Abstract
Background
This report describes the use of combined laser photocoagulation and intravitreal bevacizumab administration for aggressive zone I retinopathy of prematurity (ROP).
Methods
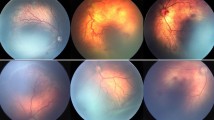
A male patient, born at 25 weeks gestation with a birth weight of 884 g, received indirect laser photocoagulation and a 0.75 mg intravitreal bevacizumab injection to each eye for aggressive stage 3 zone I ROP. Structural outcomes were evaluated 3 months after treatment.
Results
At 3-month follow-up, treatment had resulted in ROP regression, prompt resolution of plus signs and neovascular proliferation in both eyes, and no signs of systemic or ocular adverse events.
Conclusions
The combination of indirect laser photocoagulation and intravitreal bevacizumab injection was well tolerated and induced prompt regression of aggressive zone I ROP.


Similar content being viewed by others
References
Stenkuller PG, Du L, Gilbert C et al (1999) Childhood blindness. J AAPOS 3:26–32
Cryotherapy for Retinopathy of Prematurity Cooperative Group (1990) Multicenter trial of cryotherapy for retinopathy of prematurity. Three-month outcome. Arch Ophthalmol 108:195–204
Early Treatment for Retinopathy of Prematurity Cooperative Group (2003) Revised indication for the treatment of retinopathy of prematurity: results of early treatment for ROP randomized trial. Arch Ophthalmol 121:1684–1696
Flynn JT, Chan-Ling T (2006) Retinopathy of prematurity: Two distinct mechanisms that underlie zone 1 and zone 2 disease. Am J Ophthalmol 142:46–59
Katz X, Kychenthal A, Dorta P (2000) Zone I retinopathy of prematurity. J AAPOS 4:373–376
O’Keefe M, Lanigan B, Long VW (2003) Outcome of zone I retinopathy of prematurity. Acta Ophthalmol Scand 81:614–616
Kychenthal A, Dorta P, Katz X (2006) Zone I retinopathy of prematurity: clinical characteristics and treatment outcomes. Retina 26:S11–S15
Pierce EA, Avery RL, Foley ED et al (1995) Vascular endothelial growth factor/vascular permeability factor expression in a mouse model of retinal neovascularization. Proc Natl Acad Sci USA 92:905–909
Aiello LP, Avery RL, Arrigg PG et al (1994) Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med 331:1480–1487
Pierce EA, Foley ED, Smith LE (1996) Regulation of vascular endothelial growth factor by oxygen in a model of retinopathy of prematurity. Arch Ophthalmol 114:1219–1228
Okamoto N, Tobe T, Hackett SF et al (1997) Animal model. Transgenic mice with increased expression of vascular endothelial growth factor in the retina: a new model of intraretinal and subretinal neovascularization. Am J Pathol 151:281–291
Brady-McCreery KM, McCreery CJ, Sriram V et al (2001) Serum vascular endothelial growth factor and threshold retinopathy of prematurity [abstract]. In: Annual meeting of the American Academy of Ophthalmology; 4th–8th November 2001. American Academy of Ophthalmology, vol 3. Abstract, New Orleans, San Francisco, p 113
Lynch SS, Cheng CM (2007) Bevacizumab for neovascular ocular diseases. Ann Pharmacother 41:614–625
Fung AE, Rosenfeld PJ, Reichel E (2006) The International Intravitreal Bevacizumab Safety Survey: using the internet to assess drug safety worldwide. Br J Ophthalmol 90:1344–1349
Naug H, Browning J, Gole G, Gobe G (2000) Vitreal macrophages express VEGF165 in oxygen-induced retinopathy. Clin Exp Optom 28:48–52
Yourey PA, Gohari S, Su JL, Alderson RF (2000) Vascular endothelial cell growth factors promote the in vitro development of rat photoreceptor cells. J Neurosci 15:6781–6788
Murata T, Nakagawa K, Khalil A, Ishibashi T, Inomata H, Sueishi K (1996) The temporal and spatial vascular endothelial growth factor expression in retinal vasculogenesis of rat neonates. Lab Invest 74:68–79
Shahar J, Avery RL, Heilweil G et al (2006) Electrophysiologic and retinal penetration studies following intravitreal injection of bevacizumab (Avastin). Retina 26:262–269
Manzano RP, Peyman GA, Khan P, Kivilcim M (2006) Testing intravitreal toxicity of bevacizumab (Avastin). Retina 26:257–261
Bakri SJ, Cameron JD, McCannel CA, Pulido JS, Marler RJ (2006) Absence of histologic retinal toxicity of intravitreal bevacizumab in a rabbit model. Am J Ophthalmol 142:162–164
Shah PK, Narendran V, Tawansy KA, Raghuram A, Narendran K (2007) Intravitreal bevacizumab (Avastin) for post laser anterior segment ischemia in aggressive posterior retinopathy of prematurity. Indian J Ophthalmol 55:75–76
Travassos A, Teixeira S, Ferreira P et al (2007) Intravitreal bevacizumab in aggressive posterior retinopathy of prematurity. Ophthalmic Surg Lasers Imaging 38:233–237
Avery RL, Pearlman J, Pieramici DJ et al (2006) Intravitreal bevacizumab (Avastin) in the treatment of proliferative diabetic retinopathy. Ophthalmology 113:1695–1705
Beer PM, Wong SJ, Hammad AM, Falk NS, O’Malley MR, Khan S (2006) Vitreous levels of unbound bevacizumab and unbound vascular endothelial growth factor in two patients. Retina 26:871–876
Repka MX, Palmer EA, Tung B (2000) Involution of retinopathy of prematurity. Arch Ophthalmol 118:645–649
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chung, E.J., Kim, J.H., Ahn, H.S. et al. Combination of laser photocoagulation and intravitreal bevacizumab (Avastin®) for aggressive zone I retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol 245, 1727–1730 (2007). https://doi.org/10.1007/s00417-007-0661-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-007-0661-y