Abstract
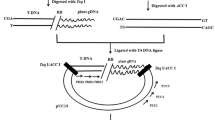
The stability of the inserted transgenes and particularly the junction regions of transgenic events is a concern of food labeling, traceability and post release monitoring, as these regions are used for development of event-specific DNA-based detection methods. During the standard agricultural breeding practices, the transgenic lines can be exposed to completely different conditions than those in the laboratory environment. Some of these conditions have the potential to affect the stability of the transgenic locus and the surrounding DNA. As tissue culture is recognized as a stressful and mutagenic factor, we have analyzed the effect of this process on the stability of the junction regions at nucleotide level in five Arabidopsis thaliana transgenic lines in comparison with the respective integration loci in ColO and C24 ecotypes. No indication of any kind of alteration at nucleotide level of the junctions was found. The relevance of the stability of the plant–T-DNA junction regions for application of the DNA-based methods in commercial transgenic plants is discussed.

Similar content being viewed by others
References
Alonso JM et al (2003) Genome wide insertional mutagenesis of Arabidopsis thaliana. Science 301:653–657
Arencibia A, Gentinetta E, Cuzzoni E, Castiglione S, Kohli A, Vain P, Leech M, Christou P, Sala F (1998) Molecular analysis of the genome of transgenic rice (Oryza sativa L.) plants produced by paricle bombardment or intact cell electroporation. Mol Breed 4:99–109
Brettel RIS, Dennis ES, Scowcroft WR, Peacock WJ (1986) Molecular analysis of a somaclonal mutant of maize alcohol dehydrogenase. Mol Gen Genet 202:235–239
Brown PTH, Göbel E, Lörtz H (1991) RFLP analysis of Zea mays callus cultures and their regenerated plants. Theor Appl Genet 81:227–232
Carmona ER, Arencibia AD, Lopez J, Simpson J, Vargas D, Sala F (2005) Analysis of genomic variability in transgenic sugar cane plants produced by Agrobacterium mediated transformation. Plant Breed 124:33–38
De Buck S, Windels P, De Loose M, Depicker A (2004) Single-copy T-DNAs integrated at different positions in the Arabidopsis genome display uniform and comparable β-glucuronidase accumulation levels. Cell Mol Life Sci 61:2632–2645
De Buck S, Jakobs A, Van Montagu M, Depicker A (1998) Agrobacterium tumefaciens transformation and cotransformation in Arabidopsis thaliana root explants and tobacco protoplasts. Mol Plant Microbe Interact 11:449–457
Dellaporta S, Wood J, Hicks JB (1983) A plant DNA minipreparation: version 2. Plant Mol Biol Rep 1:19–22
Dennis ES, Brettel RIS, Peacock WJ (1987) A tissue culture induced Adh1 null mutant of maize results from a single base pair change. Mol Gen Genet 210:181–183
Forsbach B, Schubert D, Lechtenberg B, Gils M, Schmidt R (2003) A comprehensive characterization of single-copy T-DNA insertions in the Arabidopsis thaliana genome. Plant Mol Biol 52:161–176
Gorbunova V, Levy AA (1997) Non-homologous DNA end joining in plant cells is associated with deletions and filler DNA insertions. Nucleic Acids Res 25:4650–4657
Harms CT, Armour SL, DiMairo JJ, Middlesteadt LA, Murray D, Negrotto DV, Thompson-Taylor H, Weymann K, Montoya AL, Shillito RD, Jen GC (1992) Herbicide resistance due to amplification of a mutant acetohydroxyacid synthase gene. Mol Gen Genet 233:427–435
Holst-Jensen A, Rønning S, Løvseth A, Berdal K (2003) PCR technology for screening and quantification of genetically modified organisms (GMOs). Anal Bioanal Chem 375:985–993
Kohli A, Twyman R, Abranches R, Wegel E, Stoger E, Christou P (2003) Transgene integration, organization and interaction in plants. Plant Mol Biol 52:247–258
Kovalchuk I, Kovalchuk O, Hohn B (2000) Genome-wide variation of the somatic mutation frequency in transgenic plants. EMBO J 17:4431–4438
Larkin PJ, Scowcroft WR (1981) Somaclonal variation—a novel source of variability from cell cultures for plant improvement. Theor Appl Genet 60:197–214
Lee KY, Townsend J, Tepperman J, Black M, Chui CF, Mazur B, Dunsmuir P, Bedbrook J (1988) The molecular basis of sulfonylurea herbicide resistance in tobacco. EMBO J 7:1241–1248
Madlung A, Comai L (2004) The effect of stress on genome regulation and structure. Ann Bot 94:481–495
McCallum C, Comai L, Greene EA, Henikoff S (2000) Targeted screening for induced mutations. Nat Biotechnol 18:455–457
Nakano A, Suzuki G, Yamamoto M, Turnbull K, Rahman S, Mukai Y (2005) Rearrangements of large-insert in transgenic rice T-DNA. Mol Gen Genet 273:123–129
Nickerson D, Tobe V, Taylor S (1997) PolyPhred: authomating the detection and genotyping of single nucleotide substitutions using fluorescence-based sequencing. Nucleic Acid Res 25:2745–2751
Phillips RL, Kaepler SM, Olhoft P (1994) Genetic instability of plant tissue culture: breakdown of normal controls. Proc Natl Acad Sci USA 91:5222–5226
Puchta H, Swoboda P, Gal S, Blot M, Hohn B (1995) Somatic intrachromosomal homologous recombination effects in populations of plant siblings. Plant Mol Biol 28:281–192
Risseeuw E, Franke-Van Dijk MEI, Hooykaas P (1997) Gene targeting and instability of Agrobacterium T-DNA loci in the plant genome. Plant J 5:2599–2607
Rozen S, Skaletsky HJ (2000) Primer3 on the WWW for general users and for biologist programmers. In: Krawetz S, Misener S (eds) Bioinformatics methods and protocols: methods in molecular biology. Humana Press, Totowa, NJ, pp 365–386
Theuns I, Windels P, De Buck S, Depicker A, Van Bockstaele E, De Loose M (2002) Identification and characterization of T-DNA inserts via T-DNA fingerprinting. Euphytica 123:75–84
Valvekens D, Van Montagu M, Van Lisebettens M (1988) Agrobacterium tumefaciens mediated transformation of Arabidopsis thaliana root explants using kanamycin selection. Proc Natl Acad Sci USA 85:5536–5540
Windels P, De Buck S, Van Bockstaele E, De Loose M, Depicker A (2003) T-DNA integration in Arabidopsis chromosomes: presence and origin of filler DNA sequences. Plant Physiol 133:2061–2068
Zucchi MI, Arizono H, Morais VA, Fungaro MHP, Vieira MLC (2002) Genetic instability of sugarcane plants derived from meristem cultures. Genet Mol Biol 25(1):91–96
Acknowledgements
This work is performed in the frame of project “Development of methods for the detection, identification and quantification of genetically modified organisms,” contact no. 18687-2001-11 F1ED ISP BE, funded by EC-JRC. We thank Elien Glorieus, Kristien Janssens and Cindy Merckaert for their excellent experimental work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Puigdomenech
Rights and permissions
About this article
Cite this article
Papazova, N., Windels, P., Depicker, A. et al. Sequence stability of the T-DNA – plant junctions in tissue culture in Arabidopsis transgenic lines. Plant Cell Rep 25, 1362–1368 (2006). https://doi.org/10.1007/s00299-006-0192-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-006-0192-8