencing in plants can be inherited independently of the RNA trigger and requires Met1 for maintenance. Curr Biol 11:747–757
Linn F, Heidmann I, Saedler H, Meyer P (1990) Epigenetic changes in the expression of the maize A1 gene in Petunia hybrida: role of numbers of integrated gene copies and state of methylation. Mol Gen Genet 222:329–336
Lippman Z, May B, Yordan C, Singer T, Martienssen R (2003) Distinct mechanisms determine transposon inheritance and methylation via small interfering RNA and histone modification. PLOS Biol 1:E67
Mallory AC, Mlotshwa S, Bowman LH, Vance VB (2003) The capacity of transgenic tobacco to send a systemic RNA silencing signal depends on the nature of the inducing transgene locus. Plant J 35:82–92
Marathe R, Smith TH, Anandalakshmi R, Bowman LH, Fagard M, Mourrain P, Vaucheret H, Vance VB (2000) Plant viral suppressors of post-transcriptional silencing do not suppress transcriptional silencing. Plant J 22:51–59
Matzke MA, Primig M, Trnovsky J, Matzke AJ (1989) Reversible methylation and inactivation of marker genes in sequentially transformed tobacco plants. EMBO J 8:643–649
Mette MF, Aufsatz W, van der Winden J, Matzke MA, Matzke AJ (2000) Transcriptional silencing and promoter methylation triggered by double-stranded RNA. EMBO J 19:5194–5201
Mette MF, Matzke AJ, Matzke MA (2001) Resistance of RNA-mediated TGS to HC-Pro, a viral suppressor of PTGS, suggests alternative pathways for dsRNA processing. Curr Biol 11:1119–1123
Mette MF, van der Winden J, Matzke MA, Matzke AJ (1999) Production of aberrant promoter transcripts contributes to methylation and silencing of unlinked homologous promoters in trans. EMBO J 18:241–248
Morel JB, Godon C, Mourrain P, Beclin C, Boutet S, Feuerbach F, Proux F, Vaucheret H (2002) Fertile hypomorphic ARGONAUTE (ago1) mutants impaired in post-transcriptional gene silencing and virus resistance. Plant Cell 14:629–639
Morel JB, Mourrain P, Beclin C, Vaucheret H (2000) DNA methylation and chromatin structure affect transcriptional and post-transcriptional transgene silencing in Arabidopsis. Curr Biol 10:1591–1594
Napoli C, Lemieux C, Jorgensen R (1990) Introduction of a chimeric chalcone sythase gene into petunia results in reversible co-suppression of homologous gene in trans. Plant Cell 2:279–289
Palauqui JC, Elmayan T, Pollien JM, Vaucheret H (1997) Systemic acquired silencing: transgene-specific post-transcriptional silencing is transmitted by grafting from silenced stocks to non-silenced scions. EMBO J 16:4738–4745
Park YD, Papp I, Moscone EA, Iglesias VA, Vaucheret H, Matzke AJ, Matzke MA (1996) Gene silencing mediated by promoter homology occurs at the level of transcription and results in meiotically heritable alterations in methylation and gene activity. Plant J 9:183–194
Peragine A, Yoshikawa M, Wu G, Albrecht HL, Poethig RS (2004) SGS3 and SGS2/SDE1/RDR6 are required for juvenile development and the production of trans-acting siRNAs in Arabidopsis. Genes Dev 18:2368–2379
Roth BM, Pruss GJ, Vance VB (2004) Plant viral suppressors of RNA silencing. Virus Res 102:97–108
Sijen T, Plasterk RH (2003) Transposon silencing in the Caenorhabditis elegans germ line by natural RNAi. Nature 426:310–314
Sijen T, Vijn I, Rebocho A, van Blokland R, Roelofs D, Mol JN, Kooter JM (2001) Transcriptional and posttranscriptional gene silencing are mechanistically related. Curr Biol 11:436–440
Smith CJ, Watson CF, Bird CR, Ray J, Schuch W, Grierson D (1990) Expression of a truncated tomato polygalacturonase gene inhibits expression of the endogenous gene in transgenic plants. Mol Gen Genet 224:477–481
Thierry D, Vaucheret H (1996) Sequence homology requirements for transcriptional silencing of 35S transgenes and post-transcriptional silencing of nitrite reductase (trans) genes by the tobacco 271 locus. Plant Mol Biol 32:1075–1083
Tomari Y, Zamore PD (2005) Perspective: machines for RNAi. Genes Dev 19:517–529
van der Krol AR, Mur LA, Beld M, Mol JN, Stuitje AR (1990) Flavonoid genes in petunia: addition of a limited number of gene copies may lead to a suppression of gene expression. Plant Cell 2:291–299
Van Houdt H, Bleys A, Depicker A (2003) RNA target sequences promote spreading of RNA silencing. Plant Physiol 131:245–253
Vance V, Vaucheret H (2001) RNA silencing in plants—defense and counterdefense. Science 292:2277–2280
Vaucheret H (1993) Identification of a general silencer for 19S and 35S promoters in a transgenic tobacco plant: 90 pb of homology in the promoter sequences are sufficient for trans-inactivation. C R Acad Sci Paris 316:1471–1483
Vaucheret H (2006) Post-transcriptional small RNA pathways in plants: mechanisms and regulations. Genes Dev 20:759–771
Vaucheret H, Kronenberger J, Lepingle A, Vilaine F, Boutin JP, Caboche M (1992) Inhibition of tobacco nitrite reductase activity by expression of antisense RNA. Plant J 2:559–569
Vaucheret H, Palauqui JC, Elmayan T, Moffatt B (1995a) Molecular and genetic analysis of nitrite reductase co-suppression in transgenic tobacco plants. Mol Gen Genet 248:311–317
Vaucheret H, Mourrain P, Robalo J-C, Pollien J-M (1995b) Nitrite reductase silencing as a tool for selecting spontaneous haploid plants. Plant Cell Rep 15:12–16
Vaucheret H, Vazquez F, Crete P, Bartel DP (2004) The action of ARGONAUTE1 in the miRNA pathway and its regulation by the miRNA pathway are crucial for plant development. Genes Dev 18:1187–1197
Vazquez F, Vaucheret H, Rajagopalan R, Lepers C, Gasciolli V, Mallory AC, Hilbert JL, Bartel DP, Crete P (2004) Endogenous trans-acting siRNAs regulate the accumulation of Arabidopsis mRNAs. Mol Cell 16:69–79
Voinnet O, Vain P, Angell S, Baulcombe DC (1998) Systemic spread of sequence-specific transgene RNA degradation in plants is initiated by localized introduction of ectopic promoterless DNA. Cell 95:177–187
Waterhouse PM, Graham MW, Wang MB (1998) Virus resistance and gene silencing in plants can be induced by simultaneous expression of sense and antisense RNA. Proc Natl Acad Sci USA 95:13959–13964
Xie Z, Allen E, Wilken A, Carrington JC (2005) DICER-LIKE 4 functions in trans-acting small interfering RNA biogenesis and vegetative phase change in Arabidopsis thaliana. Proc Natl Acad Sci USA 102:12984–12989
Xie Z, Johansen LK, Gustafson AM, Kasschau KD, Lellis AD, Zilberman D, Jacobsen SE, Carrington JC (2004) Genetic and functional diversification of small RNA pathways in plants. PLOS Biol 2:642–652
Yoshikawa M, Peragine A, Park MY, Poethig RS (2005) A pathway for the biogenesis of trans-acting siRNAs in Arabidopsis. Genes Dev 19:2164–2175
Zilberman D, Cao X, Jacobsen SE (2003) ARGONAUTE4 control of locus-specific siRNA accumulation and DNA and histone methylation. Science 299:716–719
Acknowledgments
We thank Jean-Marie Pollien (INRA, Versailles, France) for all graftings and Allison Mallory (Whitehead Institute, Cambridge, MA, USA) for fruitful discussions. This work was partly funded by the European Union Biotechnology Programm on Gene Silencing (B104-CT96-0253).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mourrain, P., van Blokland, R., Kooter, J.M. et al. A single transgene locus triggers both transcriptional and post-transcriptional silencing through double-stranded RNA production. Planta 225, 365–379 (2007). https://doi.org/10.1007/s00425-006-0366-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-006-0366-1