Abstract
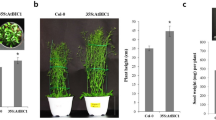
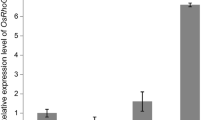
The phytochromes are a family of red/far-red light absorbing photoreceptors that control plant developmental and metabolic processes in response to changes in the light environment. We report here the overexpression of Arabidopsis thaliana PHYTOCHROME A (PHYA) gene in a commercially important indica rice variety (Oryza sativa L. Pusa Basmati-1). The expression of the transgene was driven by the light-regulated and tissue-specific rice rbcS promoter. Several independent homozygous sixth generation (T5) transgenic lines were characterized and shown to accumulate relatively high levels of PHYA protein in the light. Under both far-red and red light, PHYA-overexpressing lines showed inhibition of the coleoptile extension in comparison to non-transgenic seedlings. Furthermore, compared with non-transgenic rice plants, mature transgenic plants showed significant reduction in plant height, internode length and internode diameter (including differences in cell size and number), and produced an increased number of panicles per plant. Under greenhouse conditions, rice grain yield was 6–21% higher in three PHYA-overexpressing lines than in non-transgenic plants. These results demonstrate the potential of manipulating light signal-transduction pathways to minimize the problems of lodging in basmati/aromatic rice and to enhance grain productivity.




Similar content being viewed by others
Abbreviations
- FR:
-
Far-red light
- PBNT:
-
Pusa Basmati-1
- Pfr:
-
Far-red-absorbing form of phytochrome
- PHYA :
-
Phytochrome A gene
- phyA :
-
Phytochrome A mutant
- PHYA:
-
Phytochrome A apoprotein
- phyA:
-
Phytochrome A holoprotein
- PHYB :
-
Phytochrome B gene
- Pr:
-
Red-absorbing form of phytochrome
- R:
-
Red light
- R:FR:
-
Quantum ratio of red to far-red light
References
Aggarwal RK, Shenoy V, Ramadevi J, Rajkumar R, Singh L (2002) Molecular characterization of some Indian Basmati and other elite rice genotypes using fluorescent-AFLP. Theor Appl Genet 105:680–690
Ballare CL (2001) Arabidopsis mutants and other model systems in plant physiological ecology. Trends Plant Sci 6:99
Boccalandro HE, Ploschuk EL, Yanovsky MJ, Sanchez RA, Gatz C, Casal JJ (2003) Increased phytochrome B alleviates density effects on tuber yield of field potato crops. Plant Physiol 133:1539–1546
Boylan MT, Quail PH (1989) Oat phytochrome is biologically active in transgenic tomatoes. Plant Cell 1:765–773
Boylan MT, Quail PH (1991) Phytochrome A overexpression inhibits hypocotyl elongation in transgenic Arabidopsis. Proc Natl Acad Sci USA 88:10806–10810
Casal JJ (1996) Phytochrome A enhances the promotion of hypocotyl growth caused by reductions in levels of phytochrome B in its far-red-light-absorbing form in light-grown Arabidopsis thaliana. Plant Physiol 112:965–973
Clack T, Mathews S, Sharrock R (1994) The phytochrome apoprotein family in Arabidopsis is encoded by five genes: the sequences and expression of PHYD and PHYE. Plant Mol Biol 25:413–427
Clough RC, Vierstra RD (1997) Phytochrome degradation. Plant Cell Environ 20:713–721
Clough RC, Casal JJ, Jordan ET, Christou P, Vierstra RD (1995) Expression of a functional oat phytochrome A in transgenic rice. Plant Physiol 109:1039–1045
Davis SJ, Bhoo SH, Durski AM, Walker JM, Vierstra RD (2001) The heme-oxygenase family required for phytochrome chromophore biosynthesis is necessary for proper photomorphogenesis in higher plants. Plant Physiol 126:656–669
Dehesh K, Tepperman J, Christensen AH, Quail PH (1991) PHYB is evolutionarily conserved and constitutively expressed in rice seedling shoots. Mol Gen Genet 225:305–313
Franklin KA, Davis SJ, Stoddart WM, Vierstra RD, Whitelam GC (2003) Mutant analyses define multiple roles for phytochrome C in Arabidopsis photomorphogenesis. Plant Cell 15:1981–1989
Garg AK, Kim J-K, Owens TG, Ranwala AP, Choi YD, Kochian LV, Wu RJ (2002) Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc Natl Acad Sci USA 99:15898–15903
Halliday KJ, Thomas B, Whitelam GC (1997) Expression of heterologous phytochrome A, B or C in transgenic tobacco plants alters vegetative development and flowering time. Plant J 12:1079–1090
Halliday KJ, Bolle C, Chua NH, Whitelam GC (1999) Overexpression of rice phytochrome A partially complements phytochrome B deficiency in Arabidopsis. Planta 207:401–409
Hirschfeld M, Tepperman JM, Clack T, Quail PQ, Sharrock RA (1998) Coordination of phytochrome levels in phyB mutants of Arabidopsis as revealed by apoprotein-specific monoclonal antibodies. Genetics 149:523–535
Izawa T, Oikawa T, Tokutomi S, Okuno K, Shimamoto K (2000) Phytochromes confer the photoperiodic control of flowering in rice (a short-day plant). Plant J 22:391–399
Johnson E, Bradley M, Harberd NP, Whitelam GC (1994) Photoresponses of light-grown phyA mutants of Arabidopsis. Plant Physiol 105:141–149
Jordan ET, Hatfield PM, Hondred D, Talon M, Zeevaart JAD, Vierstra RD (1995) Phytochrome A overexpression in transgenic tobacco: Correlation of dwarf phenotype with high concentrations of phytochrome in vascular tissue and attenuated gibberellin levels. Plant Physiol 107:797–805
Kay SA, Nagatani A, Keith B, Deak M, Furuya M, Chua N-H (1989) Rice phytochrome is biologically active in transgenic tobacco. Plant Cell 1:775–782
Keller JM, Shanklin J, Vierstra RD, Hershey HP (1989) Expression of a functional monocotyledonous phytochrome gene in transgenic tobacco plants. EMBO J 8:1005–1012
Khush GS, dela Cruz N (2001) Developing Basmati rices with high yield potential. In: Chaudhary RC, Tran DV, Duffy R (eds) Speciality rices of the world: breeding production and marketing. Science Publishers, NH, pp 15–18
Komari T, Hiei Y, Saito Y, Murai N, Kumashiro T (1996) Vectors carrying two separate T-DNAs for co-transformation of higher plants mediated by Agrobacterium tumefaciens and segregation of transformants free from selection markers. Plant J 10:165–174
Kong SG, Lee DS, Kwak SN, Kim JK, Sohn JK, Kim IS (2004) Characterization of sunlight-grown transgenic rice plants expressing Arabidopsis phytochrome A. Mol Breed 14:35–45
Kyozuka J, McElroy D, Hayakawa T, Xie Y, Wu R, Shimamoto K (1993) Light-regulated and cell-specific expression of tomato rbcS-gusA and rice rbcS-gusA fusion genes in transgenic rice. Plant Physiol 102: 991–1000
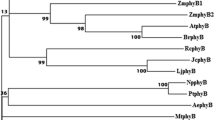
Mathews S, Sharrock RA (1996) The phytochrome gene family in grasses (Poaceae): a phylogeny and evidence that grasses have a subset of the loci found in dicot angiosperms. Mol Biol Evol 13:1141–1150
McCormac AC, Cherry JR, Hershey HP, Vierstra RD, Smith H (1991) Photobiology of tobacco altered by expression of oat phytochrome. Planta 185:162–170
McCormac AC, Wagner D, Boylan MT, Quail PH, Smith H, Whitelam GC (1993) Photoresponses of transgenic Arabidopsis seedlings expressing introduced phytochrome B-encoding cDNAs: evidence that phytochrome A and phytochrome B have distinct photoregulatory functions. Plant J 4:19–27
Monte E, Alonso JM, Ecker JR, Zhang Y, Li X, Young J, Austin-Phillips S, Quail PH (2003) Isolation and characterization of phyC mutants in Arabidopsis reveals complex crosstalk between phytochrome signaling pathways. Plant Cell 15:1962–1980
Nagaraju J, Kathirvel M, Kumar RR, Siddiq EA, Hasnain SE (2002) Genetic analysis of traditional and evolved Basmati and non-Basmati rice varieties by using fluorescence-based ISSR-PCR and SSR markers. Proc Natl Acad Sci USA 99:5836–5841
Nagatani A, Reed JW, Chory J (1993) Isolation and initial characterization of Arabidopsis mutants that are deficient in phytochrome A. Plant Physiol 102:269–277
Nagatani A, Kay SA, Deak M, Chua N-H, Furuya M (1991) Rice type I phytochrome regulates hypocotyl elongation in transgenic tobacco seedlings. Proc Natl Acad Sci USA 88:5207–5211
Olsen JE, Juntilla O, Nilsen J, Eriksson ME, Martinussen I, Olsson O, Sandberg G, Moritz T (1997) Ectopic expression of oat phytochrome A in hybrid aspen changes critical day length for growth and prevents cold acclimation. Plant J 12:1339–1350
Parks BM, Quail PH (1993) hy8, a new class of Arabidopsis long hypocotyl mutants deficient in functional phytochrome A. Plant Cell 5:39–48
Quail PH (2002) Phytochrome photosensory signalling networks. Nat Rev Mol Cell Biol 3:85–93
Rani NS, Krishnaiah K (2001) Current status and future prospects for improvement of aromatic rices in India. In: Chaudhary RC, Tran DV, Duffy R (eds) Speciality rices of the world: breeding production and marketing. Science Publishers, NH, pp 49–78
Reed JW, Nagpal P, Poole DS, Furuya M, Chory J (1993) Mutations in the gene for the red/far-red light receptor phytochrome B alter cell elongation and physiological responses throughout Arabidopsis development. Plant Cell 5:147–157
Robson PR, McCormac AC, Irvine AS, Smith H (1996) Genetic engineering of harvest index in tobacco through overexpression of a phytochrome gene. Nat Biotechnol 14:995–998
Sawers RJ, Sheehan MJ, Brutnell TP (2005) Cereal phytochromes: targets of selection, targets for manipulation? Trends Plant Sci 10:138–43
Sharrock R A, Clack T (2002) Patterns of expression and normalized levels of the five Arabidopsis phytochromes. Plant Physiol 130:442–456
Sharrock RA, Quail PH (1989) Novel phytochrome sequences in Arabidopsis thaliana: structure, evolution, and differential expression of a plant regulatory photoreceptor family. Genes Dev 3:1745–1757
Shlumukov LR, Barro F, Barcelo P, Lazzeri P, Smith H (2001) Establishment of far-red high irradiance responses in wheat through transgenic expression of an oat phytochrome A gene. Plant Cell Environ 24:703–712
Singh RK, Khush GS, Singh US, Singh AK, Singh S (2000) Breeding aromatic rice for high yield, improved aroma and grain quality. In: Singh RK, Singh US, Khush GS (eds) Aromatic rices. Science Publishers, NH pp 71–105
Smith H (1995) Physiological and ecological function within the phytochrome family. Ann Rev Plant Physiol Plant Mol Biol 46:289–315
Smith H (2000) Phytochromes and light signal perception by plants—an emerging synthesis. Nature 407:585–591
Takano M, Kanegae H, Shinomura T, Miyao A, Hirochika H, Furuya M (2001) Isolation and characterization of rice phytochrome A mutants. Plant Cell 13:521–534
Terry MJ (1997) Phytochrome chromophore-deficient mutants. Plant Cell Environ 20:740–745
Thiele A, Herold M, Lenk I, Quail PH, Gatz C (1999) Heterologous expression of Arabidopsis phytochrome B in transgenic potato influences photosynthetic performance and tuber development. Plant Physiol 120:73–81
Wagner D, Tepperman J, Quail PH (1991) Overexpression of phytochrome B induces a short hypocotyl phenotype in transgenic Arabidopsis. Plant Cell 3:1275–1288
Wang H, Deng, XW (2003) Phytochrome A-regulated signaling network and photomorphogenesis. Trends Plant Sci 8:172–178
Whitelam GC, Johnson E, Peng J, Carol P, Anderson ML, Cowl JS, Harberd NP (1993) phytochrome A null mutants of Arabidopsis display a wild-type phenotype in white light. Plant Cell 5:757–768
Yanovsky MJ, Alconada-Magliano TM, Mazzella MA, Gatz C, Thomas B, Casal J (1998) Phytochrome A affects stem growth, anthocyanin synthesis, sucrose-phosphate-synthase activity and neighbour detection in sunlight-grown potato. Planta 205:235–241
Acknowledgements
We are grateful to Dr. Peter H. Quail (University of California, Berkeley and the USDA Plant Gene Expression Center, Albany, CA) for his generous gift of Arabidopsis PHYA cDNA and two monoclonal antibodies (1.9B5A, 073D). We thank Drs. Andre T. Jagendorf, Thomas G. Owens, Maureen Hanson and Tim L. Setter (all from Cornell University) for critical review of the manuscript. We also thank Mike Stoko, Laura Fabrizio, Joan Lee, Julie Batley, Jenny Lee, Carole Dougherty and Anita Aluisio for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garg, A.K., Sawers, R.J.H., Wang, H. et al. Light-regulated overexpression of an Arabidopsis phytochrome A gene in rice alters plant architecture and increases grain yield. Planta 223, 627–636 (2006). https://doi.org/10.1007/s00425-005-0101-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-005-0101-3