Abstract
Objectives
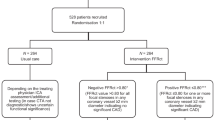
More than 3.5 million invasive coronary angiographies (ICA) are performed in Europe annually. Approximately 2 million of these invasive procedures might be reduced by noninvasive tests because no coronary intervention is performed. Computed tomography (CT) is the most accurate noninvasive test for detection and exclusion of coronary artery disease (CAD). To investigate the comparative effectiveness of CT and ICA, we designed the European pragmatic multicentre DISCHARGE trial funded by the 7th Framework Programme of the European Union (EC-GA 603266).
Methods
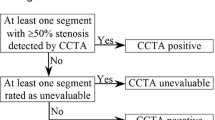
In this trial, patients with a low-to-intermediate pretest probability (10–60 %) of suspected CAD and a clinical indication for ICA because of stable chest pain will be randomised in a 1-to-1 ratio to CT or ICA. CT and ICA findings guide subsequent management decisions by the local heart teams according to current evidence and European guidelines.
Results
Major adverse cardiovascular events (MACE) defined as cardiovascular death, myocardial infarction and stroke as a composite endpoint will be the primary outcome measure. Secondary and other outcomes include cost-effectiveness, radiation exposure, health-related quality of life (HRQoL), socioeconomic status, lifestyle, adverse events related to CT/ICA, and gender differences.
Conclusions
The DISCHARGE trial will assess the comparative effectiveness of CT and ICA.
Key Points
• Coronary artery disease (CAD) is a major cause of morbidity and mortality.
• Invasive coronary angiography (ICA) is the reference standard for detection of CAD.
• Noninvasive computed tomography angiography excludes CAD with high sensitivity.
• CT may effectively reduce the approximately 2 million negative ICAs in Europe.
• DISCHARGE addresses this hypothesis in patients with low-to-intermediate pretest probability for CAD.





Similar content being viewed by others
References
WHO (2008) The global burden of disease. WHO, Geneva
Moschovitis A, Cook S, Meier B (2010) Percutaneous coronary interventions in Europe in 2006. EuroIntervention 6:189–194
Patel MR, Peterson ED, Dai D et al (2010) Low diagnostic yield of elective coronary angiography. N Engl J Med 362:886–895
Noto TJ Jr, Johnson LW, Krone R et al (1991) Cardiac catheterization 1990: a report of the registry of the Society for Cardiac Angiography and Interventions (SCA&I). Catheter Cardiovasc Diagn 24:75–83
Scanlon PJ, Faxon DP, Audet AM et al (1999) ACC/AHA guidelines for coronary angiography. A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines (Committee on Coronary Angiography). Developed in collaboration with the Society for Cardiac Angiography and Interventions. J Am Coll Cardiol 33:1756–1824
Schuetz GM, Zacharopoulou NM, Schlattmann P, Dewey M (2010) Meta-analysis: noninvasive coronary angiography using computed tomography versus magnetic resonance imaging. Ann Intern Med 152:167–177
von Ballmoos MW, Haring B, Juillerat P, Alkadhi H (2011) Meta-analysis: diagnostic performance of low-radiation-dose coronary computed tomography angiography. Ann Intern Med 154:413–420
Montalescot G, Sechtem U, Achenbach S et al (2013) 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J 34:2949–3003
Genders TS, Ferket BS, Dedic A et al (2012) Coronary computed tomography versus exercise testing in patients with stable chest pain: comparative effectiveness and costs. Int J Cardiol. doi:10.1016/j.ijcard.2012.03.151,
Dewey M, Hamm B (2007) Cost effectiveness of coronary angiography and calcium scoring using CT and stress MRI for diagnosis of coronary artery disease. Eur Radiol 17:1301–1309
Schlattmann P, Schuetz GM, Dewey M (2011) Influence of coronary artery disease prevalence on predictive values of coronary CT angiography: a meta-regression analysis. Eur Radiol 21:1904–1913
Fox K, Garcia MA, Ardissino D et al (2006) Guidelines on the management of stable angina pectoris: executive summary: The Task Force on the Management of Stable Angina Pectoris of the European Society of Cardiology. Eur Heart J 27:1341–1381
Douglas PS, Hoffmann U, Patel MR et al (2015) Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med 372:1291–1300
The SCOT-HEART Investigators (2015) CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet 385:2383–2391
Chan AW, Tetzlaff JM, Gotzsche PC et al (2013) SPIRIT 2013 explanation and elaboration: guidance for protocols of clinical trials. BMJ 346:e7586
Chan AW, Tetzlaff JM, Altman DG et al (2013) SPIRIT 2013 statement: defining standard protocol items for clinical trials. Ann Intern Med 158:200–207
Lawlor DA, Adamson J, Ebrahim S (2003) Performance of the WHO Rose angina questionnaire in post-menopausal women: are all of the questions necessary? J Epidemiol Community Health 57:538–541
Perk J, De Backer G, Gohlke H et al (2012) European guidelines on cardiovascular disease prevention in clinical practice (version 2012): The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur Heart J 33:1635–1701
Tunis SR, Stryer DB, Clancy CM (2003) Practical clinical trials: increasing the value of clinical research for decision making in clinical and health policy. JAMA 290:1624–1632
Mullins CD, Whicher D, Reese ES, Tunis S (2010) Generating evidence for comparative effectiveness research using more pragmatic randomized controlled trials. Pharmacoeconomics 28:969–976
Thorpe KE, Zwarenstein M, Oxman AD et al (2009) A pragmatic-explanatory continuum indicator summary (PRECIS): a tool to help trial designers. CMAJ 180:E47–E57
Zimmermann E, Germershausen C, Greupner J et al (2010) Improvement of skills and knowledge by a hands-on cardiac CT course: before and after evaluation with a validated questionnaire and self-assessment. Röfo 182:589–593
Maruish ME (2012) User’s manual for the SF-12v2 health survey, 3rd edn. QualityMetric, Lincoln
EuroQolGroup (1990) EuroQol-a new facility for the measurement of health-related quality of life. Health Policy 16:199–208
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67:361–370
Griffin S, Claxton K, Sculpher M (2008) Decision analysis for resource allocation in health care. J Health Serv Res Policy 13:23–30
Ramsey SD, Willke RJ, Glick H et al (2015) Cost-effectiveness analysis alongside clinical trials II-An ISPOR Good Research Practices Task Force report. Value Health 18:161–172
Schuetz GM, Schlattmann P, Achenbach S et al (2013) Individual patient data meta-analysis for the clinical assessment of coronary computed tomography angiography: protocol of the Collaborative Meta-Analysis of Cardiac CT (CoMe-CCT). Syst Rev 2:13
Kernan WN, Viscoli CM, Makuch RW, Brass LM, Horwitz RI (1999) Stratified randomization for clinical trials. J Clin Epidemiol 52:19–26
Therneau TM, Grambsch P, Pankratz VS (2003) Penalized survival models and frailty. J Comput Graph Stat 12:156–175
Therneau T (2012) coxme: mixed effects Cox models. R package version 2.2-3. http://CRAN.R-project.org/package=coxme
R Development Core Team (2012) R: a language and environment for statistical computing. http://www.R-project.org. R Foundation for Statistical Computing, Vienna, Austria
Schlattmann P (2009) Medical applications of finite mixture models. Springer, Berlin
Schafer JL (1999) Multiple imputation: a primer. Stat Methods Med Res 8:3–15
Keaven Anderson (2012) gsDesign: group sequential design. R package version 2.7-04. http://CRAN.R-project.org/package=gsDesign
Dewey M, Rief M, Martus P et al (2016) Evaluation of computed tomography in patients with atypical angina or chest pain clinically referred for invasive coronary angiography: randomised controlled trial. BMJ 355:i5441
Williams MC, Hunter A, Shah AS et al (2016) Use of coronary computed tomographic angiography to guide management of patients with coronary disease. J Am Coll Cardiol 67:1759–1768
Kip KE, Hollabaugh K, Marroquin OC, Williams DO (2008) The problem with composite end points in cardiovascular studies: the story of major adverse cardiac events and percutaneous coronary intervention. J Am Coll Cardiol 51:701–707
The DISCHARGE Trial (2015) www.dischargetrial.eu. Accessed 25 Mar 2015
Chow BJ, Small G, Yam Y et al (2015) Prognostic and therapeutic implications of statin and aspirin therapy in individuals with nonobstructive coronary artery disease: results from the CONFIRM (coronary CT angiography evaluation for clinical outcomes: an international multicenter registry) registry. Arterioscler Thromb Vasc Biol. doi:10.1161/ATVBAHA.114.304351
Dewey M, Zimmermann E, Deissenrieder F et al (2009) Noninvasive coronary angiography by 320-row CT with lower radiation exposure and maintained diagnostic accuracy: comparison of results with cardiac catheterization in a head-to-head pilot investigation. Circulation 120:867–875
Pontone G, Andreini D, Bartorelli AL et al (2013) A long-term prognostic value of CT angiography and exercise ECG in patients with suspected CAD. JACC Cardiovasc Imaging 6:641–650
Linde JJ, Hove JD, Sorgaard M et al (2015) Long-term clinical impact of coronary CT angiography in patients with recent acute-onset chest pain: the randomized controlled CATCH trial. JACC Cardiovasc Imaging 8:1404–1413
Schuetz GM, Schlattmann P, Dewey M (2012) Use of 3x2 tables with an intention to diagnose approach to assess clinical performance of diagnostic tests: meta-analytical evaluation of coronary CT angiography studies. BMJ 345:e6717
Acknowledgments
The scientific guarantor of this publication is Prof. Marc Dewey, M.D. Ph.D. The authors of this manuscript declare relationships with the following companies, foundations and government or university grants: Abbott, Academy of Finland, Centre of Excellence programs, Actelion, American Thoracic Society, Amgen, AP Møller og hustru Chastine McKinney Møllers Fond, Astra-Zeneca, Athera Biotechnologies AB, B.Braun Medical, Bayer, Bayer-Schering, Berlin Chemie, Beuth Hochschule für Technik Berlin, Biosensors, Biotie Therapies, Biotronik, Boehringer-Ingelheim, Boston Scientific, Bracco, Braun, Cardiac MR Academy Berlin, CardiRad Ltd, Comac-Medical, Cordis, Covance, Directorate-General for Health and Food Safety, Eckert & Ziegler Radiopharma GmbH, Edwards Lifesciences, Endocyte Inc, European Regional Development Fund, European Society of Cardiology, European Union, F.Hoffmann-La Roche, Finnish Foundation for Cardiovascular Research, Fundação para a Ciência e Tecnologia, Portugal, GE Healthcare, Gedeon Richter, German Federal Ministry of Education and Research (BMBF), German Research Foundation (DFG), Guerbet, Guidotti,Intermeetings Padova, Irish Lung Foundation, Jansen-Cilag, Johnson & Johnson, KRKA, Lantheus Inc, Medtronic, Medtronik, Menarini, Merck, Merck Sharp & Dohme, MSD, National Heart, Lung, and Blood Institute (NHLBI), Novartis, Orion Pharma, Pfizer, Pharma Swiss, Philips, Piramal, Portuguese Society of Cardiology, Quintiles, Research Council of Rigshopitalet, Roche, Sandoz, Sanofi Aventis, Schering, Servier, Siemens, Sigma Tau, Southeastern Health and Social Care Trust Innovation Research Development Group Fund, Spanish Society of Cardiology, St. Jude Medical, Stiftung Charité (Berlin Institute of Health), Takeda,TEKES Finland, The Danish Agency for Science Technology and Innovation by The Danish Council for Strategic Research, The Danish Heart Foundation, The John and Birthe Meyer Foundation, The Lundbeck Foundation, Toshiba Medical Systems, Turku University Hospital research funds, VITAL Images Inc., Zealand A/S
This study has received funding by the Seventh Framework Programme of the European Union (EC-GA 603266). Prof. Peter Schlattmann kindly provided statistical advice for this manuscript. Institutional review board approval was obtained. Written informed consent will be obtained from all subjects (patients) for the study. Study subjects or cohorts have not been previously reported in another journal/study. Methodology: prospective/retrospective, randomised controlled trial, multicenter study.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Napp, A.E., Haase, R., Laule, M. et al. Computed tomography versus invasive coronary angiography: design and methods of the pragmatic randomised multicentre DISCHARGE trial. Eur Radiol 27, 2957–2968 (2017). https://doi.org/10.1007/s00330-016-4620-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4620-z