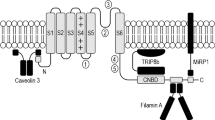
Abstract
HCN channels are the molecular subunits of native funny (f-) channels of cardiac pacemaker cells and neurons. Although funny channels were first functionally described in cardiac cells in the late 1970s, cloning of HCN channels, of which four subunits are known today (HCN1-4), had to wait some 20 years to be accomplished, which delayed the investigation of HCN-related channelopathies. In cardiac pacemaker cells, the main function of f-channels is to contribute substantially to the generation of spontaneous activity of pacemaker cells and control of heart rate. Given this role in cardiac rhythm, it is natural to expect that defective f-channels (or their molecular correlates HCN4 channels) might be responsible for inheritable forms of cardiac arrhythmogenic diseases. Indeed, the recent search for HCN4-related inheritable arrhythmias has resulted in the finding of four different mutations of the hHcn4 gene, which have been reported to be associated with bradycardia and/or more complex arrhythmic conditions. In neurons, HCN channels display a variety of functions including the regulation of excitability, dendritic integration, plasticity, motor learning, generation of repetitive firing, and others. Defective HCN channels may therefore in principle also contribute to pathological conditions in the nervous system. While full evidence for neuronal HCN channelopathies is not yet available, several indications point to a link between temporal lobe and absence epilepsies and altered distribution of HCN1/HCN2 isoforms. Here we briefly review the current knowledge of HCN-related channelopathies in the heart and the brain.



Similar content being viewed by others
References
Clapham DE (1998) Not so funny anymore: pacing channels are cloned. Neuron 21:5–7
DiFrancesco D (1981) A study of the ionic nature of the pace-maker current in calf Purkinje fibres. J Physiol 314:377–393
DiFrancesco D (1981) A new interpretation of the pace-maker current in calf Purkinje fibres. J Physiol 314:359–376
DiFrancesco D (1985) The cardiac hyperpolarizing-activated current, if. Origins and developments. Prog Biophys Mol Biol 46:163–183
DiFrancesco D, Ferroni A, Mazzanti M et al (1986) Properties of the hyperpolarizing-activated current (if) in cells isolated from the rabbit sino-atrial node. J Physiol 377:61–88
Baruscotti M, Barbuti A, Bucchi A (2009) The cardiac pacemaker current. J Mol Cell Cardiol
Mangoni ME, Nargeot J (2008) Genesis and regulation of the heart automaticity. Physiol Rev 88:919–982
DiFrancesco D, Tortora P (1991) Direct activation of cardiac pacemaker channels by intracellular cyclic AMP. Nature 351:145–147
DiFrancesco D (1993) Pacemaker mechanisms in cardiac tissue. Annu Rev Physiol 55:455–472
Bucchi A, Baruscotti M, Robinson RB et al (2003) I(f)-dependent modulation of pacemaker rate mediated by cAMP in the presence of ryanodine in rabbit sino-atrial node cells. J Mol Cell Cardiol 35:905–913
Bucchi A, Baruscotti M, Robinson RB et al (2007) Modulation of rate by autonomic agonists in SAN cells involves changes in diastolic depolarization and the pacemaker current. J Mol Cell Cardiol 43:39–48
DiFrancesco D, Ducouret P, Robinson RB (1989) Muscarinic modulation of cardiac rate at low acetylcholine concentrations. Science 243:669–671
Tardif JC, Ford I, Tendera M et al (2005) Efficacy of ivabradine, a new selective I(f) inhibitor, compared with atenolol in patients with chronic stable angina. Eur Heart J 26:2529–2536
Borer JS, Fox K, Jaillon P et al (2003) Antianginal and antiischemic effects of ivabradine, an I(f) inhibitor, in stable angina: a randomized, double-blind, multicentered, placebo-controlled trial. Circulation 107:817–823
Milanesi R, Baruscotti M, Gnecchi-Ruscone T et al (2006) Familial sinus bradycardia associated with a mutation in the cardiac pacemaker channel. N Engl J Med 354:151–157
Nof E, Luria D, Brass D et al (2007) Point mutation in the HCN4 cardiac ion channel pore affecting synthesis, trafficking, and functional expression is associated with familial asymptomatic sinus bradycardia. Circulation 116:463–470
Schulze-Bahr E, Neu A, Friederich P et al (2003) Pacemaker channel dysfunction in a patient with sinus node disease. J Clin Invest 111:1537–1545
Ueda K, Nakamura K, Hayashi T et al (2004) Functional characterization of a trafficking-defective HCN4 mutation, D553N, associated with cardiac arrhythmia. J Biol Chem 279:27194–27198
Robinson RB, Siegelbaum SA (2003) Hyperpolarization-activated cation currents: from molecules to physiological function. Annu Rev Physiol 65:453–480
Wahl-Schott C, Biel M (2009) HCN channels: structure, cellular regulation and physiological function. Cell Mol Life Sci 66:470–494
Biel M, Wahl-Schott C, Michalakis S et al (2009) Hyperpolarization-activated cation channels: from genes to function. Physiol Rev 89:847–885
Ashcroft FM (1999) Ion channels and disease. Academic Press, London
Jackson HA, Marshall CR, Accili EA (2007) Evolution and structural diversification of hyperpolarization-activated cyclic nucleotide-gated channel genes. Physiol Genomics 29:231–245
Santoro B, Grant SG, Bartsch D et al (1997) Interactive cloning with the SH3 domain of N-src identifies a new brain specific ion channel protein, with homology to Eag and cyclic nucleotide-gated channels. Proc Natl Acad Sci USA 94:14815–14820
Santoro B, Liu DT, Yao H et al (1998) Identification of a gene encoding a hyperpolarization-activated pacemaker channel of brain. Cell 93:717–729
Gauss R, Seifert R, Kaupp UB (1998) Molecular identification of a hyperpolarization-activated channel in sea urchin sperm. Nature 393:583–587
Ludwig A, Zong X, Jeglitsch M et al (1998) A family of hyperpolarization-activated mammalian cation channels. Nature 393:587–591
Vaccari T, Moroni A, Rocchi M et al (1999) The human gene coding for HCN2, a pacemaker channel of the heart. Biochim Biophys Acta 1446:419–425
Seifert R, Scholten A, Gauss R et al (1999) Molecular characterization of a slowly gating human hyperpolarization-activated channel predominantly expressed in thalamus, heart, and testis. Proc Natl Acad Sci USA 96:9391–9396
Ludwig A, Zong X, Stieber J et al (1999) Two pacemaker channels from human heart with profoundly different activation kinetics. EMBO J 18:2323–2329
Zagotta WN, Olivier NB, Black KD et al (2003) Structural basis for modulation and agonist specificity of HCN pacemaker channels. Nature 425:200–205
Brelidze TI, Carlson AE, Zagotta WN (2009) Absence of direct cyclic nucleotide modulation of mEAG1 and hERG1 channels revealed with fluorescence and electrophysiological methods. J Biol Chem 284:27989–27997
Baker K, Warren KS, Yellen G et al (1997) Defective “pacemaker” current (Ih) in a zebrafish mutant with a slow heart rate. Proc Natl Acad Sci USA 94:4554–4559
Warren KS, Baker K, Fishman MC (2001) The slow mo mutation reduces pacemaker current and heart rate in adult zebrafish. Am J Physiol Heart Circ Physiol 281:H1711–H1719
Viscomi C, Altomare C, Bucchi A et al (2001) C terminus-mediated control of voltage and cAMP gating of hyperpolarization-activated cyclic nucleotide-gated channels. J Biol Chem 276:29930–29934
Giorgetti A, Carloni P, Mistrik P et al (2005) A homology model of the pore region of HCN channels. Biophys J 89:932–944
Shin KS, Rothberg BS, Yellen G (2001) Blocker state dependence and trapping in hyperpolarization-activated cation channels: evidence for an intracellular activation gate. J Gen Physiol 117:91–101
Doyle DA, Morais CJ, Pfuetzner RA et al (1998) The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280:69–77
Sigworth FJ (1994) Voltage gating of ion channels. Q Rev Biophys 27:1–40
Bezanilla F (2000) The voltage sensor in voltage-dependent ion channels. Physiol Rev 80:555–592
Chen J, Mitcheson JS, Tristani-Firouzi M et al (2001) The S4–S5 linker couples voltage sensing and activation of pacemaker channels. Proc Natl Acad Sci USA 98:11277–11282
Decher N, Chen J, Sanguinetti MC (2004) Voltage-dependent gating of hyperpolarization-activated, cyclic nucleotide-gated pacemaker channels: molecular coupling between the S4–S5 and C-linkers. J Biol Chem 279:13859–13865
Vaca L, Stieber J, Zong X et al (2000) Mutations in the S4 domain of a pacemaker channel alter its voltage dependence. FEBS Lett 479:35–40
Long SB, Campbell EB, MacKinnon R (2005) Voltage sensor of Kv1.2: structural basis of electromechanical coupling. Science 309:903–908
Prole DL, Yellen G (2006) Reversal of HCN channel voltage dependence via bridging of the S4–S5 linker and Post-S6. J Gen Physiol 128:273–282
Ferrer T, Rupp J, Piper DR et al (2006) The S4–S5 linker directly couples voltage sensor movement to the activation gate in the human ether-a'-go-go-related gene (hERG) K+ channel. J Biol Chem 281:12858–12864
Lu Z, Klem AM, Ramu Y (2002) Coupling between voltage sensors and activation gate in voltage-gated K+ channels. J Gen Physiol 120:663–676
DiFrancesco D (1999) Dual allosteric modulation of pacemaker (f) channels by cAMP and voltage in rabbit SA node. J Physiol 515(Pt 2):367–376
Altomare C, Bucchi A, Camatini E et al (2001) Integrated allosteric model of voltage gating of HCN channels. J Gen Physiol 117:519–532
Barbuti A, Baruscotti M, Altomare C et al (1999) Action of internal pronase on the f-channel kinetics in the rabbit SA node. J Physiol 520(Pt 3):737–744
Wainger BJ, DeGennaro M, Santoro B et al (2001) Molecular mechanism of cAMP modulation of HCN pacemaker channels. Nature 411:805–810
Zhou L, Siegelbaum SA (2007) Gating of HCN channels by cyclic nucleotides: residue contacts that underlie ligand binding, selectivity, and efficacy. Structure 15:655–670
Ulens C, Tytgat J (2001) Functional heteromerization of HCN1 and HCN2 pacemaker channels. J Biol Chem 276:6069–6072
Chen S, Wang J, Siegelbaum SA (2001) Properties of hyperpolarization-activated pacemaker current defined by coassembly of HCN1 and HCN2 subunits and basal modulation by cyclic nucleotide. J Gen Physiol 117:491–504
Much B, Wahl-Schott C, Zong X et al (2003) Role of subunit heteromerization and N-linked glycosylation in the formation of functional hyperpolarization-activated cyclic nucleotide-gated channels. J Biol Chem 278:43781–43786
Altomare C, Terragni B, Brioschi C et al (2003) Heteromeric HCN1–HCN4 channels: a comparison with native pacemaker channels from the rabbit sinoatrial node. J Physiol 549:347–359
Qu J, Kryukova Y, Potapova IA et al (2004) MiRP1 modulates HCN2 channel expression and gating in cardiac myocytes. J Biol Chem 279:43497–43502
Barbuti A, Gravante B, Riolfo M et al (2004) Localization of pacemaker channels in lipid rafts regulates channel kinetics. Circ Res 94:1325–1331
Pian P, Bucchi A, Decostanzo A et al (2007) Modulation of cyclic nucleotide-regulated HCN channels by PIP(2) and receptors coupled to phospholipase C. Pflugers Arch 455:125–145
Baruscotti M, Bucchi A, DiFrancesco D (2005) Physiology and pharmacology of the cardiac pacemaker (“funny”) current. Pharmacol Ther 107:59–79
Lakatta EG, DiFrancesco D (2009) What keeps us ticking: a funny current, a calcium clock, or both? J Mol Cell Cardiol 47:157–170
Mangoni ME, Nargeot J (2001) Properties of the hyperpolarization-activated current (I(f)) in isolated mouse sino-atrial cells. Cardiovasc Res 52:51–64
Verkerk AO, Wilders R, van Borren MM et al (2007) Pacemaker current (I(f)) in the human sinoatrial node. Eur Heart J 28:2472–2478
Baruscotti M, Robinson RB (2007) Electrophysiology and pacemaker function of the developing sinoatrial node. Am J Physiol Heart Circ Physiol 293:H2613–H2623
Wiese C, Grieskamp T, Airik R et al (2009) Formation of the sinus node head and differentiation of sinus node myocardium are independently regulated by Tbx18 and Tbx3. Circ Res 104:388–397
Mommersteeg MT, Hoogaars WM, Prall OW et al (2007) Molecular pathway for the localized formation of the sinoatrial node. Circ Res 100:354–362
Espinoza-Lewis RA, Yu L, He F et al (2009) Shox2 is essential for the differentiation of cardiac pacemaker cells by repressing Nkx2-5. Dev Biol 327:376–385
Brioschi C, Micheloni S, Tellez JO et al (2009) Distribution of the pacemaker HCN4 channel mRNA and protein in the rabbit sinoatrial node. J Mol Cell Cardiol 47:221–227
Liu J, Dobrzynski H, Yanni J et al (2007) Organisation of the mouse sinoatrial node: structure and expression of HCN channels. Cardiovasc Res 73:729–738
Marionneau C, Couette B, Liu J et al (2005) Specific pattern of ionic channel gene expression associated with pacemaker activity in the mouse heart. J Physiol 562:223–234
Shi W, Wymore R, Yu H et al (1999) Distribution and prevalence of hyperpolarization-activated cation channel (HCN) mRNA expression in cardiac tissues. Circ Res 85:e1–e6
Chandler NJ, Greener ID, Tellez JO et al (2009) Molecular architecture of the human sinus node: insights into the function of the cardiac pacemaker. Circulation 119:1562–1575
Camm AJ, Lau CP (2003) Electrophysiological effects of a single intravenous administration of ivabradine (S 16257) in adult patients with normal baseline electrophysiology. Drugs R D 4:83–89
Moosmang S, Biel M, Hofmann F et al (1999) Differential distribution of four hyperpolarization-activated cation channels in mouse brain. Biol Chem 380:975–980
Moosmang S, Stieber J, Zong X et al (2001) Cellular expression and functional characterization of four hyperpolarization-activated pacemaker channels in cardiac and neuronal tissues. Eur J Biochem 268:1646–1652
Thollon C, Bedut S, Villeneuve N et al (2007) Use-dependent inhibition of hHCN4 by ivabradine and relationship with reduction in pacemaker activity. Br J Pharmacol 150:37–46
Liu J, Noble PJ, Xiao G et al (2008) Role of pacemaking current in cardiac nodes: insights from a comparative study of sinoatrial node and atrioventricular node. Prog Biophys Mol Biol 96:294–304
Dobrzynski H, Nikolski VP, Sambelashvili AT et al (2003) Site of origin and molecular substrate of atrioventricular junctional rhythm in the rabbit heart. Circ Res 93:1102–1110
Greener ID, Tellez JO, Dobrzynski H et al (2009) Ion channel transcript expression at the rabbit atrioventricular conduction axis. Circ Arrhythm Electrophysiol 2:305–315
Gaborit N, Le Bouter S, Szuts V et al (2007) Regional and tissue specific transcript signatures of ion channel genes in the non-diseased human heart. J Physiol 582:675–693
Han W, Bao W, Wang Z et al (2002) Comparison of ion-channel subunit expression in canine cardiac Purkinje fibers and ventricular muscle. Circ Res 91:790–797
Shi W, Yu H, Wu J, Zuckerman J, Wymore R (2000) The distribution and prevalence of HCN isoforms in the canine heart and their relation to the voltage dependence of If. Biophys J 78:353A (Abstract)
Remme CA, Verkerk AO, Hoogaars WM et al (2009) The cardiac sodium channel displays differential distribution in the conduction system and transmural heterogeneity in the murine ventricular myocardium. Basic Res Cardiol 104:511–522
Moroni A, Gorza L, Beltrame M et al (2001) Hyperpolarization-activated cyclic nucleotide-gated channel 1 is a molecular determinant of the cardiac pacemaker current I(f). J Biol Chem 276:29233–29241
Ludwig A, Zong X, Hofmann F et al (1999) Structure and function of cardiac pacemaker channels. Cell Physiol Biochem 9:179–186
Robinson RB, Yu H, Chang F et al (1997) Developmental change in the voltage-dependence of the pacemaker current, if, in rat ventricle cells. Pflugers Arch 433:533–535
Hoppe UC, Beuckelmann DJ (1998) Characterization of the hyperpolarization-activated inward current in isolated human atrial myocytes. Cardiovasc Res 38:788–801
Sartiani L, Stillitano F, Cerbai E et al (2009) Electrophysiologic changes in heart failure: focus on pacemaker channels. Can J Physiol Pharmacol 87:84–90
Zhang Q, Huang A, Lin YC et al (2009) Associated changes in HCN2 and HCN4 transcripts and I(f) pacemaker current in myocytes. Biochim Biophys Acta 1788:1138–1147
Chang F, Cohen IS, DiFrancesco D et al (1991) Effects of protein kinase inhibitors on canine Purkinje fibre pacemaker depolarization and the pacemaker current i(f). J Physiol 440:367–384
Tester DJ, Ackerman MJ (2009) Cardiomyopathic and channelopathic causes of sudden unexplained death in infants and children. Annu Rev Med 60:69–84
Stieber J, Herrmann S, Feil S et al (2003) The hyperpolarization-activated channel HCN4 is required for the generation of pacemaker action potentials in the embryonic heart. Proc Natl Acad Sci USA 100:15235–15240
Herrmann S, Stieber J, Stockl G et al (2007) HCN4 provides a ‘depolarization reserve’ and is not required for heart rate acceleration in mice. EMBO J 26:4423–4432
Hoesl E, Stieber J, Herrmann S et al (2008) Tamoxifen-inducible gene deletion in the cardiac conduction system. J Mol Cell Cardiol 45:62–69
Brown HF, DiFrancesco D, Noble SJ (1979) How does adrenaline accelerate the heart? Nature 280:235–236
Pape HC (1996) Queer current and pacemaker: the hyperpolarization-activated cation current in neurons. Annu Rev Physiol 58:299–327
Muller F, Scholten A, Ivanova E et al (2003) HCN channels are expressed differentially in retinal bipolar cells and concentrated at synaptic terminals. Eur J Neurosci 17:2084–2096
Notomi T, Shigemoto R (2004) Immunohistochemical localization of Ih channel subunits, HCN1-4, in the rat brain. J Comp Neurol 471:241–276
Milligan CJ, Edwards IJ, Deuchars J (2006) HCN1 ion channel immunoreactivity in spinal cord and medulla oblongata. Brain Res 1081:79–91
Santoro B, Chen S, Luthi A et al (2000) Molecular and functional heterogeneity of hyperpolarization-activated pacemaker channels in the mouse CNS. J Neurosci 20:5264–5275
Doan TN, Stephans K, Ramirez AN et al (2004) Differential distribution and function of hyperpolarization-activated channels in sensory neurons and mechanosensitive fibers. J Neurosci 24:3335–3343
Wickenden AD, Maher MP, Chaplan SR (2009) HCN pacemaker channels and pain: a drug discovery perspective. Curr Pharm Des 15:2149–2168
Catterall WA, Dib-Hajj S, Meisler MH et al (2008) Inherited neuronal ion channelopathies: new windows on complex neurological diseases. J Neurosci 28:11768–11777
Reid CA, Berkovic SF, Petrou S (2009) Mechanisms of human inherited epilepsies. Prog Neurobiol 87:41–57
Mulley JC, Scheffer IE, Petrou S et al (2003) Channelopathies as a genetic cause of epilepsy. Curr Opin Neurol 16:171–176
Heron SE, Scheffer IE, Berkovic SF et al (2007) Channelopathies in idiopathic epilepsy. Neurotherapeutics 4:295–304
Planells-Cases R, Jentsch TJ (2009) Chloride channelopathies. Biochim Biophys Acta 1792:173–189
Chen K, Aradi I, Thon N et al (2001) Persistently modified h-channels after complex febrile seizures convert the seizure-induced enhancement of inhibition to hyperexcitability. Nat Med 7:331–337
Chen K, Baram TZ, Soltesz I (1999) Febrile seizures in the developing brain result in persistent modification of neuronal excitability in limbic circuits. Nat Med 5:888–894
Brewster A, Bender RA, Chen Y et al (2002) Developmental febrile seizures modulate hippocampal gene expression of hyperpolarization-activated channels in an isoform- and cell-specific manner. J Neurosci 22:4591–4599
Brewster AL, Bernard JA, Gall CM et al (2005) Formation of heteromeric hyperpolarization-activated cyclic nucleotide-gated (HCN) channels in the hippocampus is regulated by developmental seizures. Neurobiol Dis 19:200–207
Santoro B, Baram TZ (2003) The multiple personalities of h-channels. Trends Neurosci 26:550–554
Budde T, Caputi L, Kanyshkova T et al (2005) Impaired regulation of thalamic pacemaker channels through an imbalance of subunit expression in absence epilepsy. J Neurosci 25:9871–9882
Kuisle M, Wanaverbecq N, Brewster AL et al (2006) Functional stabilization of weakened thalamic pacemaker channel regulation in rat absence epilepsy. J Physiol 575:83–100
Strauss U, Kole MH, Brauer AU et al (2004) An impaired neocortical Ih is associated with enhanced excitability and absence epilepsy. Eur J Neurosci 19:3048–3058
Ludwig A, Budde T, Stieber J et al (2003) Absence epilepsy and sinus dysrhythmia in mice lacking the pacemaker channel HCN2. EMBO J 22:216–224
Peng BW, Justice JA, Zhang K et al (2010) Increased basal synaptic inhibition of hippocampal area CA1 pyramidal neurons by an antiepileptic drug that enhances I(H). Neuropsychopharmacol 35:464–472
Acknowledgments
This work was supported by grants from the European Union (LSHM-CT-2006-018676 NORMACOR) and the Ministero dell’ Istruzione dell’Università e della Ricerca (FIRB RBLA035A4X and PRIN 2006055828 to DD and PRIN 2007WB35CW to MB). We wish to thank A. Barbuti and A. Bucchi for their comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baruscotti, M., Bottelli, G., Milanesi, R. et al. HCN-related channelopathies. Pflugers Arch - Eur J Physiol 460, 405–415 (2010). https://doi.org/10.1007/s00424-010-0810-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-010-0810-8