Abstract
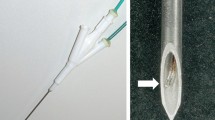
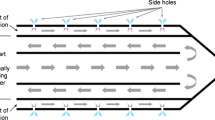
This experimental study was undertaken to compare radiofrequency (RF) ablation zones created by internally cooled (IC) and internally cooled wet (ICW) electrodes. IC and ICW electrodes with a 2-cm exposed active tip were used to induce 30 ablation zones in 10 explanted bovine livers with a 12-min ablation time, respectively. In addition, two kinds of electrodes produced 16 ablation zones in five living porcine livers, respectively. In explanted bovine livers using IC and ICW electrodes, the mean long-axis diameter, short-axis diameter, volume, and variable coefficient of long-axis diameters of the ablation zones were 3.01 cm, 2.62 cm, 11.08 cm3, 10%, 5.28 cm, 5.07 cm, 73.48 cm3, and 14%, respectively. In living porcine livers using IC and ICW electrodes, the corresponding values were 2.62 cm, 2.00 cm, 5.76 cm3, 15%, 3.84 cm, 2.89 cm, 18.50 cm3, and 25%, respectively. In both ex vivo and in vivo livers, long-axis diameters, short-axis diameters, volumes, and variable coefficients for the use of ICW electrodes were significantly greater than for the use of IC electrodes (each p < 0.05). ICW electrodes produced significantly larger ablation zones than IC electrodes in both ex vivo and in vivo livers, but the ablation zones induced by IC electrodes were more reproducible.



Similar content being viewed by others
References
Dodd GD III, Soulen MC, Kane RA et al (2000) Minimally invasive treatment of malignant hepatic tumors: at the threshold of a major breakthrough. Radiographics 20:9–27
Tateishi R, Shiina S, Teratani T et al (2005) Percutaneous radiofrequency ablation for hepatocellular carcinoma. An analysis of 1000 cases. Cancer 103:1201–1209
Choi D, Lim HK, Rhim H, Kim YS et al (2007) Percutaneous radiofrequency ablation for early-stage hepatocellular carcinoma as a first-line treatment: long-term results and prognostic factors in a large single-institution series. Eur Radiol 17:684–692
de Baere T, Denys A, Wood BJ et al (2001) Radiofrequency liver ablation: experimental comparative study of water-cooled versus expandable systems. AJR 176:187–192
Goldberg SN, Stein MC, Gazelle GS, Sheiman RG, Kruskal JB, Clouse ME (1999) Percutaneous radiofrequency tissue ablation: optimization of pulsed-radiofrequency technique to increase coagulation necrosis. J Vasc Interv Radiol 10:907–916
Kim SK, Gu MS, Hong HP, Choi D, Chae SW (2007) CT findings after radiofrequency ablation in rabbit livers: comparison of internally cooled electrodes, perfusion electrodes, and internally cooled perfusion electrodes. J Vasc Interv Radiol 18:1417–1427
Lee JM, Han JK, Kim SH et al (2005) Bipolar radiofrequency ablation using wet-cooled electrodes: an in vitro experimental study in bovine liver. AJR 184:391–397
Lee JM, Han JK, Kim SH et al (2005) Radiofrequency ablation in the liver using two cooled-wet electrodes in the bipolar mode. Eur Radiol 15:2163–2170
Lee JM, Han JK, Kim SH et al (2004) Optimization of wet radiofrequency ablation using a perfused-cooled electrode: a comparative study in ex vivo bovine livers. Korean J Radiol 5:250–257
Lee JM, Han JK et al (2004) Comparison of wet radiofrequency ablation with dry radiofrequency ablation and radiofrequency ablation using hypertonic saline preinjection: ex vivo bovine liver. Korean J Radiol 5:258–265
Lee JM, Han JK, Kim SH, Sohn KL, Choi SH, Choi BI (2005) Bipolar radiofrequency ablation in ex vivo bovine liver with the open-perfused system versus the cooled-wet system. Eur Radiol 15:759–764
Lorentzen T (1996) A cooled needle electrode for radiofrequency tissue ablation: thermodynamic aspects of improved performance compared with conventional needle design. Acad Radiol 3:556–563
Miao Y, Ni Y, Yu J, Marchal G (2000) A comparative study on validation of a novel cooled-wet electrode for radiofrequency liver ablation. Invest Radiol 35:438–444
Ni Y, Miao Y, Mulier S, Yu J, Baert AL, Marchal G (2000) A novel “cooled-wet” electrode for radiofrequency ablation. Eur Radiol 10:852–854
Solbiati L, Goldberg SN, Ierace T et al (1997) Hepatic metastases: percutaneous radio-frequency ablation with cooled-tip electrodes. Radiology 205:367–373
Burdio F, Guemes A, Burdio JM et al (2003) Bipolar saline-enhanced electrode for radiofrequency ablation: results of experimental study of in vivo porcine liver. Radiology 229:447–456
McGahan JP, Brock JM, Tesluk H, Gu WZ, Schneider P, Browning PD (1992) Hepatic ablation with use of radio-frequency electrocautery in the animal model. J Vasc Interv Radiol 3:291–297
Lencioni R, Cioni D, Crocetti L et al (2005) Early-stage hepatocellular carcinoma in patients with cirrhosis: long-term results of percutaneous image-guided radiofrequency ablation. Radiology 234:961–967
Yamakado K, Nakatsuka A, Ohmori S et al (2002) Radiofrequency ablation combined with chemoembolization in hepatocellular carcinoma: treatment response based on tumor size and morphology. J Vasc Interv Radiol 13:1225–1232
Hoey MF, Mulier PM, Leveillee RJ, Hulbert JC (1997) Transurethral prostate ablation with saline electrode allows controlled production of larger lesions than conventional methods. J Endourol 11:279–284
Livraghi T, Goldberg SN, Monti F et al (1997) Saline-enhanced radio-frequency tissue ablation in the treatment of liver metastases. Radiology 202:205–210
Acknowledgment
This work was supported by a 2008 research grant from the Korean Liver Cancer Study Group.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cha, J., Choi, D., Lee, M.W. et al. Radiofrequency Ablation Zones in Ex Vivo Bovine and In Vivo Porcine Livers: Comparison of the Use of Internally Cooled Electrodes and Internally Cooled Wet Electrodes. Cardiovasc Intervent Radiol 32, 1235–1240 (2009). https://doi.org/10.1007/s00270-009-9600-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-009-9600-0