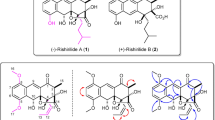
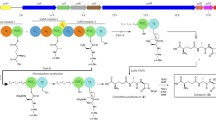
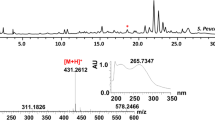
Abstract
Mupirocin is a polyketide antibiotic produced by Pseudomonas fluorescens. The biosynthetic cluster encodes 6 type I polyketide synthase multifunctional proteins and 29 single function proteins. The biosynthetic pathway belongs to the trans-AT group in which acyltransferase activity is provided by a separate polypeptide rather than in-cis as found in the original type I polyketide synthases. Special features of this group are in-cis methyltransferase domains and a trans-acting HMG-CoA synthase-cassette which insert α- and β- methyl groups respectively while enoyl reductase domains are absent from the condensing modules. In addition, for the mupirocin system, there is no obvious loading mechanism for initiation of the polyketide chain and many aspects of the pathway remain to be elucidated. Mupirocin inhibits isoleucyl-tRNA synthetase and has been used since 1985 to help prevent infection by methicillin-resistant Staphylococcus aureus, particularly within hospitals. Resistance to mupirocin was first detected in 1987 and high-level resistance in S. aureus is due to a plasmid-encoded second isoleucyl-tRNA synthetase, a more eukaryotic-like enzyme. Recent analysis of the biosynthetic pathway for thiomarinols from marine bacteria opens up possibilities to modify mupirocin so as to overcome this resistance.



Similar content being viewed by others
References
Alexander RG, Clayton JP, Luk K, Rogers NH, King TJ (1978) The chemistry of pseudomonic acid. Part 1. The absolute configuration of pseudomonic acid A. J Chem Soc Perkin Trans I:561–565
Aron ZD, Fortin PD, Calderone CT, Walsh CT (2007) FenF: servicing the Mycosubtilin synthetase assembly line in trans. Chembiochem 8:613–616
Basker MJ, Comber KR, Clayton PJ, Hannan PT, Mizen LW, Rogers NH, Slocombe B, Sutherland R (1980) Ethylmonate A: a semisynthetic antibiotic derived from pseudomonic acid A. In: Nelson JD, Grassi C (eds) Current chemotherapy and infectious disease, vol 1. American Society for Microbiology, Washington, pp 471–473
Bumpus SB, Magarvey NA, Kelleher NL, Walsh CT, Calderone CT (2008) Polyunsaturated fatty-acid-like trans-enoyl reductases utilized in polyketide biosynthesis. J Americal Chem Soc 130:11614–11616
Bush K, Macielag M (2000) New approaches in the treatment of bacterial infections. Curr Opin Chem Biol 4:433–439
Butcher RA, Schroeder FC, Fischbach MA, Straight PD, Kolter R, Walsh CT, Clardy J (2007) The identification of bacillaene, the product of the PksX megacomplex in Bacillus subtilis. Proc Natl Acad Sci USA 104(5):1506–1509
Caffrey AR, Quilliam BJ, LaPlante KL (2010) Risk factors associated with mupirocin resistance in meticillin-resistant Staphylococcus aureus. J Hosp Infect. doi:10.1016/j.jhin.2010.06.023
Cane DE (2010) Programming of erythromycin biosynthesis by a modular polyketide synthase. J Biol Chem 285(36):27515–27523
Chain EB, Mellows G (1974) Structure of pseudomonic acid, an antibiotic from Pseudomonas fluorescens. Journal of the Chemical Society, Chemical Communications 1974:847–848
Chain EB, Mellows G (1977) Pseudomonic acid. Part 1. The structure of pseudomonic acid A, a novel antibiotic produced by Pseudomonas fluorescens. J Chem Soc Perkin Trans I:294–309
Chang Z, Sitachitta N, Rossi JV, Roberts MA, Flatt PM, Jia J, Sherman DH, Gerwick WH (2004) Biosynthetic pathway and gene cluster analysis of curacin A, an antitubulin natural product from the tropical marine cyanobacterium Lyngbya majuscula. J Nat Prod 67:1356–1367
Chen X-H, Vater J, Piel J, Franke P, Scholz R, Schneider K, Koumoutsi A, Hitzeroth G, Grammel N, Strittmatter AW, Gottschalk G, Süssmuth RD, Borriss R (2006) Structural and functional characterization of three polyketide synthase gene clusters in Bacillus amyloliquefaciens FZB 42. J Bacteriol 188(11):4024–4036
Cheng Y-Q, Tang G-L, Shen B (2003) Type I polyketide synthase requiring a discrete acyltransferase for polyketide biosynthesis. Proc Natl Acad Sci USA 100(6):3149–3154
Cookson BD, Lacey RW, Noble WC, Reeves DS, Wise R, Redhead RJ (1990) Mupirocin-resistant Staphylococcus aureus. Lancet 335:1095–1096
Cooper SM, Cox RJ, Crosby J, Crump MP, Hothersall J, Laosripaiboon W, Simpson TJ, Thomas CM (2005a) Mupirocin W, a novel pseudomonic acid produced by targeted mutation of the mupirocin biosynthetic gene cluster. The Royal Society of Chemistry Chemical Communications 2005:1179–1181
Cooper SM, Laosripaiboon W, Rahman AS, Hothersall J, El-Sayed AK, Winfield C, Crosby J, Cox RJ, Simpson TJ, Thomas CM (2005b) Shift to pseudomonic acid B production in P. fluorescens NCIMB10586 by mutation of mupirocin tailoring genes mupO, mupU, mupV, and macpE. Chem Biol 12:825–833
Critchley IA, Ochsner UA (2008) Recent advances in the preclinical evaluation of the topical antibacterial agent REP8839. Curr Opin Chem Biol 12:409–417
Dacre JE, Emmerson AM, Jenner EA (1983) Nasal carriage of gentamicin and methicillin resistant Staphylococcus aureus treated with topical pseudomonic acid. Lancet 322(8357):1036
DelVecchio F, Petkovic H, Kendrew SG, Low L, Wilkinson B, Lill R, Cortés J, Rudd BAM, Staunton J, Leadlay PF (2003) Active-site residue, domain and module swaps in modular polyketide synthases. J Ind Microbiol Biotechnol 30:489–494
Dyke KGH, Curnock SP, Golding M, Noble WC (1991) Cloning of the gene conferring resistance to mupirocin in Staphylococcus aureus. FEMS Microbiol Lett 77:195–198
Edwards DJ, Marquez BL, Nogle LM, McPhail K, Goeger DE, Roberts MA, Gerwick WH (2004) Structure and biosynthesis of the jamaicamides, new mixed polyketide-peptide neurotoxins from the marine cyanobacterium Lyngbya majuscula. Chem Biol 11:817–833
El-Sayed AK, Hothersall J, Thomas CM (2001) Quorum-sensing-dependent regulation of biosynthesis of the polyketide antibiotic mupirocin in Pseudomonas fluorescens NCIMB 10586. Microbiology 147:2127–2139
El-Sayed AK, Hothersall J, Cooper SM, Stephens E, Simpson TJ, Thomas CM (2003) Characterization of the mupirocin biosynthesis gene cluster from Pseudomonas fluorescens NCIMB 10586. Chem Biol 10:419–430
Eltringham I (1997) Mupirocin resistance and methicillin-resistant Staphylococcus aureus (MRSA). J Hosp Infect 35:1–8
Farmer TH, Gilbart J, Elson SW (1992) Biochemical basis of mupirocin resistance in strains of Staphylococcus aureus. J Antimicrob Chemother 30:587–596
Fawley WN, Parnell P, Hall J, Wilcox MH (2006) Surveillance for mupirocin resistance following introduction of routine peri-operative prophylaxis with nasal mupirocin. J Hosp Infect 62:327–332
Feline TC, Jones RB, Mellows G, Phillips L (1977) Pseudomonic acid. Part 2. Biosynthesis of pseudomonic acid A. J Chem Soc Perkin Trans I:309–318
Fleming A (1945) Penicillin. Nobel lecture
Fuller AT, Mellows G, Woolford M, Banks GT, Barrow KD, Chain EB (1971) Pseudomonic acid: an antibiotic produced by Pseudomonas fluorescens. Nature 234:416–417
Fuqua WC, Winans SC, Greenberg EP (1994) Quorum sensing in bacteria: the LuxR-LuxI family of cell density-responsive transcriptional regulators. J Bacteriol 176(2):269–275
Gilbart J, Perry CR, Slocombe B (1993) High-level mupirocin resistance in Staphylococcus aureus: evidence for two distinct isoleucyl-tRNA synthetases. Antimicrob Agents Chemother 37(1):32–38
Gilpin DF, Small S, Bakkshi S, Kearney MP, Cardwell C, Tunney MM (2010) Efficacy of a standard meticillin-resistant Staphylococcus aureus decolonisation protocol in routine clinical practice. J Hosp Infect 75:93–98
GlaxoSmithKlein (2010) Bactroban. http://public.gsk.co.uk/products/bactroban/. Accessed 9 August 2010
Gregory MA, Petkovic H, Lill RE, Moss SJ, Wilkinson B, Gaisser S, Leadlay PF, Sheridan RM (2005) Mutasynthesis of rapamycin analogues through the manipulation of a gene governing starter unit biosynthesis. Angew Chem Int Ed 44:4757–4760
Gupta S, Lakshmanan V, Kim BS, Fecik R, Reynolds KA (2008) Generation of novel pikromycin antibiotic products through mutasynthesis. Chembiochem 9:1609–1616
Hertweck C (2009) The biosynthetic logic of polyketide diversity. Angew Chem Int Ed 48:4688–4716
Hill AM, Staunton J (2010) Type I modular PKS. In: Mander L, Liu HW (eds) Comprehensive natural products II: chemistry and biology, vol 1. Oxford, Elsevier, pp 385–452
Hodgson JE, Curnock SP, Dyke KGH, Morris R, Sylvester DR, Gross MS (1994) Molecular characterization of the gene encoding high-level mupirocin resistance in Staphylococcus aureus J2870. Antimicrob Agents Chemother 38(5):1205–1208
Hothersall J, Wu JE, Rahman AS, Shields JA, Haddock J, Johnson N, Cooper SM, Stephens ER, Cox RJ, Crosby J, Willis CL, Simpson TJ, Thomas CM (2007) Mutational analysis reveals that all tailoring region genes are required for production of polyketide antibiotic mupirocin by Pseudomonas fluorescens. J Biol Chem 282(21):15451–15461
Hughes J, Mellows G (1978) Inhibition of isoleucyl-transfer ribonucleic acid synthetase in Escherichia coli by pseudomonic acid. Biochem J 179:305–318
Kopp M, Irschik H, Pradella S, Müller R (2005) Production of the tubulin destabilizer disorazol in Sorangium cellulosum: biosynthetic machinery and regulatory genes. Chembiochem 6:1277–1286
Lal R, Kumari R, Kaur H, Khanna R, Dhingra N, Tuteja D (2000) Regulation and manipulation of the gene clusters encoding type-I PKSs. Trends Biotechnol 18:264–274
Larkin M (2003) Daptomycin approved for skin and skin-structure infections. Lancet 3:677
Liu Q-Z, Wu Q, Zhang Y-B, Liu M-N, Hu F-P, Xu X-G, Zhu D-M, Ni Y-X (2010a) Prevalence of clinical meticillin-resistant Staphylococcus aureus (MRSA) with high-level mupirocin resistance in Shanghai and Wenzhou, China. Int J Antimicrob Agents 35:114–118
Liu TG, Vora H, Khosla C (2010b) Quantitative analysis and engineering of fatty acid biosynthesis in E. coli. Metab Eng 12:378–386
Lugtenberg BJJ, Bloemberg GV (2004) Life in the rhizospere. In: Ramos J-L (ed) Pseudomonas, vol 1. Kluwer Academic/Plenum Publishers, New York, pp 403–430
Magnuson K, Jackowski S, Rock C, John E, Cronan J (1993) Regulation of fatty acid biosynthesis in Escherichia coli. Microbiol Rev 57(3):522–542
Maier T, Leibundgut M, Ban N (2008) The crystal structure of a mammalian fatty acid synthase. Science 321:1315–1322
Marion O, Gao X, Marcus S, Hall DG (2009) Synthesis and preliminary antibacterial evaluation of simplified thiomarinol analogs. Bioorg Med Chem 17:1006–1017
Martin FM, Simpson TJ (1989) Biosynthetic studies on pseudomonic acid (mupirocin), a novel antibiotic metabolite of Pseudomonas fluorescens. J Chem Soc Perkin Trans I:207–209
Medimetriks (2008) Products. http://medimetriks.com/content/blogsection/6/36/. Accessed 9 August 2010
Mochizuki S, Hiratsu K, Suwa M, Ishii T, Sugino F, Yamada K, Kinashi H (2003) The large linear plasmid pSLA2-L of Streptomyces rochei has an unusually condensed gene organization for secondary metabolism. Mol Microbiol 48(6):1501–1510
Nakama T, Nureki O, Yokoyama S (2001) Structural basis for the recognition of isoleucyl-adenylate and an antibiotic, mupirocin, by isoleucyl-tRNA synthetase. J Biol Chem 276(50):47387–47393
Neu HC (1992) The crisis in antibiotic resistance. Science 257:1064–1073
Nguyen KT, Ritz D, Gu J-Q, Alexander D, Chu M, Miao V, Brian P, Baltz RH (2006) Combinatorial biosynthesis of novel antibiotics related to daptomycin. Proc Natl Acad Sci USA 103(46):17462–17467
Orrett FA (2008) The emergence of mupirocin resistance among clinical isolates of methicillin-resistant Staphylococcus aureus in Trinidad: a first report. Jpn J Infect Dis 61:107–110
Partida-Martinez LP, Hertweck C (2007) A gene cluster encoding rhizoxin biosynthesis in “Burkholderia rhizoxina”, the bacterial endosymbiont of the fungus Rhizopus microsporus. Chembiochem 8:41–45
Patel JB, Gorwitz R, Jernigan JA (2009) Mupirocin resistance. Clin Infect Dis 49:935–941
Perez-Roth E, Kwong SM, Alcoba-Florez J, Firth N, Mendez-Alvarez S (2010) Complete nucleotide sequence and comparative analysis of pPR9, a 41.7-kilobase conjugative staphylococcal multiresistance plasmid conferring high-level mupirocin resistance. Antimicrob Agents Chemother 54(5):2252–2257
Perlova O, Gerth K, Kaiser O, Hans A, Müller R (2006) Identification and analysis of the chivosazol biosynthetic gene cluster from the myxobacterial model strain Sorangium cellulosum So ce56. J Biotechnol 121:147–191
Piel J (2002) A polyketide synthase-peptide synthetase gene cluster from an uncultured bacterial symbiont of Paederus beetles. Proc Natl Acad Sci USA 99(22):14002–14007
Piel J (2010) Biosynthesis of polyketides by trans-AT polyketide synthases. Nat Prod Rep 27:996–1047
Prescott LM, Harley JP, Klein DA (2005) Microbiology, 6th edn. McGraw-Hill, New York
Pulsawat N, Kitani S, Nihira T (2007) Characterization of biosynthetic gene cluster for the production of virginiamycin M, a streptogramin type A antibiotic, in Streptomyces virginiae. Gene 393:31–42
Rahman M, Noble WC, Cookson B (1987) Mupirocin-resistant Staphylococcus aureus. Lancet 330(8555):377–378
Rahman AS, Hothersall J, Crosby J, Simpson TJ, Thomas CM (2005) Tandemly duplicated acyl carrier proteins, which increase polyketide antibiotic production, can apparently function either in parallel or in series. J Biol Chem 280(8):6399–6408
Ramsey MA, Bradley SF, Kauffman CA, Morton TM (1996) Identification of chromosomal location of mupA gene, encoding low-level mupirocin resistance in staphylococcal isolates. Antimicrob Agents Chemother 40(12):2820–2823
Reeves CD, Murli S, Ashley GW, Piagentini M, Hutchinson CR, McDaniel R (2001) Alteration of the substrate specificity of a modular polyketide synthase acyltransferase domain through site-specific mutations. Biochemistry 40(51):15464–15470
Ridley CP, Lee HY, Khosla C (2008) Evolution of polyketide synthases in bacteria. PNAS 105(12):4595–4600
Schneider K, Chen X-H, Vater J, Franke P, Nicholson G, Borriss R, Süssmuth RD (2007) Macrolactin is the polyketide biosynthesis product of the pks2 cluster of Bacillus amyloliquefaciens FZB42. J Nat Prod 70:1417–1423
Schujman GE, Dd M (2008) Regulation of type II fatty acid synthase in Gram-positive bacteria. Curr Opin Microbiol 11:148–152
Shiozawa H, Kagasaki T, Kinoshita T, Haruyama H, Domon H, Utsui Y, Kodama K, Takahashi S (1993) Thiomarinol, a new hybrid antimicrobial antibiotic produced by a marine bacterium. J Of Antibiot 46(12):1834–1842
Simunovic V, Zapp J, Rachid S, Krug D, Meiser P, Müller R (2006) Myxovirescin A biosynthesis is directed by hybrid polyketide synthases/nonribosomal peptide synthetase, 3-hydroxy-3-methylglutaryl–CoA synthases, and trans-acting acyltransferases. Chembiochem 7:1206–1220
Slocombe B, Perry C (1991) The antimicrobial activity of mupirocin—an update on resistance. J Hosp Infect 19(Supp2):19–25
Sudek S, Lopanik NB, Waggoner LE, Hildebrand M, Anderson C, Liu H, Patel A, Sherman DH, Haygood MG (2007) Identification of the putative bryostatin polyketide synthase gene cluster from “Candidatus Endobugula sertula”, the uncultivated microbial symbiont of the marine bryozoan Bugula neritina. J Nat Prod 70(1):67–74
Sutherland R, Boon RJ, Griffin KE, Masters PJ, Slocombe B, White AR (1985) Antibacterial activity of mupirocin (pseudomonic acid), a new antibiotic for topical use. Antimicrob Agents Chemother 27(4):495–498
Tang G-L, Cheng Y-Q, Shen B (2004a) Leinamycin biosynthesis revealing unprecedented architectural complexity for a hybrid polyketide synthase and nonribosomal peptide synthetase. Chem Biol 11:33–45
Tang Y, Lee TS, Lee HY, Khosla C (2004b) Exploring the biosynthetic potential of bimodular aromatic polyketide synthases. Tetrahedron 60:7659–7671
TEVA (2003) Press Release: Teva Announces Approval Of Mupirocin Ointment http://www.tevapharm.com/pr/2003/pr_421.asp. Accessed 9 August 2010
Torvaldsen S, Roberts C, Riley TV (1999) The continuing evolution of methicillin-resistant Staphylococcus aureus in Western Australia. Infect Control Hosp Epidemiol 20(2):133–135
Udo EE, Farook VS, Mokadas EM, Jacob LE, Sanyal SC (1999) Molecular fingerprinting of mupirocin-resistant methicillin-resistant Staphylococcus aureus from a burn unit. Int J Infect Dis 3(2):82–87
Upton A, Lang S, Heffernan H (2003) Mupirocin and Staphylococcus aureus: a recent paradigm of emerging antibiotic resistance. J Antimicrob Chemother 51:613–617
Vasquez JE, Walker ES, Franzus BW, Overbay BK, Reagan DR, Sarubbi FA (2000) The epidemiology of mupirocin resistance among methicillin-resistant Staphylococcus aureus at a Veterans’ Affairs Hospital. Infect Control Hosp Epidemiol 21(7):459–464
Weber T, Laiple KJ, Pross EK, Textor A, Grond S, Welzel K, Pelzer S, Vente A, Wohlleben W (2008) Molecular analysis of the kirromycin biosynthetic gene cluster revealed beta-alanine as precursor of the pyridone moiety. Chem Biol 15:175–188
Whatling C, Hodgson JE, Burnham MKR, Clarke NJ, Franklin CH, Thomas CM (1995) Identification of a 60 kb region of the chromosome of Pseudomonas fluorescens NCIB 10586 required for the biosynthesis of pseudomonic acid (mupirocin). Microbiology 141:973–982
Wu J, Cooper SM, Cox RJ, Crosby J, Crump MP, Hothersall J, Simpson TJ, Thomas CM, Willis CL (2007) Mupirocin H, a novel metabolite resulting from mutation of the HMG-CoA synthase analogue, mupH in Pseudomonas fluorescens. Chem Commun 2007:2040–2042
Wu J, Hothersall J, Mazzetti C, O'Connell Y, Shields JA, Rahman AS, Cox RJ, Crosby J, Simpson TJ, Thomas CM, Willis CL (2008) In vivo mutational analysis of the mupirocin gene cluster reveals labile points in the biosynthetic pathway: the “leaky hosepipe” mechanism. Chembiochem 9:1500–1508
Wuite J, Davies BI, Go M, Lambers J, Jackson D, Mellows G (1983) Pseudomonic acid: a new topical antimicrobial agent. Lancet 322(8346):394
Xie X, Watanabe K, Wojcicki WA, Wang CCC, Tang Y (2006) Biosynthesis of lovastatin analogs with a broadly specific acyltransferase. Chem Biol 13:1161–1169
Yadav G, Gokhale RS, Mohanty D (2003) Computational approach for prediction of domain organisation and substrate specificity of modular polyketide synthases. J Mol Biol 328:335–363
Yanagisawa T, Kawakami M (2003) How does Pseudomonas fluorescens avoid suicide from its antibiotic pseudomonic acid? J Biol Chem 278(28):25887–25894
Yanagisawa T, Lee JT, Wu HC, Kawakami M (1994) Relationship of protein structure of isoleucyl-tRNA synthetase with pseudomonic acid resistance of Escherichia coli. J Biol Chem 269(39):24304–24309
Acknowledgments
The work on mupirocin and thiomarinol being carried out in the authors' lab is funded by UK Research Councils BBSRC and EPSRC under grant number E021611. RG is funded by a BBSRC Doctoral Training Centre Studentship. We are grateful to Joanne Hothersall for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gurney, R., Thomas, C.M. Mupirocin: biosynthesis, special features and applications of an antibiotic from a Gram-negative bacterium. Appl Microbiol Biotechnol 90, 11–21 (2011). https://doi.org/10.1007/s00253-011-3128-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3128-3